Last updated on Apr 10, 2026

Get the free FDA Prior Notice Data Sheet
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is fda prior notice data
The FDA Prior Notice Data Sheet is a government form used by companies to submit information about imported products to the U.S. Food and Drug Administration for regulatory compliance.
pdfFiller scores top ratings on review platforms




Who needs fda prior notice data?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to fda prior notice data
What is the FDA Prior Notice Data Sheet?
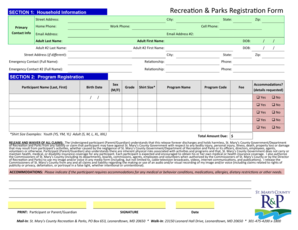
The FDA Prior Notice Data Sheet is a vital component in the process of ensuring compliance with U.S. Food and Drug Administration regulations for imported goods. This form is required by the FDA to gather essential information about the products being imported, including the submitting company’s details and product-specific information. The accuracy of this form is crucial as it plays a significant role in regulatory compliance, thereby facilitating smooth customs processing for imported products.
Included on the form are sections that require details such as the company name, address, and the FDA registration number. Completing this FDA import form accurately helps prevent delays and potential penalties during the importation process.
Purpose and Benefits of the FDA Prior Notice Data Sheet
The FDA Prior Notice Data Sheet serves several critical functions in import compliance. First, it ensures that all necessary information is provided to customs, enabling faster processing times for shipments. By using this U.S. import compliance form, individuals and companies can help safeguard public health and adhere to FDA’s regulatory requirements.
In addition to streamlining the import process, this form provides legal protection for companies that comply with FDA regulations. Benefits extend beyond regulatory compliance; quick approvals may lead to increased operational efficiency and greater reliability in supply chain logistics.
Who Needs to Complete the FDA Prior Notice Data Sheet?
Various entities must complete the FDA Prior Notice Data Sheet when importing goods into the United States. Primarily, businesses and individuals importing food, beverages, and pharmaceuticals are obligated to ensure the form is accurately filled out.
It's particularly relevant for businesses in the food industry, pharmaceuticals, and any other sectors dealing with imported products. These importers are responsible for meeting all regulatory compliance requirements associated with their shipments, which includes correctly completing the FDA Prior Notice Data Sheet.
How to Fill Out the FDA Prior Notice Data Sheet Online (Step-by-Step)
Filling out the FDA Prior Notice Data Sheet online is made simple through resources like pdfFiller. Follow these steps to ensure accuracy in each part of the form:
-
Begin by gathering essential information about your company and the products being imported.
-
Access the electronic form on pdfFiller.
-
Complete sections such as 'Submitting Company Name and Address' and 'FDA Registration Number'.
-
Pay special attention to confusing fields like 'Common Name' and 'FDA Product Code'.
-
Review all entries before submission to avoid errors.
By preparing ahead and following these steps, users can minimize confusion during the completion process.
Common Mistakes to Avoid When Completing the FDA Prior Notice Data Sheet
Errors in completing the FDA Prior Notice Data Sheet can lead to significant delays and complication in the import process. Some of the most common mistakes users make include:
-
Missing crucial information such as company details or product descriptions.
-
Incorrectly entering the FDA registration number or common name.
-
Failing to review the form for clarity and accuracy before submission.
Double-checking completed forms can prevent such errors, ensuring a smoother submission process and avoiding potential penalties associated with incorrect submissions.
Submission Methods for the FDA Prior Notice Data Sheet
Once the FDA Prior Notice Data Sheet is completed, users have multiple methods to submit the form. Submissions can be made electronically through platforms like pdfFiller or via physical submission. Each method has its own processes and timelines.
It's crucial to adhere to submission deadlines to prevent penalties, which can occur if the form is submitted late. There may also be associated fees for certain submission methods that users should be aware of.
What Happens After You Submit the FDA Prior Notice Data Sheet?
After submission of the FDA Prior Notice Data Sheet, users should expect confirmation of their submission through tracking options available. This acknowledgment is important for monitoring the status of the import.
Processing times may vary, and it is essential to be prepared for any follow-up requirements if additional information is requested. In cases where a form is rejected, users will need to understand the correction process to ensure compliance and resubmit accordingly.
How pdfFiller Can Help You with the FDA Prior Notice Data Sheet
PdfFiller offers a comprehensive solution for completing the FDA Prior Notice Data Sheet efficiently. Users can benefit from features such as form filling, eSigning, and document management all in one platform.
With strong security measures including 256-bit encryption, pdfFiller prioritizes the safe handling of sensitive documents. The user-friendly online interface ensures that users can access and complete crucial forms without hassle, enhancing the overall experience during the importation process.
Final Thoughts on Completing the FDA Prior Notice Data Sheet
Utilizing the FDA Prior Notice Data Sheet is essential for anyone involved in the import process. This form not only safeguards compliance with FDA regulations but also facilitates efficient border crossing for goods.
By simplifying form completion and submissions, pdfFiller helps users navigate regulations with ease, making it a practical choice for businesses and individuals alike. Start using pdfFiller today for a seamless experience managing your FDA documentation.
How to fill out the fda prior notice data
-
1.Access the FDA Prior Notice Data Sheet on pdfFiller by visiting the website and searching for the form title.
-
2.Once the form is open, familiarize yourself with the layout and labeled input fields.
-
3.Gather necessary information before you start filling in the form, including the submitting company's name and address, FDA registration number, shipment details, and product-specific information.
-
4.Begin filling in the form by entering the submitting company's name and address in the designated fields.
-
5.Input the FDA registration number in the corresponding field to establish compliance for your product.
-
6.Provide product-specific information, such as the Common, Usual or Market Name, quantity, and FDA product code in the appropriate sections.
-
7.If any details are missing, refer to your Customs Invoice for guidance, following any prompts provided in the form.
-
8.Once all fields are completed, review the form for accuracy, ensuring all required fields are filled correctly.
-
9.Utilize pdfFiller's tools to finalize the form, checking for any errors or omissions.
-
10.Save your completed form as a PDF document. You can download it directly from pdfFiller for submission or email it to the relevant authorities.
-
11.Submit the form as per the FDA guidelines, either electronically or print it out for physical submission as necessary.

Who is eligible to submit the FDA Prior Notice Data Sheet?
The FDA Prior Notice Data Sheet can be submitted by any business or individual involved in importing food products to the U.S. that requires FDA compliance, including manufacturers, importers, and customs brokers.
Is there a deadline for submitting the FDA Prior Notice Data Sheet?
Yes, the FDA requires that the prior notice be submitted at least two hours before the arrival of the shipment into the U.S. This ensures compliance and avoids delays at customs.
How do I submit the FDA Prior Notice Data Sheet?
You can submit the FDA Prior Notice Data Sheet electronically through the FDA's Prior Notice System Interface (PNSI) or use a third-party filing service for submission. Ensure all information is accurate before submission.
What supporting documents do I need for the FDA Prior Notice Data Sheet?
You may need supporting documents such as your Customs Invoice, bills of lading, and product specifications to provide the necessary details for completion of the FDA Prior Notice Data Sheet.
What are common mistakes to avoid when completing the form?
Common mistakes include omitting required fields, inaccuracies in product information, or providing an incorrect FDA registration number. Always double-check all entries for completeness and accuracy.
What is the processing time for the FDA Prior Notice Data Sheet?
Processing time for the FDA Prior Notice is generally quick, but you must allow at least two hours for the notice to be accepted before importing. Issues with the form can lead to delays.
What happens if the FDA Prior Notice Data Sheet is not submitted?
Failure to submit the FDA Prior Notice Data Sheet prior to import can result in denied entry of the goods into the U.S., potential fines, and additional processing delays.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.