Last updated on Apr 4, 2026

Get the free Study Calendar Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is study calendar form
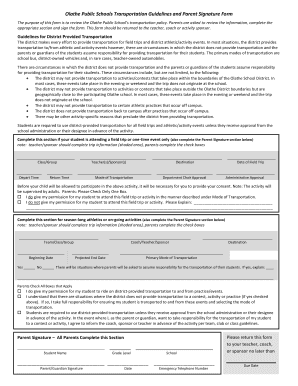
The Study Calendar Form is a medical document used by researchers to outline study-related services within clinical trials, aiding in budget preparation and informed consent.
pdfFiller scores top ratings on review platforms




Who needs study calendar form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to study calendar form
What is the Study Calendar Form?
The Study Calendar Form is a critical tool in clinical research, serving to document study-related services comprehensively. This form plays a significant role in aligning the Research Budget with the Informed Consent Form, ensuring clear communication and expectations for all parties involved. Essential components of the Study Calendar Form include services, procedures, codes, and pricing, contributing to the efficient management of clinical trials.
Purpose and Benefits of the Study Calendar Form
The Study Calendar Form is instrumental in organizing study services and clarifying billing responsibilities for clinical trials. By streamlining research protocols, the form enhances compliance with regulatory standards and minimizes billing issues and misunderstandings. Utilizing this form not only simplifies the management of study activities but also promotes transparency among researchers and participants.
Key Features of the Study Calendar Form
This form offers various fillable sections to capture necessary details regarding services, codes, and visit schedules. Users benefit from explicit instructions provided within the form, which facilitate accurate completion. Furthermore, customization options enable researchers to tailor the form to fit specific studies and protocols, enhancing its usability in a dynamic research environment.
Who Needs the Study Calendar Form?
The target audience for the Study Calendar Form includes researchers, coordinators, and compliance officers. It is essential in scenarios where detailed documentation of clinical research activities is required. Various fields within clinical research protocols necessitate the use of this form to ensure all aspects are thoroughly covered and compliant with regulatory demands.
How to Fill Out the Study Calendar Form Online (Step-by-Step)
To fill out the Study Calendar Form on pdfFiller, follow these steps:
-
Access the form directly on pdfFiller's platform.
-
Begin filling out the required fields, paying close attention to the provided instructions.
-
Save your progress frequently to prevent data loss.
-
Review each entry for accuracy before final submission.
Common Errors and How to Avoid Them
While completing the Study Calendar Form, users often encounter common pitfalls, including:
-
Omitting required fields or details.
-
Incorrectly coding services or pricing.
To mitigate errors, double-check all entries and utilize available resources for validation. If discrepancies arise, seek assistance to ensure all information is correct before submission.
Digital Signature Requirements for the Study Calendar Form
Understanding the signature requirements for the Study Calendar Form is crucial. Users may need either a digital or wet signature, depending on the guidelines set forth in their study protocol. pdfFiller offers efficient eSignature capabilities, ensuring secure handling of documents during the signing process.
Submission Methods and Delivery for the Study Calendar Form
Once the Study Calendar Form is completed, users can submit it electronically via pdfFiller. If applicable, mailing options are also available, complete with necessary address details for traditional delivery. It is essential to confirm submission and track its status to ensure timely processing of the document.
Security and Compliance for the Study Calendar Form
Data security is paramount when handling the Study Calendar Form. pdfFiller adheres to strict security measures, including compliance with HIPAA and GDPR. Maintaining privacy and following record retention requirements are critical for protecting sensitive information during the document management process.
Get Started with pdfFiller to Complete Your Study Calendar Form
pdfFiller provides numerous features to simplify the completion of the Study Calendar Form. Users can benefit from a variety of tools designed for seamless document editing, eSigning, and sharing. Testimonials from satisfied users underscore the platform's effectiveness in managing clinical research documentation.
How to fill out the study calendar form
-
1.To access the Study Calendar Form on pdfFiller, visit the pdfFiller website and use the search bar to locate the form by typing 'Study Calendar Form'.
-
2.Once you find the correct form, click on it to open the interactive PDF editor.
-
3.Begin filling in the form by clicking on the first field, where you will list study procedures. Hold your cursor over the designated area to expose fillable fields.
-
4.Gather necessary information before starting to fill the form, which includes details of services, corresponding codes, and pricing data related to the study.
-
5.Continue to navigate through the fields by clicking on them sequentially; use the tab key to move quickly between sections for a more efficient process.
-
6.Pay close attention to any specific instructions indicated at the top of the form to ensure accuracy in your entries.
-
7.Once you complete all required fields, review the entered information thoroughly for any errors or omissions.
-
8.To finalize the form, save your progress regularly and utilize the preview feature on pdfFiller to see the completed document before submission.
-
9.After reviewing, either download the form directly to your device or submit it through the designated channels provided within pdfFiller.

Who needs to fill out the Study Calendar Form?
The Study Calendar Form is essential for clinical trial coordinators, research administrators, healthcare providers involved in clinical studies, and budgeting specialists to ensure proper documentation of study-related services.
What information is required to fill out this form?
You will need to provide details about services, procedures, codes, prices, and visits concerning the study, alongside any instructions specific to the clinical trial.
Can this form be submitted online?
Yes, this form can be completed online using pdfFiller and submitted through the available online submission methods or downloaded for physical submission.
What are common mistakes to avoid when filling out this form?
Common mistakes include omitting required fields, entering inaccurate codes or prices, and failing to review the form for errors before submission.
Are there any deadlines associated with submitting the form?
Deadlines can vary based on the specific study protocol. It’s crucial to confirm submission timelines with the overseeing research institution or the clinical trial sponsor.
Is notarization required for the Study Calendar Form?
No, the Study Calendar Form does not require notarization, making it easier for users to complete and submit without additional steps.
How long does it take to process the Study Calendar Form?
Processing times depend on the reviewing institution's workload, but you can typically expect a response within a few business days after submission.
Related Catalogs
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.