Stamp Digital Sign Clinical Trial Agreement Template For Free




Users trust to manage documents on pdfFiller platform
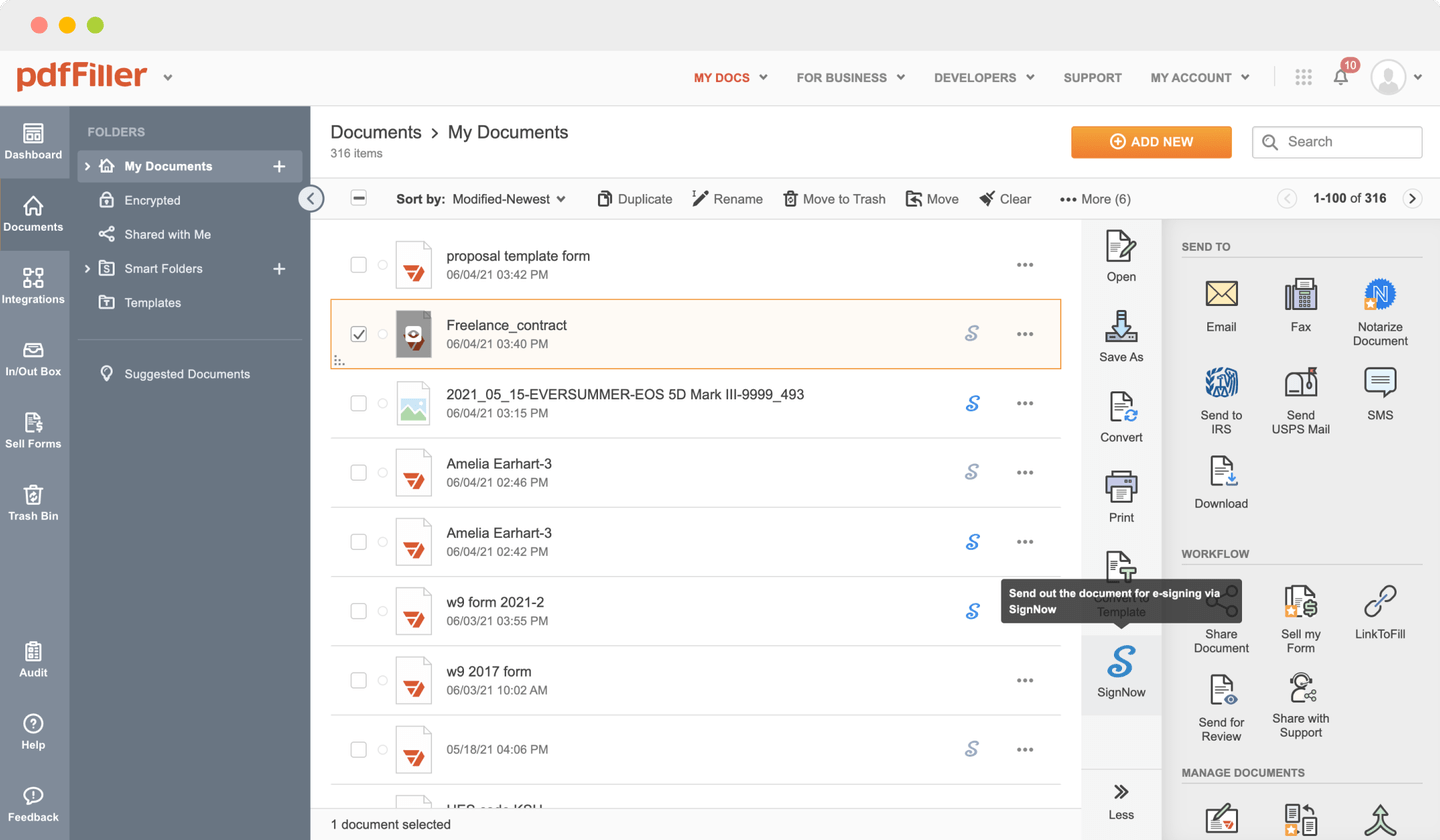
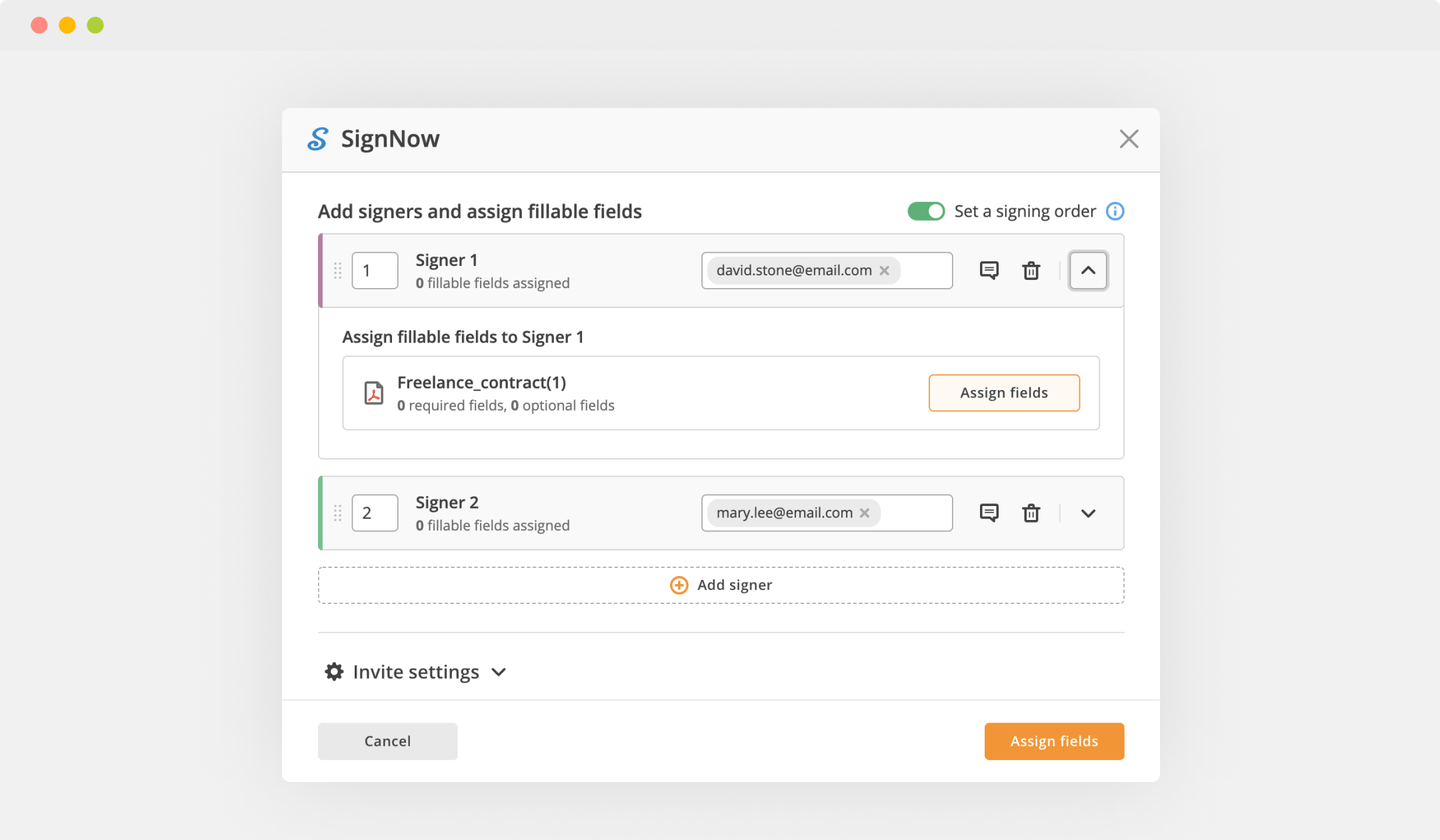
Send documents for eSignature with signNow
Watch a quick video tutorial on how to Stamp Digital Sign Clinical Trial Agreement Template
pdfFiller scores top ratings in multiple categories on G2
Stamp Digital Sign Clinical Trial Agreement Template in minutes
pdfFiller allows you to Stamp Digital Sign Clinical Trial Agreement Template quickly. The editor's handy drag and drop interface allows for fast and intuitive document execution on any device.
Ceritfying PDFs online is a quick and safe method to verify papers at any time and anywhere, even while on the fly.
Go through the detailed instructions on how to Stamp Digital Sign Clinical Trial Agreement Template electronically with pdfFiller:
Upload the form you need to sign to pdfFiller from your device or cloud storage.
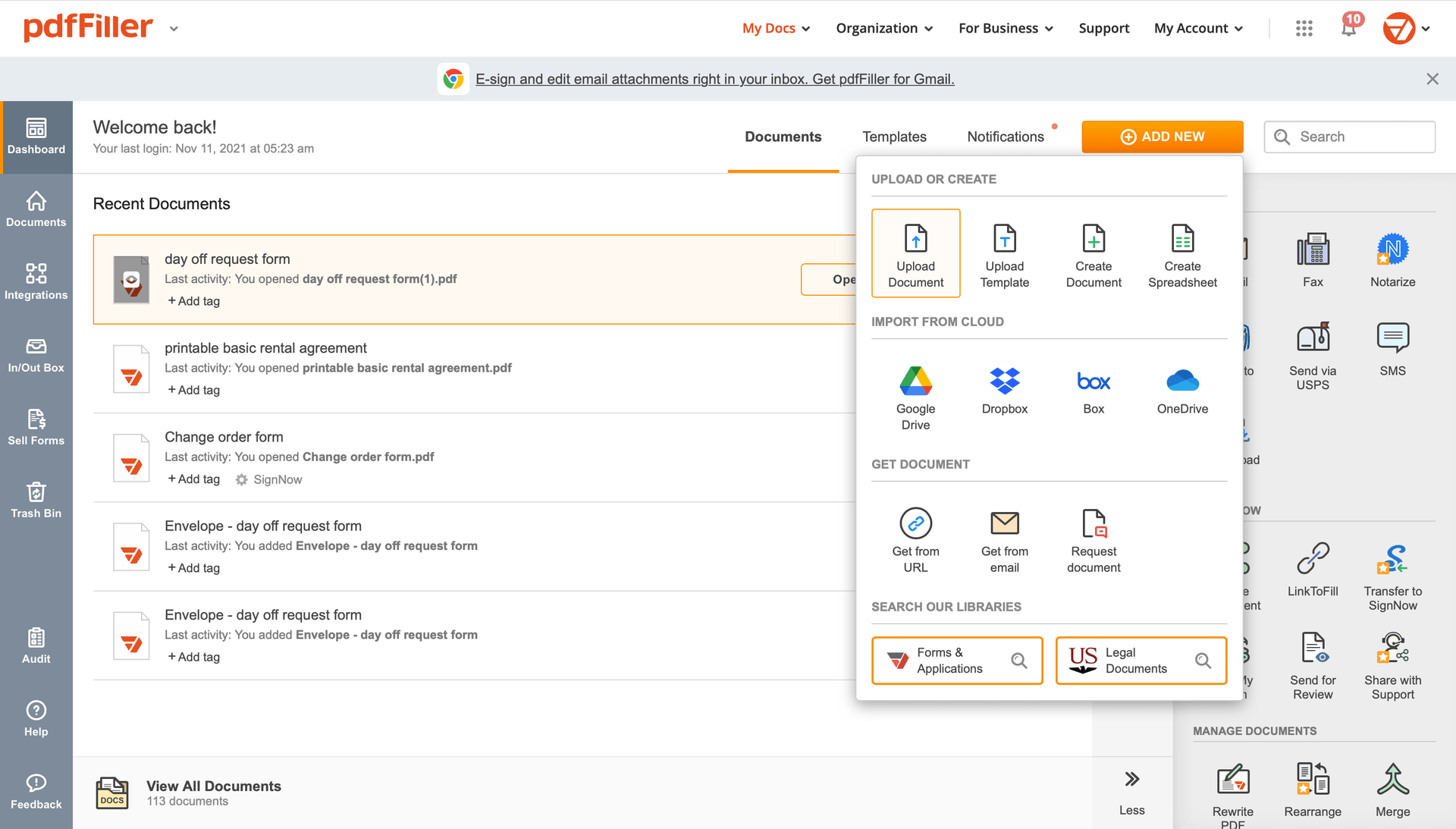
Once the document opens in the editor, click Sign in the top toolbar.

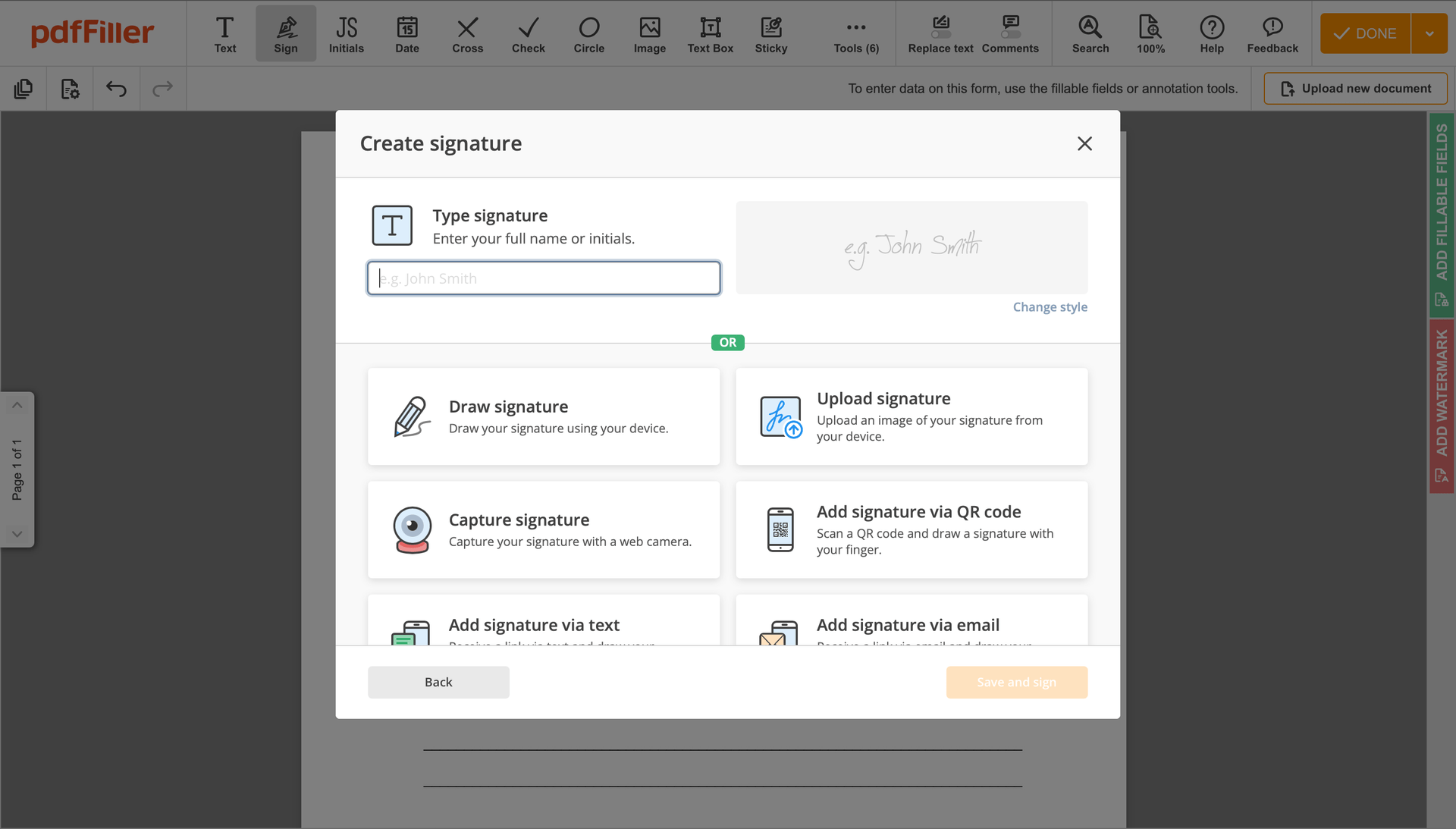
Create your electronic signature by typing, drawing, or adding your handwritten signature's photo from your laptop. Then, click Save and sign.
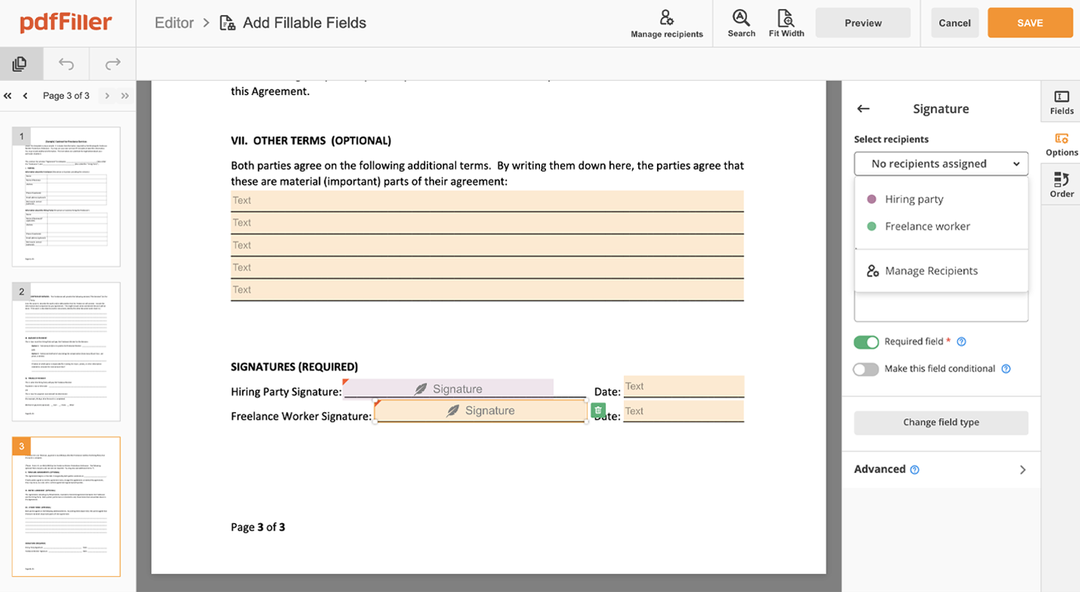
Click anywhere on a form to Stamp Digital Sign Clinical Trial Agreement Template. You can move it around or resize it using the controls in the floating panel. To apply your signature, hit OK.
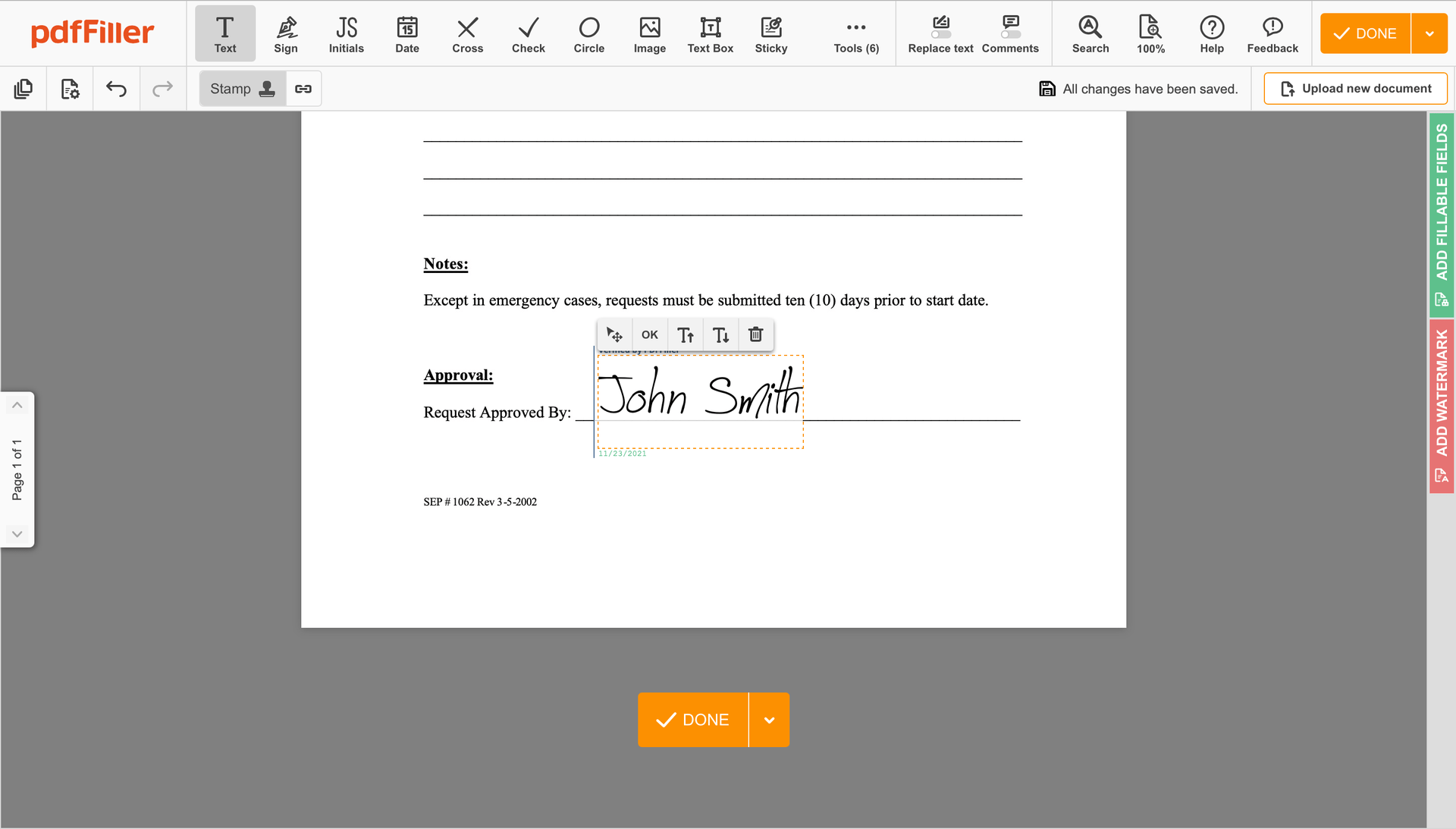
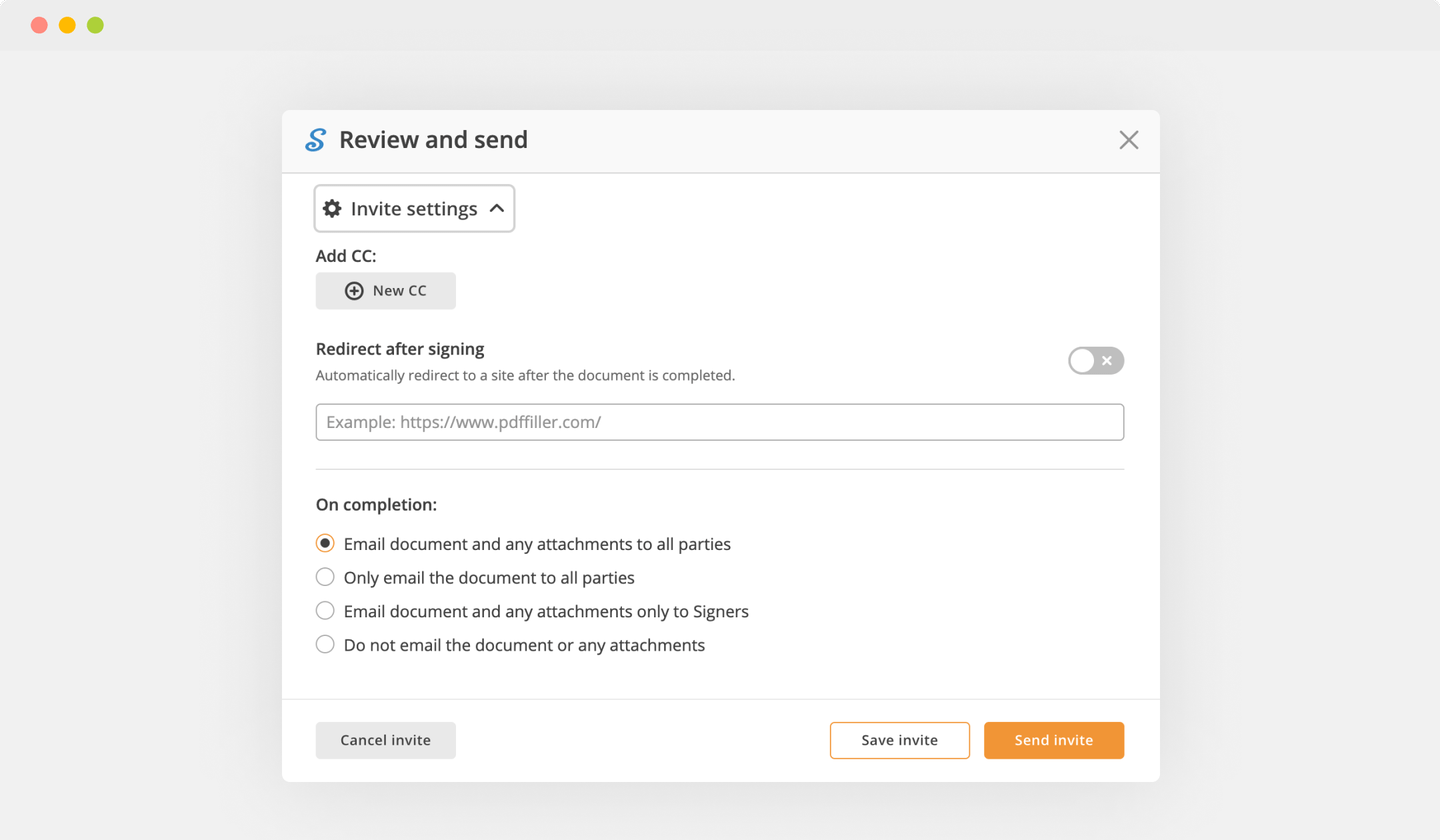
Complete the signing process by hitting DONE below your form or in the top right corner.
Next, you'll return to the pdfFiller dashboard. From there, you can get a completed copy, print the form, or send it to other parties for review or approval.
Still using different applications to sign and manage your documents? Use our solution instead. Use our document editor to make the process simple. Create forms, contracts, make templates and many more features, within your browser. You can Stamp Digital Sign Clinical Trial Agreement Template with ease; all of our features are available to all users. Have an advantage over those using any other free or paid applications. The key is flexibility, usability and customer satisfaction. We deliver on all three.
How to edit a PDF document using the pdfFiller editor:
How to Send a PDF for eSignature
What our customers say about pdfFiller