Last updated on Mar 23, 2026
VA P3039A free printable template
pdfFiller is not affiliated with any government organization
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is VA P3039A
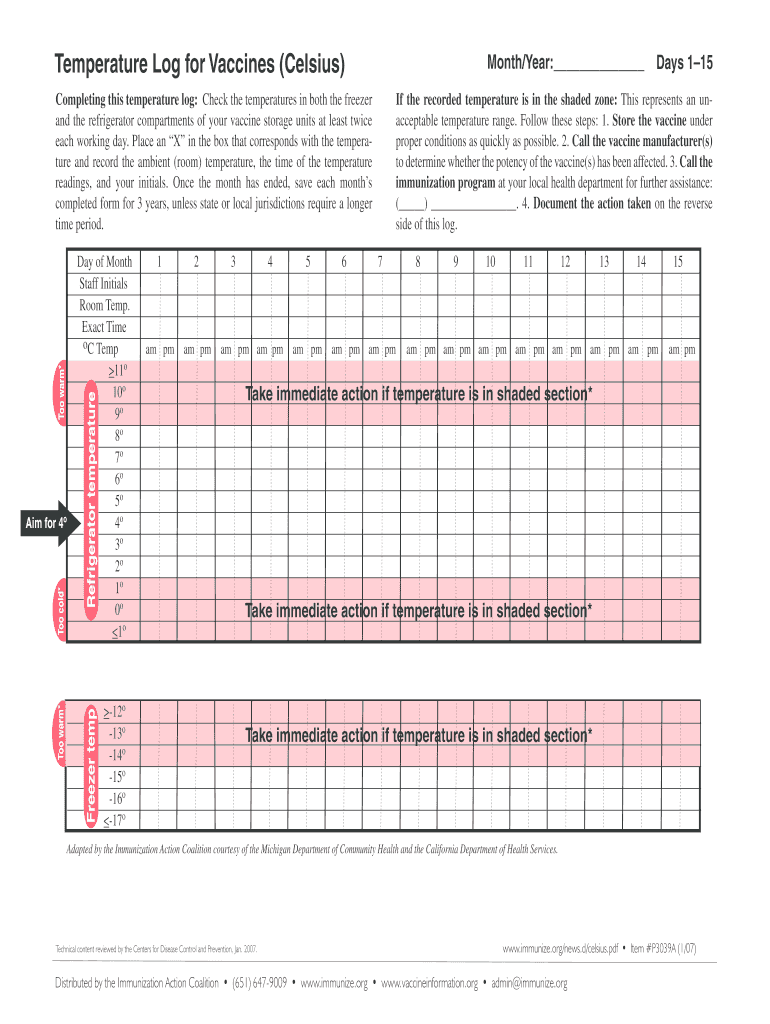
The Vaccine Temperature Log Form is a healthcare document used by providers to monitor and record the temperature of vaccine storage units.
pdfFiller scores top ratings on review platforms




Who needs VA P3039A?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to VA P3039A
What is the Vaccine Temperature Log Form?
The Vaccine Temperature Log Form is essential for healthcare providers to monitor and record the temperature of vaccine storage units. This form is crucial in ensuring that vaccines remain effective by documenting temperatures for both freezer and refrigerator compartments. It helps maintain proper storage conditions, which is vital for vaccine efficacy and safety.
Monitoring temperature is not just a procedural requirement; it directly impacts vaccine potency. Proper documentation allows for timely interventions if temperatures fall outside acceptable ranges, minimizing potential risks to vaccine effectiveness.
Why is Proper Temperature Monitoring Essential for Vaccines?
Temperature significantly affects the effectiveness of vaccines. When vaccines are stored outside their recommended temperature ranges, their efficacy can be compromised, leading to ineffective immunizations. Various health authorities have established guidelines to ensure vaccine safety, which require strict compliance with temperature monitoring protocols.
Improper storage can jeopardize public health initiatives and lead to unnecessary health risks. Adherence to temperature logging practices not only fulfills legal obligations but also safeguards the integrity of vaccinations administered to patients.
Who Needs the Vaccine Temperature Log Form?
The Vaccine Temperature Log Form is vital for a range of users in the healthcare sector. Primarily, healthcare providers and organizations responsible for storing vaccines must utilize this form to ensure proper temperature monitoring. Additionally, pharmacies and clinics that administer vaccinations also need to maintain accurate logs.
Educational institutions with vaccination programs are another key group that benefit from the Vaccine Temperature Log Form, ensuring their compliance with health standards.
How to Fill Out the Vaccine Temperature Log Form Online (Step-by-Step)
Filling out the Vaccine Temperature Log Form is a straightforward process. Follow these steps:
-
Access the form and input daily temperature readings for both freezer and refrigerator.
-
Initial each entry to confirm the data accuracy and indicate who conducted the temperature check.
-
Document any actions taken if temperatures are outside acceptable ranges, such as notifications to supervisors or health authorities.
-
Review all entries for completeness and accuracy before saving the completed form.
-
Save the form as a PDF for electronic records or print it for physical retention.
Key Features of the Vaccine Temperature Log Form
The Vaccine Temperature Log Form includes several critical features that enhance its usability and compliance. Users will find fillable fields designed for recording temperature data, dates, and staff initials for accountability. Clear instructions are provided to guide users through the completion process.
Additionally, the form specifies the required actions to take when temperatures are outside acceptable limits, emphasizing the importance of thorough record retention for compliance with health regulations.
Understanding Temperature Ranges and Required Actions
Familiarizing yourself with acceptable temperature ranges is essential for effective vaccine storage. Vaccines typically require specific temperature settings to maintain their potency, often within a range of 2°C to 8°C for refrigeration and below -15°C for freezers.
If temperatures deviate from these ranges, immediate actions must be taken. These actions include:
-
Reassessing the storage unit's functionality.
-
Taking corrective measures to restore appropriate temperatures.
-
Documenting all actions taken and communications made.
State-Specific Requirements and Compliance
In addition to general guidelines, various state-specific regulations may govern vaccine storage and temperature logging. It is imperative for healthcare providers to be aware of their local jurisdiction's requirements to ensure compliance with laws regarding record retention and monitoring.
Healthcare organizations should regularly consult local health department resources or professional networks to stay updated on any changes to regulations regarding temperature logging.
Security and Privacy in Handling Vaccine Temperature Logs
Maintaining the security and privacy of vaccine temperature logs is crucial. Utilizing platforms such as pdfFiller ensures that sensitive data is protected through advanced encryption methods compliant with HIPAA and GDPR. It's vital to implement best practices for securely sharing and storing these logs.
Users should prioritize confidentiality and data protection when managing documentation related to vaccine storage temperatures.
How to Save, Share, and Submit the Vaccine Temperature Log Form
Once you complete the Vaccine Temperature Log Form, saving and sharing it effectively is essential. Here’s how to do it:
-
Download the completed form as a PDF to your device.
-
Share the form securely with stakeholders or health authorities via email or designated platforms.
-
Submit the form according to your organization’s preferred methods, whether electronically or in a physical format.
Maximize Efficiency with pdfFiller: Your Go-To Tool for Forms
pdfFiller provides a comprehensive solution for managing the Vaccine Temperature Log Form. Its capabilities include editing, signing, and sharing forms seamlessly through a cloud-based interface. This streamlined approach enhances user efficiency and helps ensure compliance with temperature documentation regulations.
Getting started with pdfFiller empowers organizations to effectively manage their temperature logs, ensuring higher standards of care and compliance.
How to fill out the VA P3039A
-
1.To begin, access the Vaccine Temperature Log Form on pdfFiller by searching for the form name in the template library.
-
2.Once you locate the form, open it in pdfFiller's editor interface where you can see all the fields you need to complete.
-
3.Before filling the form, gather necessary information such as the daily temperature readings and the staff initials for verification.
-
4.Navigate through the form fields to enter the current temperature for both the freezer and refrigerator compartments, ensuring accuracy.
-
5.Fill in the required fields, including spaces for any actions taken if the recorded temperature is outside the acceptable range.
-
6.Review all the entered information for completeness and accuracy, making any necessary corrections as you go.
-
7.Once satisfied with the information, finalize the form by saving or downloading it in your preferred format.
-
8.If required, submit your completed form according to your organization's protocols, or print it for physical records.

Who is required to fill out the Vaccine Temperature Log Form?
The Vaccine Temperature Log Form must be completed by healthcare providers responsible for vaccine storage, including pharmacists and public health officials overseeing vaccination programs.
What are the deadlines for submitting the Vaccine Temperature Log Form?
The form must be filled out daily to ensure compliance with health regulations, and it should be retained for a minimum of three years unless local laws state otherwise.
How do I submit the Vaccine Temperature Log Form after completing it?
You can submit the Vaccine Temperature Log Form electronically via email, or print and fax it to the relevant department as per your organization's submission guidelines.
Are there any supporting documents needed with this form?
Typically, no additional supporting documents are required; however, maintaining a record of temperature readings may be beneficial for compliance purposes.
What common mistakes should I avoid when completing this form?
Avoid omitting temperature recordings or staff initials, as these are crucial for the form's validity. Double-check the temperature readings entered to ensure they fall within the acceptable range.
How long does it take to process the Vaccine Temperature Log Form?
Processing time may vary, but as it is primarily a recording document, it is advisable to complete and submit it daily for timely review and compliance.
What actions should be taken if the temperature is outside the acceptable range?
If temperatures fall outside the acceptable range, it is essential to immediately contact the vaccine manufacturer and local health department for guidance on maintaining vaccine integrity.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.




















