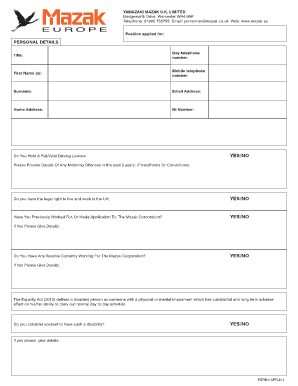
Last updated on Nov 12, 2014

Get the free Research Summary for IRB Application
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is IRB Research Summary
The Research Summary for IRB Application is a document used by researchers at Ashford University to outline their study for Institutional Review Board (IRB) review and ensure ethical research standards.
pdfFiller scores top ratings on review platforms




Who needs IRB Research Summary?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to IRB Research Summary
What is the Research Summary for IRB Application?
The Research Summary for IRB Application is a critical form utilized by researchers aimed at achieving compliance within ethical research practices. This form plays a significant role in the Institutional Review Board (IRB) application process, ensuring that proposed studies are ethically sound. Key sections included in the form address the purpose, methodology, risks and benefits, and informed consent processes.
This form is primarily used by both researchers and subjects, ensuring that all parties are informed about the study’s framework and ethical commitments. Its structured design helps maintain clarity throughout the research process, emphasizing the need for a thorough understanding of ethical standards in research.
Purpose and Benefits of the Research Summary for IRB Application
The Research Summary serves to explain the significance of adhering to ethical standards in human subjects research. This form is essential in ensuring that researchers uphold ethical principles and institutional compliance throughout their studies. By utilizing this form, researchers can clearly outline their methodologies, enhancing transparency and protection for both participants and researchers.
Adhering to IRB approval criteria not only fosters trust between researchers and participants but also reinforces the overall integrity of the research conducted. The clarity provided by this document significantly lowers risks associated with unethical practices, benefiting all stakeholders involved.
Key Features of the Research Summary for IRB Application
This form consists of several major sections critical for a well-rounded submission. Each section includes:
-
Purpose and significance of the study
-
Methodology employed in the research
-
Assessment of risks and benefits
-
Procedures for subject recruitment
-
The informed consent process
The fillable format streamlines the application process, allowing for easy completion and review. Additionally, it mandates signatures from both researchers and subjects, ensuring all parties acknowledge their roles and responsibilities in the study.
Who Needs the Research Summary for IRB Application?
The primary users of the Research Summary include researchers and research subjects. Researchers play a critical role in detailing their proposed studies, while subjects contribute vital information about their participation. Eligibility requirements for this form apply to various academic settings, including those specific to Ashford University.
Understanding the responsibilities of each role ensures compliance with IRB protocols, which helps in the ethical management of research involving human subjects.
How to Fill Out the Research Summary for IRB Application Online (Step-by-Step)
To successfully complete the Research Summary, follow these essential steps:
-
Gather necessary information using the pre-filing checklist.
-
Complete each section of the form systematically, ensuring clarity.
-
Review for common errors, such as incomplete fields or lack of signatures.
-
Submit the completed form as directed.
By adhering to these steps, users can mitigate potential pitfalls while ensuring that their submissions are accurate and well-prepared.
Submission Methods and What Happens After You Submit
Once you complete the Research Summary, you can submit it using various methods, including online and offline options. After submission, users can expect to receive confirmation of receipt and information regarding processing times.
Tracking options are available to monitor the status of the IRB application. Understanding these procedures is vital to ensure timely responses and approvals.
Security and Compliance When Using the Research Summary for IRB Application
Data security and compliance are paramount when handling sensitive information through the Research Summary. pdfFiller employs robust measures to ensure the secure handling of documents, complying with regulations like HIPAA and GDPR.
Maintaining privacy throughout the research process is essential. Researchers must be diligent in safeguarding participant data, ensuring a trustworthy research environment.
How pdfFiller Facilitates the Research Summary for IRB Application
pdfFiller assists users in completing the Research Summary with a variety of user-friendly features. These include the ability to edit text, fill in fields, and eSign documents seamlessly.
Furthermore, pdfFiller allows users to save, share, and track their submissions efficiently. Leveraging cloud-based services enhances accessibility for researchers, providing a streamlined workflow for managing research documentation.
Final Thoughts on the Research Summary for IRB Application
Utilizing pdfFiller for completing the Research Summary can simplify the IRB application process. With its intuitive platform and strong security measures, researchers are encouraged to begin their IRB submissions with confidence.
Exploring other related forms and resources available through pdfFiller can further support ethical research endeavors and compliance with institutional guidelines.
How to fill out the IRB Research Summary
-
1.To access the Research Summary for IRB Application, visit pdfFiller and search for the form by name.
-
2.Once located, click on the form to open it in the pdfFiller editor.
-
3.Begin by thoroughly reviewing the form to familiarize yourself with its sections.
-
4.Gather necessary information, such as study purpose, methodology, and details on risks and benefits.
-
5.Use the fillable fields to enter information relevant to your research proposal as prompted.
-
6.Utilize the tips and instructions provided within the form to ensure you provide complete and accurate information.
-
7.Once you’ve filled out all necessary fields, review the form for any missing information or errors.
-
8.After verification, save your changes in pdfFiller to ensure your progress is not lost.
-
9.You can choose to download the completed form for your records or submit it directly through pdfFiller if required.

Who is eligible to complete the Research Summary for IRB Application?
Eligibility is primarily for researchers at Ashford University who are conducting studies involving human subjects and require IRB approval.
What is the submission procedure for this form?
The form can be submitted electronically through pdfFiller, or by downloading and sending it directly to the Institutional Review Board for review.
What supporting documents are required with the form?
While specific supporting documents are not listed, typically, additional information about your research methodology and consent procedures may be required.
What are common mistakes to avoid when filling this form?
Common mistakes include skipping sections, providing vague information, and failing to gather required data before initiating the form.
How long does it take to process the Research Summary for IRB Application?
Processing times can vary; typically, allow several weeks for review by the IRB. It's advisable to submit early to accommodate feedback or revisions.
Can students complete this form without faculty guidance?
While students can complete the form independently, it's highly recommended to consult with faculty advisors to ensure all research proposals meet ethical guidelines.
Is notarization required for this form?
No, notarization is not required for the Research Summary for IRB Application as per the metadata provided.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.