Last updated on Apr 4, 2026

Get the free 8b Continuing Review Worksheet
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is 8b continuing review worksheet
The 8b Continuing Review Worksheet is a healthcare form used by researchers to review protocols involving human subjects and ensure compliance with regulatory criteria.
pdfFiller scores top ratings on review platforms




Who needs 8b continuing review worksheet?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to 8b continuing review worksheet
What is the 8b Continuing Review Worksheet?
The 8b Continuing Review Worksheet is a specialized form essential for human subjects research. It serves as a comprehensive tool for documenting ongoing compliance in research protocols involving human subjects, ensuring regulatory adherence. This worksheet plays a crucial role in the research process by fulfilling key compliance requirements laid out by the Human Investigation Committee (HIC). Researchers utilize this form to maintain standards essential to ethical research practices.
Purpose and Benefits of the 8b Continuing Review Worksheet
The main purpose of the 8b Continuing Review Worksheet is to facilitate adherence to ethical research standards. By using this research compliance document, institutions enhance the likelihood of obtaining timely approvals from the Human Investigation Committee (HIC). This not only streamlines the review process but also serves to actively protect the rights and welfare of human subjects involved in research undertakings.
-
Ensures compliance with ethical standards in research
-
Facilitates approvals from HIC
-
Protects the welfare of human subjects
Key Features of the 8b Continuing Review Worksheet
The 8b Continuing Review Worksheet includes several critical sections designed for thorough documentation. Among these are protocol details, conflict of interest disclosures, and descriptions of the consent process related to a medical consent form. The form also addresses data safety monitoring and vulnerable populations, offering checkboxes and specified fields that simplify the completion process.
-
Protocol details and conflicts of interest
-
Consent process linked to HIPAA findings
-
Monitoring the safety of vulnerable populations
Who Needs the 8b Continuing Review Worksheet?
This worksheet is primarily intended for principal investigators and their research teams engaged in human subjects research. It is essential for institutions that require HIC reviews to ensure compliance with ethical standards. Additionally, individuals responsible for overseeing ethical compliance within research settings are also key users of this document.
How to Fill Out the 8b Continuing Review Worksheet Online
To complete the 8b Continuing Review Worksheet online, users should follow a series of step-by-step instructions to ensure accuracy. Initially, gather all necessary information to fill out the required fields efficiently. Each section must be addressed meticulously, as the accuracy and completeness of submissions are vital for regulatory compliance.
-
Gather all required documentation and information.
-
Navigate through each section of the worksheet.
-
Fill in all fields and check for accuracy.
Review and Validation Checklist for the 8b Continuing Review Worksheet
A review and validation checklist can significantly improve the accuracy of the completed worksheet. Users should ensure they fill in all required fields and be aware of common pitfalls that may hinder the review process. Understanding what reviewers look for during HIC evaluations will aid in constructing a thorough submission.
-
Checklist of required fields and common errors
-
Suggestions for reviewing the worksheet
Submission Methods for the 8b Continuing Review Worksheet
Users can submit the 8b Continuing Review Worksheet through various methods, including both digital and physical options. Knowing the deadlines associated with submissions and any potential fees is essential for timely processing. While digital submissions tend to streamline the process, understanding physical submission requirements is also critical.
Security and Compliance for the 8b Continuing Review Worksheet
Data safety and compliance with regulations are paramount when handling the 8b Continuing Review Worksheet. Compliance with HIPAA and data protection laws ensures that sensitive information remains secure throughout the research process. Adopting best practices for handling both personal and research data is vital for maintaining confidentiality.
Using pdfFiller to Complete the 8b Continuing Review Worksheet
pdfFiller offers a user-friendly platform to complete the 8b Continuing Review Worksheet with several helpful features. Functions such as eSign, convert, and share significantly enhance the form-filling process, allowing users to efficiently edit and share their worksheets while maintaining confidentiality and security.
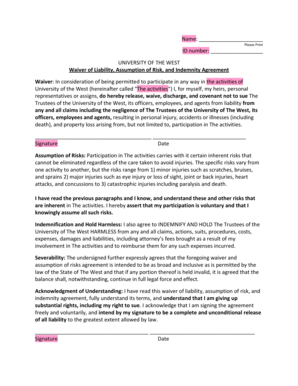
Sample of a Completed 8b Continuing Review Worksheet
Providing a sample of a completed 8b Continuing Review Worksheet serves as an invaluable reference for users. This visual representation of a filled-out form highlights key sections and filled fields, aiding users in comprehending the completion requirements. Utilizing the sample as a guide rather than a direct template is advisable for optimal form completion.
How to fill out the 8b continuing review worksheet
-
1.Access the 8b Continuing Review Worksheet on pdfFiller by navigating to the provided link or searching for the form in the pdfFiller interface.
-
2.Open the form by clicking on it, which will load it into pdfFiller's editing workspace.
-
3.Familiarize yourself with the sections of the form, including protocol details, conflict of interest, consent processes, and any applicable checkboxes.
-
4.Gather all necessary information before starting, such as HIC Protocol number, principal investigator details, and documentation regarding consent processes and confidentiality.
-
5.Fill out each field by clicking on the blank spaces or checkboxes, and entering the required information or selecting the appropriate options.
-
6.Use the toolbar in pdfFiller to navigate through multiple pages of the form and easily edit fields as needed.
-
7.Review the completed form for any errors or missing information, ensuring that all relevant sections are filled out accurately.
-
8.Finalize the form by clicking on the 'Save' button, followed by the 'Download' option or 'Submit' if applicable.
-
9.Consider saving a copy for your records and ensure that you follow any further submission protocols required by your institution.
Who is eligible to use the 8b Continuing Review Worksheet?
Principal investigators conducting research involving human subjects and members of the Human Investigation Committee (HIC) are primarily eligible to use the 8b Continuing Review Worksheet.
What information do I need before filling out this form?
Before completing the form, gather the HIC Protocol number, details about the principal investigator, the consent process, findings related to HIPAA, and any information regarding vulnerable populations included in your study.
How do I submit the completed form?
Once you have filled out the 8b Continuing Review Worksheet on pdfFiller, you can submit it electronically if required by your institution or download it for physical submission as per their guidelines.
Are there any common mistakes to avoid when filling out this form?
Common mistakes include leaving fields blank, not fully checking consent processes, and failing to review the form for accuracy before submission. Make sure all information is complete and correct.
Will I need supporting documents to complete this form?
It's advisable to have supporting documents such as consent forms and protocol details ready while filling out the 8b Continuing Review Worksheet, as they provide critical context for your responses.
What are the typical processing times for this form?
Processing times can vary depending on the review committee's schedule. Generally, expect a few weeks for review and feedback after submission, so plan accordingly.
What happens if my protocol involves vulnerable populations?
If your protocol includes vulnerable populations, ensure you provide thorough details about consent processes and risk assessments in the 8b Continuing Review Worksheet to address ethical concerns.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.