Last updated on Apr 4, 2026

Get the free FDA Retort Inspection Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is fda retort inspection form
The FDA Retort Inspection Form is a government document used by inspectors to evaluate food processing safety in steam hydrostatic retorts.
pdfFiller scores top ratings on review platforms




Who needs fda retort inspection form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to fda retort inspection form
What is the FDA Retort Inspection Form?
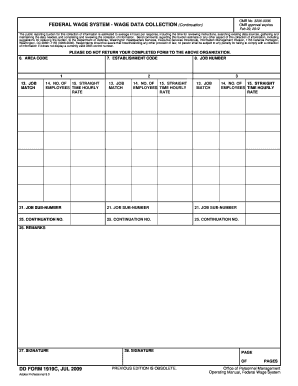
The FDA Retort Inspection Form, also known as the FDA 3511f form, is crucial for maintaining food safety standards in the processing industry. This form serves to evaluate the operation of food processing equipment, specifically steam hydrostatic retorts. Key components of the form include details on the retort description, controls, and temperature-recording devices such as thermometers.
Compliance with FDA regulations is essential for food processors, making this form a vital tool for ensuring safe food production practices.
Purpose and Benefits of the FDA Retort Inspection Form
The FDA Retort Inspection Form is essential for food processors as it ensures adherence to safety standards and regulatory compliance. By utilizing this form, inspectors can verify critical aspects of retort operation, particularly temperature monitoring during processing. This form not only aids in compliance with FDA mandates but also aligns with broader health regulations and OSHA requirements, fostering a safer work environment.
Additionally, the form assists food processors in confidently documenting their practices, which can enhance their reputation with health officials and consumers alike.
Key Features of the FDA Retort Inspection Form
The FDA Retort Inspection Form is structured to include several important sections that facilitate thorough inspections. Notable features of this form encompass:
-
Vents and gauges that monitor pressure and temperature.
-
Specific diagram requirements for retort systems.
-
Fillable fields and checkboxes for ease of use.
-
Detailed instructions for accurate completion.
These elements ensure that operators provide precise measurements and maintain proper records, thereby supporting ongoing compliance with FDA standards.
Who Needs the FDA Retort Inspection Form?
This form is required for a variety of stakeholders within the food processing industry. Specifically, entities that utilize steam hydrostatic retorts must complete this form during inspections. Health and safety officials are responsible for evaluating the form as part of their duties to uphold FDA regulations. Therefore, any food processing organization seeking to ensure compliance with FDA standards should familiarize themselves with the requirements of the Retort Inspection Form.
How to Fill Out the FDA Retort Inspection Form Online (Step-by-Step)
Completing the FDA Retort Inspection Form online is a straightforward process, and following these steps will help ensure accuracy:
-
Access the form through the specified online platform.
-
Carefully fill out each critical field, paying special attention to data entry.
-
Review all entries to minimize potential errors.
-
Use the included instructions for guidance on specific sections.
By following these steps, users can effectively complete the form with confidence while avoiding common mistakes.
Submission Methods for the FDA Retort Inspection Form
Once completed, the FDA Retort Inspection Form can be submitted via several methods, ensuring flexibility for users. The available submission options include:
-
Online submission through the designated portal.
-
Mailing the form to the respective FDA office.
-
In-person delivery to ensure timely handling of documentation.
Adhering to submission deadlines is crucial, and users should confirm the submission process with the appropriate contacts to verify receipt of their form.
What Happens After You Submit the FDA Retort Inspection Form
After submission, the FDA Retort Inspection Form undergoes a review process by relevant officials. Key steps in this process include:
-
Review and validation of all information submitted on the form.
-
Follow-up requests for additional information if necessary.
-
Addressing common reasons for rejection, such as incomplete data or incorrect entries.
Being aware of these steps can help food processors prepare for what to expect after submission and ensure that their forms meet all necessary criteria.
Security and Compliance When Using the FDA Retort Inspection Form
Ensuring the privacy and security of sensitive information is paramount when using the FDA Retort Inspection Form. pdfFiller provides robust security features, including adherence to HIPAA and GDPR compliance standards. Users can rest assured that their data is securely handled throughout the submission process.
It is also essential to maintain records as required by food safety standards, which emphasizes the importance of reliable document management practices in maintaining compliance.
Helpful Resources and Examples for the FDA Retort Inspection Form
For users seeking further assistance with the FDA Retort Inspection Form, there are several valuable resources available. These include:
-
Sample or completed forms for reference.
-
Guides related to food processing inspections.
-
Contact information for expert support regarding the completion of the form.
Utilizing these resources can enhance understanding and facilitate the proper completion of the Retort Inspection Form.
Start Completing the FDA Retort Inspection Form with pdfFiller
Utilizing pdfFiller for completing the FDA Retort Inspection Form offers numerous advantages, including easy editing and secure eSigning capabilities. Users can take comfort in knowing their data is managed with the highest security standards, allowing them to focus on completing the form accurately.
Get started today using pdfFiller to efficiently fill out your FDA Retort Inspection Form.
How to fill out the fda retort inspection form
-
1.Access the FDA Retort Inspection Form on pdfFiller by searching for its name in the search bar.
-
2.Once open, use the interface to navigate through each section of the form easily.
-
3.Before starting, gather necessary information such as retort descriptions, control settings, and compliance documentation.
-
4.Begin filling in the retort description and ensure all required fields are completed accurately within pdfFiller.
-
5.Utilize features such as checkboxes for compliance checks and draw diagrams as necessary directly on the document.
-
6.After completing all sections, review the filled form for any missing information or errors to ensure accuracy.
-
7.Finalize your form by saving or downloading it as needed, or submitting it directly through pdfFiller's submission options.

Who is eligible to use the FDA Retort Inspection Form?
The FDA Retort Inspection Form can be used by food safety inspectors, compliance officers, and managers in food processing facilities that utilize steam hydrostatic retorts for food safety protocols.
What is the deadline for submitting the FDA Retort Inspection Form?
There is typically no strict deadline; however, submitting the form promptly after an inspection is recommended to ensure compliance and proper documentation.
How should I submit the completed FDA Retort Inspection Form?
The completed form can be saved and submitted through electronic means, such as via a designated FDA submission portal or directly to an FDA compliance officer if required.
What supporting documents are needed when completing the FDA Retort Inspection Form?
Typically, you may need documentation covering retort specifications, previous inspection reports, and records of calibration for measuring devices used in the inspection.
What common mistakes should I avoid when filling out the form?
Ensure all fields are completed accurately and legibly, avoid using abbreviations without definitions, and double-check all measurements before submission.
What are the processing times for inspections using this form?
Processing times can vary depending on the facility and inspector workload; however, inspections are generally completed within a few days to a few weeks.
Are there any fees associated with the FDA Retort Inspection Form?
Generally, using the form itself does not incur fees unless there are specific assessments or regulatory actions involved by the FDA.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.