Last updated on Apr 4, 2026

Get the free Exempt Committee Review Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is exempt committee review form
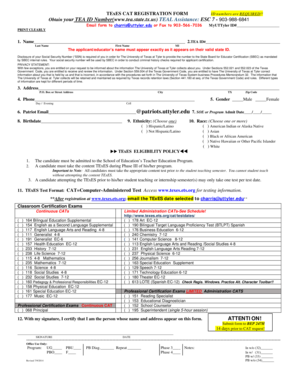
The Exempt Committee Review Form is a document used by researchers to determine if a study qualifies for exempt approval under the Common Rule Criteria (45CFR46).
pdfFiller scores top ratings on review platforms




Who needs exempt committee review form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to exempt committee review form
What is the Exempt Committee Review Form?
The Exempt Committee Review Form is a critical document used in research compliance to evaluate whether a study qualifies for exempt approval under the Common Rule Criteria (45CFR46). It serves to define the role of the form in facilitating ethical research practices.
Determining exempt approval is significant as it impacts how studies involving human subjects, minors, or sensitive data are regulated. Several types of studies may qualify for exemption, including certain educational practices and research conducted in established or commonly accepted educational settings.
Purpose and Benefits of the Exempt Committee Review Form
This form simplifies the research approval process for eligible studies. By using the exempt committee review form, researchers can expedite their submissions and minimize unnecessary delays in obtaining research approval.
One notable benefit includes a reduction in review times for studies that meet exemption criteria. Furthermore, compliance with research ethics and U.S. regulations is critical, as it ensures integrity in data handling and participant protection.
Key Features of the Exempt Committee Review Form
The Exempt Committee Review Form contains several essential sections that guide users effectively. Key features include specific inquiries regarding minors, compliance with HIPAA identifiers, and various criteria that influence exempt review decisions.
Form users will encounter YES/NO options alongside checkboxes designed for ease of completion. These features ensure that significant details are captured accurately during the application process.
Who Needs the Exempt Committee Review Form?
This form is primarily utilized by researchers across various institutions involved in studies that may impact human subjects. Research fields requiring ethical review often encompass social, behavioral, and educational research.
Examples of scenarios where this form is applicable include studies involving observational data collection or surveys conducted with minimal risk.
How to Fill Out the Exempt Committee Review Form Online
Completing the Exempt Committee Review Form online involves several straightforward steps. Begin by accessing the form on pdfFiller, then follow these guidelines:
-
Fill in basic information about the study and research team.
-
Answer all questions related to the criteria for exempt status, ensuring attention to detail.
-
Review the information for accuracy before submitting.
Common fields, such as study purpose and participant information, require precise data entries for compliance and approval accuracy.
Common Errors and How to Avoid Them
While filling out the Exempt Committee Review Form, users may encounter common mistakes that can hinder the approval process. Frequent errors include incomplete sections or inaccurate information regarding participant demographics.
To ensure a successful submission, here are some tips:
-
Double-check all entries for clarity and completeness.
-
Ensure compliance with HIPAA regulations, especially when handling sensitive information.
Submission Methods and What Happens After You Submit
The submission process for the Exempt Committee Review Form offers several options. Users can submit the form digitally via pdfFiller or traditional methods, depending on their institution's requirements.
After submission, users can expect a confirmation notification along with tracking capabilities to monitor the application status. This ensures transparency and helps researchers stay informed throughout the review process.
Security and Compliance for the Exempt Committee Review Form
pdfFiller prioritizes the security of sensitive documents submitted through the Exempt Committee Review Form. Utilizing 256-bit encryption, user data remains secure against unauthorized access.
Moreover, pdfFiller adheres to HIPAA and GDPR regulations, ensuring that all forms are handled with utmost privacy and data protection standards.
Why Choose pdfFiller for Your Exempt Committee Review Form Needs?
pdfFiller provides an array of benefits for users completing the Exempt Committee Review Form. With tools for editing, filling, and eSigning documents, pdfFiller enhances the overall form-filling experience.
The platform's capabilities streamline the completion of forms, reducing time spent on administrative tasks, and improving workflow efficiency for researchers.
Let pdfFiller Simplify Your Research Approval Process
By leveraging pdfFiller for form completion, users can navigate the complexities of research approvals with ease. The platform not only facilitates the filling out of the exempt committee review form but also enhances compliance with regulatory standards.
The combination of user-friendly features and robust support enables researchers to focus on what matters most—conducting valuable research.
How to fill out the exempt committee review form
-
1.Access pdfFiller and open the Exempt Committee Review Form by searching for its title in the platform's search bar.
-
2.Once the form is open, familiarize yourself with the layout, including sections with YES/NO options and checklist items.
-
3.Gather necessary information before starting your form, including details about your study, potential involvement of minors, and any HIPAA identifiers.
-
4.Begin filling in the form by carefully answering each question, using the checkboxes provided for yes/no responses.
-
5.Utilize pdfFiller's tools to add additional notes or comments in the specified areas, ensuring you thoroughly explain your responses where required.
-
6.After completing all fields, review the form for accuracy and completeness, checking for any missed questions or unclear responses.
-
7.Finalize your form by saving your changes and either downloading a copy for your records or submitting it directly through pdfFiller.

Who is eligible to submit the Exempt Committee Review Form?
Researchers from accredited institutions planning studies involving human subjects are eligible to submit the Exempt Committee Review Form. Ensure your study meets the criteria set by the Common Rule.
What are the submission procedures for the Exempt Committee Review Form?
You can submit the Exempt Committee Review Form electronically via pdfFiller or print it for manual submission to your Institutional Review Board (IRB). Be sure to follow your institution's specific submission guidelines.
What supporting documents are required when submitting the form?
Typically, you may need to include a study protocol, informed consent documents, and any relevant data protection plans when submitting the Exempt Committee Review Form to your IRB for review.
How long does it take to process the Exempt Committee Review Form?
Processing times vary by institution, but generally, IRBs aim to review exempt applications within a few weeks. It's advisable to submit your form well in advance of your planned study start date.
What common mistakes should I avoid when filling out the form?
Common mistakes include not fully answering all questions, failing to provide necessary details about the study, and neglecting to check all required boxes. Review your form thoroughly to avoid these issues.
Are there any fees associated with submitting the Exempt Committee Review Form?
Most institutions do not charge fees for submitting the Exempt Committee Review Form, but you should verify any potential costs with your specific IRB or research office.
What criteria does the IRB use to determine exempt status?
The IRB evaluates studies based on criteria outlined in the Common Rule (45CFR46), assessing elements like minimal risk to subjects, the nature of the data collected, and the use of deception, if applicable.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.