Last updated on May 3, 2026

Immunize P3039F 2011-2026 free printable template
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Immunize P3039F
The Vaccine Storage Temperature Log is a healthcare form used by medical facilities to monitor and record temperatures of vaccine storage units, ensuring compliance with storage guidelines.
pdfFiller scores top ratings on review platforms




PDFiller is easy to use and it allows me to delete pages; Adobe Sign does not allow me to delete pages while using their platform.
Pretty good so far just trying to learn all the ins and outs of the program.
Who needs Immunize P3039F?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Immunize P3039F
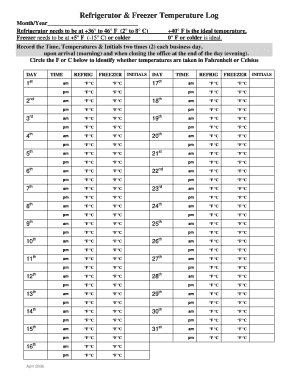
What is the Vaccine Storage Temperature Log?
The Vaccine Storage Temperature Log is an essential tool for healthcare settings, designed to maintain the integrity of vaccine storage conditions. This log serves as a record-keeping mechanism to ensure that vaccines are stored at the appropriate temperatures, critical for their efficacy and safety.
Adapted by the Immunization Action Coalition and reviewed by the CDC, it plays a vital role in monitoring vaccine storage compliance. This form is used consistently to track temperature data in refrigerators and freezers, ensuring that healthcare providers adhere to set guidelines.
Purpose and Benefits of Using the Vaccine Storage Temperature Log
Utilizing the Vaccine Storage Temperature Log is crucial for maintaining vaccine potency and safety. By ensuring proper documentation, healthcare providers can avoid potential losses due to temperature excursions.
-
This log aids in regular monitoring, aligning with vaccine storage guidelines and improving overall compliance.
-
Accurate records can also help healthcare facilities mitigate legal risks associated with improper vaccine handling.
Key Features of the Vaccine Storage Temperature Log
The Vaccine Storage Temperature Log is designed with user-friendly components to facilitate effective monitoring. Users will find fillable fields for recording temperature readings along with staff initials, ensuring accountability and accuracy.
-
Instructions are provided for managing situations where temperatures fall outside the acceptable range.
-
All completed forms must be retained for a minimum of three years, ensuring proper documentation for oversight and audits.
Who Needs the Vaccine Storage Temperature Log?
This log is vital for various stakeholders involved in vaccine management. Healthcare providers, including clinics, hospitals, and pharmacies, are primarily responsible for ensuring that their vaccine storage practices meet regulatory standards.
-
Any facility that stores vaccines must utilize the log to maintain compliance.
-
Those involved in vaccine management and storage benefit from using this essential healthcare form template.
How to Fill Out the Vaccine Storage Temperature Log Online (Step-by-Step)
Filling out the Vaccine Storage Temperature Log online is a straightforward process. Here’s how users can complete the log accurately:
-
Access the online form and enter the current date and time of the temperature reading.
-
Record the temperature value in the designated field and include your initials for verification.
-
Follow the instructions provided for any out-of-range temperatures and document the action taken.
-
Save the completed form in a secure location as required for record-keeping.
Common Errors and How to Avoid Them
Identifying typical errors in completing the Vaccine Storage Temperature Log can help ensure more accurate records. Frequent mistakes include incorrect temperature entries and missing initials.
-
Double-check all entries before finalizing the submission to minimize errors.
-
Providing training for staff on the proper use of the log can significantly reduce mistakes.
Submission Methods for the Vaccine Storage Temperature Log
Users can submit the completed Vaccine Storage Temperature Log through various methods. This flexibility allows for ease of compliance and record-keeping.
-
Submission can occur digitally via secure platforms or through physical delivery to relevant authorities.
-
It is crucial to adhere to submission deadlines to ensure compliance with healthcare regulations.
Privacy and Data Protection for Vaccine Storage Records
Ensuring the privacy and security of vaccine storage records is paramount. The Vaccine Storage Temperature Log must comply with HIPAA regulations to protect sensitive data.
-
Implementing robust security measures in handling these logs helps maintain compliance.
-
Features offered by services like pdfFiller enhance overall data protection during log completion.
How pdfFiller Simplifies the Process of Using the Vaccine Storage Temperature Log
pdfFiller provides a comprehensive solution for individuals completing the Vaccine Storage Temperature Log. As a cloud-based platform, it offers numerous advantages for users.
-
Editing and filling the log is streamlined through its intuitive interface.
-
eSigning capabilities further enhance workflow efficiency, allowing for 'virtual' signatures as needed.
Finalize Your Vaccine Storage Temperature Log Easily with pdfFiller
Leveraging pdfFiller for your Vaccine Storage Temperature Log can simplify the documentation process. The platform's user-friendly tools ensure that users can efficiently complete their logs while maintaining accuracy.
-
The investment in pdfFiller's features ultimately leads to better compliance and easier management of records.
-
Utilizing this platform guarantees support for users throughout the form completion process.
How to fill out the Immunize P3039F
-
1.Begin by accessing pdfFiller and navigating to the Vaccine Storage Temperature Log form. You can search for the form using the search bar or locate it in the healthcare forms section.
-
2.Once you open the form, familiarize yourself with the layout. The document includes multiple blank fields where you will enter temperature readings, your initials, and the date of the readings.
-
3.Before filling out the form, collect the necessary information, including the current temperature readings of the refrigerator or freezer and any specific times you need to record these values.
-
4.Start by entering the initial temperature reading in the designated field. Use the fillable text box to input the temperature accurately. Make sure to pay attention to the measurement units, usually in degrees Celsius or Fahrenheit.
-
5.Next, document the time of the reading. Locate the time field and enter the relevant time for each reading. This provides a clear record of monitoring throughout the day.
-
6.Don’t forget to initial the form in the corresponding fields to indicate you, or the responsible staff member, conducted the readings. This adds accountability to the temperature log.
-
7.Once you have filled out all required fields, review the completed form for any errors or omissions. Ensure all entries are accurate, and cross-check with your collected data.
-
8.To finalize the form, look for the save option within pdfFiller. You can either save it directly to your account, download it as a PDF, or use the submit feature to send it directly to your intended recipient.
-
9.If needed, you may also print a hard copy of the form for your records. Ensure you keep copies of all completed logs for compliance, as per guidelines.

Who is required to complete the Vaccine Storage Temperature Log?
Healthcare providers, pharmacy staff, and clinics that store vaccines are typically required to complete this log. It helps maintain proper vaccine storage and ensure safety.
What should I do if the recorded temperatures are out of range?
If temperatures are outside acceptable ranges, refer to the handling instructions provided with the form. Follow the steps outlined to report and address temperature excursions.
How long should I retain the completed forms?
You must keep completed Vaccine Storage Temperature Logs for a minimum of three years. This is crucial for compliance and safety checks.
Are there specific deadlines for submitting the Vaccine Storage Temperature Log?
While there are no standard deadlines, it is essential to complete the log daily. Regular monitoring helps to ensure proper vaccine effectiveness and safety.
What common mistakes should I avoid when filling out the log?
Avoid leaving fields blank, especially for time and initials. Ensure accuracy in temperature readings and check the form for correct entries before finalizing it.
Can I fill out the Vaccine Storage Temperature Log electronically?
Yes, you can fill out the Vaccine Storage Temperature Log electronically using pdfFiller. This provides ease of completion and saves time while maintaining accurate records.
Is notarization required for this form?
No, notarization is not required for the Vaccine Storage Temperature Log. It is a straightforward record-keeping document used internally.
Related Catalogs
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.