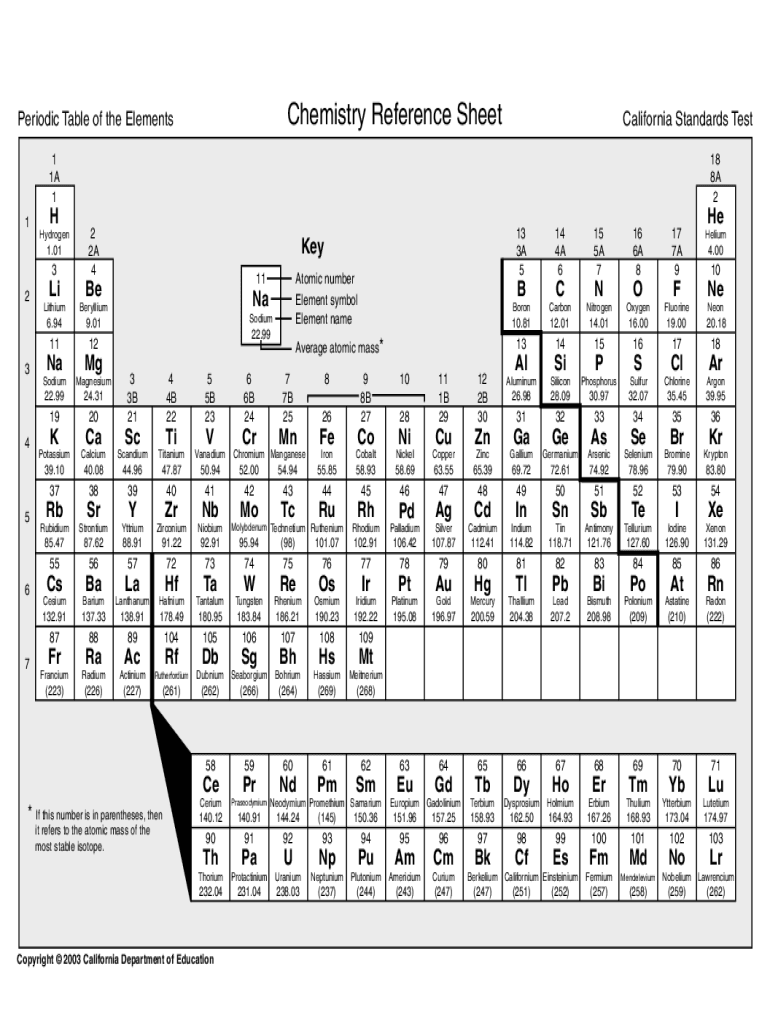
Get the free chemistry reference sheet periodic table form
Get, Create, Make and Sign

Editing chemistry reference sheet periodic table online
How to fill out chemistry reference sheet periodic
How to fill out a chemistry reference sheet periodic:
Who needs chemistry reference sheet periodic:
Video instructions and help with filling out and completing chemistry reference sheet periodic table
Instructions and Help about periodic table of elements california standards test form
Calcium is the third element in group two on the periodic table. It’s really very common. We find it all over the world in all sorts of minerals. So for example you can have bones, this is a pelvis of a sheep, I found it when I was walking one day, you can see the legs go in there and there. In here we have some calcium turnings. We’re storing them in our glove box. This glove box here is a contraption full of nitrogen, it’s dry nitrogen so that we can keep our calcium away from oxygen and away from moisture as well, because obviously what we don’t want is when we get our nice very reactive calcium surface we don’t want it to tarnish from oxides too quickly before we can react it with things. So what we have here inside the bottle are turnings of calcium. Calcium is quite hard compared to sort of the heavier group two elements, so strontium and barium, so it’s harder to file. Bones are made from calcium phosphate mixed with some organic material as well, and they’re very strong because of the structure within the bones. There are all sorts of shells of mollusks and the like where they use calcium carbonate from the seawater, getting the calcium out, reacting with carbon dioxide and making their shells. And because shells are made out of calcium carbonate, which dissolves in acid, people are quite worried about the effects of global warming. Not the heating up but when the C02 dissolves into the sea it makes it more acid, so in principle shells could start dissolving and some mollusks may not be able to survive because they just can’t make their shells. If I just pour them out, so you can see. Calcium looks metallic than the calcium metal itself because of the way the electrons behave in the solid. In fact, it is because it is a very good electrical conductor, but the compounds are all white, because calcium has no free electrons that can go from one energy level to another in the way you can say with copper. So copper sulfate is a beautiful blue color, calcium sulfate is white, and it’s actually colorless, so if you see a big crystal of calcium sulfate it just looks like glass. But the white is caused by very small crystals which scatter the light, just in the same way that any sort of white powder will scatter light, flour for example or talcum powder, anything like that. Calcium compounds themselves are actually mostly white, but calcium metal itself, as you can see we’ve got tarnished parts, but also there are some shiny bits where it’s untarnished. So you’re actually seeing the metal the luster of the metal there as well. Calcium carbonate dissolves in water saturated with C02 to make calcium bicarbonate which is more soluble. And so you can get water, so-called hard water, that has quite high levels of calcium. And when this percolates through the ground and comes to a cave it can then crystallize out, and you can get hanging down pillars of calcium carbonate, which are called stalactites. And you can have pillars growing from the floor...
Fill chemistry reference sheet periodic table : Try Risk Free
People Also Ask about chemistry reference sheet periodic table
For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
Fill out your chemistry reference sheet periodic online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.