Last updated on Apr 10, 2026

Get the free Adverse Reaction Tracking Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is adverse reaction tracking form
The Adverse Reaction Tracking Form is a healthcare document used by clinical users to collect and report patient allergy and adverse reaction data.
pdfFiller scores top ratings on review platforms




Who needs adverse reaction tracking form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to adverse reaction tracking form
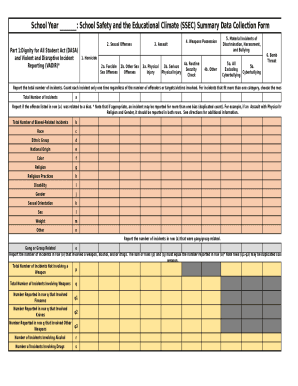
What is the Adverse Reaction Tracking Form?
The Adverse Reaction Tracking Form (ART) is a crucial tool in healthcare used to document and manage patient allergy and adverse reaction data. This form plays a vital role in improving patient safety by enabling healthcare providers to efficiently collect and report necessary information. By utilizing the ART form, clinical users, verifiers, and Pharmacy and Therapeutics (P&T) Committee members can ensure accuracy and compliance in managing patient reactions.
This patient allergy form serves not only to identify allergic reactions but also functions as an adverse drug reaction reporting form, aiding in the communication of critical data to regulatory bodies such as the FDA.
Purpose and Benefits of the Adverse Reaction Tracking Form
Employing the Adverse Reaction Tracking Form offers several key advantages in the healthcare sector. By tracking allergies and adverse drug reactions, this healthcare tracking form enhances the safety and well-being of patients. Accurate documentation allows for improved data collection and reporting processes, enabling healthcare professionals to respond swiftly to any incidents.
Additionally, utilizing this clinical data entry form streamlines the reporting to regulatory agencies, ultimately contributing to better public health outcomes.
Key Features of the Adverse Reaction Tracking Form
The ART Form is equipped with numerous features designed to facilitate efficient data entry and verification. Key characteristics include:
-
Fillable fields that allow clinical users to enter comprehensive patient data.
-
Verification sections that ensure accuracy through roles designated for verifiers.
-
Compatibility with specific software versions, essential for accessing all the available features.
These features make the ART form an effective clinical verification form that meets the requirements of regulatory standards, including those set forth by the FDA.
Who Needs the Adverse Reaction Tracking Form?
The primary users of the Adverse Reaction Tracking Form include clinical users, verifiers, and P&T committee members. Each group plays a distinct role in ensuring the form's effectiveness:
-
Clinical Users: This group, comprising doctors, nurses, and clinicians, is responsible for data entry.
-
Verifiers: Typically clinical pharmacists and dietitians, they check the accuracy of the data entered.
-
P&T Committee Members: They review the collected information, ensuring compliance and assisting in the reporting of adverse drug reactions to the FDA.
The interaction of these roles is critical in maintaining the integrity and accuracy of the data collected through the ART Form.
How to Fill Out the Adverse Reaction Tracking Form Online
To fill out the Adverse Reaction Tracking Form digitally, follow these steps:
-
Access the online platform where the form is hosted.
-
Begin entering patient reaction data into the designated fields.
-
Follow field-by-field instructions to ensure all necessary information is captured.
-
Utilize available tips for effective data input to reduce errors.
This process promotes efficiency in maintaining accurate patient records through the clinical data entry form.
Submission Methods and What Happens After You Submit
The Adverse Reaction Tracking Form can be submitted through various methods, including online submission and email options. After submitting the form, users can expect a confirmation of submission and should be aware of typical processing times, ensuring that they know when to anticipate feedback or further steps.
Security and Compliance for the Adverse Reaction Tracking Form
When handling sensitive patient data, security is paramount. The ART Form complies with HIPAA regulations and GDPR standards, reinforcing the commitment to privacy and data protection. Additionally, security features utilized by pdfFiller, such as 256-bit encryption, further safeguard sensitive information during the submission process.
Common Errors and How to Avoid Them
While filling out the Adverse Reaction Tracking Form, users may encounter several common errors. To avoid these pitfalls, consider these practical tips:
-
Ensure that all fields are filled out completely to maintain form completeness.
-
Double-check patient reaction data for accuracy before submission.
By being aware of these common errors and implementing strategies to prevent them, users can improve the accuracy of their submissions significantly.
How to Use pdfFiller for Filling and Signing the Adverse Reaction Tracking Form
pdfFiller offers an efficient solution for completing and signing the Adverse Reaction Tracking Form. Users can easily edit and annotate documents, making it convenient to prepare forms for submission. The benefits of eSigning capabilities also allow for seamless sharing and collaboration with stakeholders, streamlining the entire process of document management.
Experience Seamless Form Management with pdfFiller
Utilizing pdfFiller for form completion and management enhances the experience through its cloud-based functionality. Users can access and manage their forms from any browser, reducing barriers to document handling. With robust security features in place, pdfFiller ensures that sensitive documents are managed effectively and securely. Start filling out the Adverse Reaction Tracking Form today for a smoother documentation experience.
How to fill out the adverse reaction tracking form
-
1.Access the Adverse Reaction Tracking Form on pdfFiller by visiting their website and logging into your account.
-
2.Use the search bar to locate the form by typing 'Adverse Reaction Tracking Form' and selecting it from the dropdown options.
-
3.Once the form is open, familiarize yourself with the layout which includes various fillable fields and checkboxes.
-
4.Prepare to enter patient allergy and adverse reaction data by gathering necessary patient information such as drug details and previous reactions.
-
5.Begin filling in required fields labeled as 'Enter/Edit Patient Reaction Data'. Carefully enter all known information.
-
6.If you are verifying, select the appropriate fields for data verification using the 'Verify Patient Reaction Data' option.
-
7.Use dropdown menus to select options for 'DRUG INGREDIENT' as required for each patient's case.
-
8.Once all fields are filled out, review the form thoroughly for accuracy and completeness.
-
9.Finalize your entries by clicking on the 'Save' button. You can later download or submit the form directly through pdfFiller.
-
10.Select the 'Download' option to save a copy of the form to your device or choose the 'Submit' option to send the form electronically.

Who is eligible to complete the Adverse Reaction Tracking Form?
The form is primarily intended for clinical users, verifiers, and P&T Committee Users who report and verify patient allergy and adverse reaction data.
What is the deadline for submitting the Adverse Reaction Tracking Form?
While specific deadlines can vary based on facility policies, it's crucial to submit the form promptly to ensure timely reporting of adverse drug reactions.
How do I submit the Adverse Reaction Tracking Form?
You can submit the form electronically through pdfFiller after completing all required fields, or print and submit it based on your facility’s procedures.
What supporting documents are required with the Adverse Reaction Tracking Form?
Typically, no additional documents are needed, but confirming patient allergies or previous adverse reactions may be helpful for accuracy.
What common mistakes should I avoid when filling out this form?
Ensure all fields are filled accurately, verify drug ingredients correctly, and double-check patient data to avoid inaccuracies and delays in processing.
How long does it take to process the Adverse Reaction Tracking Form?
Processing times can vary widely based on the institution's review protocols, but you should confirm any expected timelines with your healthcare administrator.
What issues should I consider while using pdfFiller for this form?
Ensure you have the correct software versions for optimal functionality and confirm that the Progress Notes package version 2.5 is installed for generating notes.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.