Last updated on Apr 10, 2026

Get the free Georgetown University Informed Consent Template
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is georgetown university informed consent
The Georgetown University Informed Consent Template is a medical consent form used by researchers to obtain participant consent for social and behavioral research involving treatment interventions.
pdfFiller scores top ratings on review platforms




Who needs georgetown university informed consent?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to georgetown university informed consent
What is the Georgetown University Informed Consent Template?
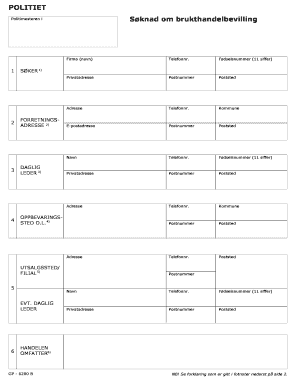
The Georgetown University Informed Consent Template serves as a critical document for social and behavioral research, primarily involving treatment interventions. This template is designed to facilitate a thorough understanding by outlining the research's nature, potential risks, benefits, and participants' rights. Its layout includes specified sections that help both researchers and participants navigate the essential information effectively.
Informed consent is fundamental in research, ensuring participants are fully aware of what their involvement entails. The importance of this template lies in its ability to standardize the consent process, contributing to ethical research practices. The clear structure of the template enables a straightforward completion, enhancing communication between parties involved.
Purpose and Benefits of the Georgetown University Informed Consent Template
The purpose of informed consent in research cannot be overstated, particularly concerning treatment interventions. This template plays a crucial role in outlining these processes, providing significant benefits to researchers and participants alike.
For researchers, utilizing this template means adhering to standardized practices, ensuring compliance with ethical guidelines. Benefits extend to research participants, granting them a clear understanding of their rights and the risks they may encounter. The clarity and transparency offered by the template lead to a more informed participant experience.
Key Features of the Georgetown University Informed Consent Template
The Georgetown University Informed Consent Template boasts several essential features, making it a valuable tool for researchers. Key sections include project details and participant information, which guide users in providing pertinent details effectively.
In addition, the template incorporates boilerplate language, simplifying the process for investigators. Its user-friendly design promotes clear communication, ensuring that all necessary information is presented in an accessible manner.
Who Needs the Georgetown University Informed Consent Template?
This template is essential for various roles within the research landscape. Subjects and researchers are the primary users, each holding specific responsibilities regarding informed consent.
It is particularly necessary in situations involving social behavioral research that requires treatment. By understanding their roles and obligations, both researchers and participants can engage in ethical research practices, safeguarding participant rights throughout the process.
How to Fill Out the Georgetown University Informed Consent Template Online
Completing the Georgetown University Informed Consent Template online using pdfFiller is an efficient process. To start, users can easily access the online filling platform, obtaining guidance through each step of the process.
-
Begin by navigating to the template within pdfFiller.
-
Follow field-by-field instructions, focusing on entering critical information accurately.
-
Avoid common errors such as omitting required fields or misinterpreting instructions.
How to Sign the Georgetown University Informed Consent Template
Signing the Georgetown University Informed Consent Template involves understanding the available options. Users can choose between a digital signature and a wet signature based on specific requirements outlined in the template.
The process for signing electronically through pdfFiller's secure platform is straightforward. Proper signing is crucial for ensuring the document's validity, reinforcing compliance with research standards.
Where to Submit the Georgetown University Informed Consent Template
Submitting the Georgetown University Informed Consent Template requires knowledge of the appropriate protocols. Various submission locations exist, including universities and Institutional Review Board (IRB) offices, guiding researchers on where to direct their documents.
Key deadlines and processing times are vital to consider, as they impact research timelines. Adopting best practices for submission enhances the likelihood of timely delivery, ensuring that all protocols are effectively followed.
Security and Compliance when Using the Georgetown University Informed Consent Template
Data protection and compliance play significant roles in the utilization of the Georgetown University Informed Consent Template. Researchers must implement robust security measures when handling confidential participant information.
Compliance with regulatory frameworks, such as HIPAA and GDPR, is essential to maintain participant trust and integrity in research practices. pdfFiller offers valuable resources designed to enhance security for sensitive documents, further safeguarding the informed consent process.
How pdfFiller Can Help You with the Georgetown University Informed Consent Template
pdfFiller simplifies the management and completion of the Georgetown University Informed Consent Template by offering a range of capabilities. Users can benefit from robust editing features, ensuring precise customization and clarity in their documents.
As a user-friendly platform, pdfFiller enhances the document management experience while emphasizing the importance of security in processing sensitive forms online. This ensures that both researchers and participants maintain a secure and efficient environment for managing consent documents.
How to fill out the georgetown university informed consent
-
1.Access the Georgetown University Informed Consent Template on pdfFiller by entering the form title in the search bar.
-
2.Open the form by clicking the document link in the search results, which will launch the pdfFiller interface.
-
3.Gather necessary information, including your project details, participant rights, and relevant treatment intervention information before starting to fill out the form.
-
4.Navigate through the form using pdfFiller's interface, clicking on each text field to enter the required information.
-
5.Complete any checkboxes pertaining to participant rights and conditions outlined in the template.
-
6.Review the filled areas for completeness and ensure all necessary information has been provided.
-
7.Finalize the form by clicking on the 'Sign' option where participants must sign and date the last page as per instructions.
-
8.Save your completed document by clicking 'Save' or 'Download' options. If needed, you can submit it through the submission methods provided by your institution.

Who needs to complete the Georgetown University Informed Consent Template?
The template must be completed by researchers who are conducting social and behavioral research, particularly those involving treatment interventions, to ensure participant understanding and consent.
What information is needed to fill out this form?
You will need participant details, study project information, risks involved, benefits, and required rights and protocols to accurately complete the form.
How do I submit the completed consent form?
After completing the form, you can save it and submit it according to guidelines provided by your institution or IRB. Ensure all necessary signatures have been obtained.
Are there any deadlines for completing and submitting this form?
Deadlines for submission can vary by project and institution. Typically, it should be completed prior to the participant's involvement in the research.
What common mistakes should I avoid when completing this form?
Be careful to fill in all blank fields, ensure all necessary signatures are acquired, and double-check for any spelling or factual errors before submission.
Is notarization required for this consent form?
No, notarization is not required for the Georgetown University Informed Consent Template as per the provided metadata.
What are the rights of participants outlined in this consent form?
Participants have rights regarding their involvement in the study, including understanding study details, potential risks, and benefits before providing consent.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.