Last updated on Apr 11, 2026

Get the free Investigator Registration Questionnaire
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is investigator registration questionnaire
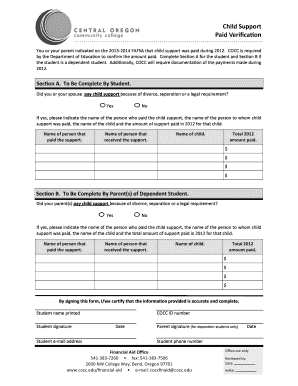
The Investigator Registration Questionnaire is a healthcare form used by ICON Clinical Research to collect necessary details from medical professionals for participation in clinical trials.
pdfFiller scores top ratings on review platforms




Who needs investigator registration questionnaire?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to investigator registration questionnaire
What is the Investigator Registration Questionnaire?
The Investigator Registration Questionnaire is a specific document utilized by ICON Clinical Research to gather essential personal and professional information from medical professionals interested in participating in clinical trials. This form is critical in determining the suitability of candidates for various research positions.
It collects various types of information, including the investigator's name, institution, contact details, medical license number, qualifications, and relevant clinical research experience. The form serves an important purpose by ensuring that only qualified medical professionals engage in clinical research activities, enhancing the overall integrity of the process.
Completing the Investigator Registration Questionnaire is vital for medical professionals aspiring to contribute to groundbreaking research within their respective fields.
Purpose and Benefits of the Investigator Registration Questionnaire
The Investigator Registration Questionnaire is an essential tool for both the investigators and ICON Clinical Research. It benefits medical professionals by facilitating their entry into the clinical research community.
By accurately completing the form, details collected streamline the recruitment process by allowing ICON to quickly access the needed information. This fosters a more efficient onboarding process for potential clinical trial investigators.
Additionally, the questionnaire plays a pivotal role in ensuring that all candidates meet the necessary regulatory compliance and vetting standards required for participation in clinical trials.
Key Features of the Investigator Registration Questionnaire
The Investigator Registration Questionnaire comprises several key sections designed to capture comprehensive information about the applicant. Main sections typically include personal information, qualifications, and specific therapeutic areas.
Questions within the questionnaire are designed to assess relevant experiences, such as electronic data capture (EDC) proficiency and past audit outcomes. Moreover, strong security measures are implemented to protect user data during the information submission process.
Who Needs the Investigator Registration Questionnaire?
The Investigator Registration Questionnaire is intended for clinical investigators and medical professionals who wish to engage in clinical research. This includes doctors, nurses, and other healthcare providers looking to participate in various therapeutic areas.
Eligibility criteria for completing the questionnaire may vary based on the specific clinical trial and typically require certifications or professional credentials relevant to the applicant’s field.
How to Fill Out the Investigator Registration Questionnaire Online (Step-by-Step)
-
Access the form through pdfFiller’s website.
-
Fill in essential fields such as your name, institution, and medical license number.
-
Pay attention to questions regarding previous experiences, particularly those related to electronic data capture (EDC).
-
Review your entries for accuracy and completeness before submission.
-
Submit the form electronically via pdfFiller by following the prompts provided.
Submission Methods and Delivery Options for the Investigator Registration Questionnaire
Users have various methods for submitting the completed Investigator Registration Questionnaire. Options include direct online submission through pdfFiller or via email to ICON Clinical Research.
It's essential to be aware of any specific submission deadlines and anticipated processing timelines associated with the form. Additionally, be prepared to include any necessary supporting documents that may be required for your submission.
Common Errors and How to Avoid Them
While completing the Investigator Registration Questionnaire, users may encounter several common errors. These often include missing fields, incorrect information entries, and failing to provide required documentation.
To avoid these issues, it’s beneficial to consult a troubleshooting guide for frequent errors and utilize a review checklist to verify the completeness and accuracy of your submission before sending it off.
Security and Compliance for the Investigator Registration Questionnaire
Data security is a significant concern when using the Investigator Registration Questionnaire. The form is secured with robust encryption features provided by pdfFiller, ensuring that sensitive information remains confidential during submission.
Moreover, compliance with relevant laws such as HIPAA and GDPR is maintained, reassuring users that their personal information will be handled with utmost respect and confidentiality throughout the process.
How to Download and Save the Investigator Registration Questionnaire PDF
Once the Investigator Registration Questionnaire has been filled out, users can easily download it for their records. Instructions for downloading the document are straightforward and can typically be completed in just a few clicks.
Users have the option to save the questionnaire in various file formats for convenience. It’s advisable to keep important documents organized and stored securely to prevent loss or unauthorized access.
Get Started with Your Investigator Registration Questionnaire Today!
The process of completing the Investigator Registration Questionnaire using pdfFiller is designed to be user-friendly and efficient. With supportive features like eSigning and secure sharing, pdfFiller enhances the overall experience.
By engaging in clinical research as an investigator, you open opportunities not just for yourself but also contribute to advancing medicine and patient care.
How to fill out the investigator registration questionnaire
-
1.To complete the Investigator Registration Questionnaire, start by accessing pdfFiller and searching for the form title. Once located, click on the form to open it in the editor.
-
2.Familiarize yourself with pdfFiller’s interface. Use the toolbar to zoom in or out for better visibility. The form will display various fields that need to be filled out.
-
3.Before you begin filling out the form, gather all necessary information such as your full name, institution details, contact information, medical license number, educational qualifications, and experience in clinical research.
-
4.Utilizing the editor, click on each field to enter the required information. For example, fill in your first and last name, and answer questions such as whether you have EDC experience by selecting 'Yes' or 'No' using the checkbox.
-
5.Once all fields are completed, review your entries for accuracy and completeness. Ensure that each section is thoroughly filled out according to the instructions provided.
-
6.After reviewing, click the 'Save' button to store your progress. Then choose the option to download or submit the form directly through pdfFiller. Ensure to follow the submission guidelines set by ICON Clinical Research.

Who is eligible to fill out the Investigator Registration Questionnaire?
Medical professionals, including doctors and researchers in clinical fields, who wish to participate in clinical trials administered by ICON Clinical Research can fill out this questionnaire.
What information is required to complete the form?
You will need to provide personal details such as your name, institution, contact information, medical license number, and qualifications, as well as information about your experience in clinical research.
How can I submit the completed questionnaire?
Once you have filled out the questionnaire on pdfFiller, you can either save and download the form or submit it directly to ICON Clinical Research through the platform, following the provided instructions.
What are common mistakes to avoid when filling out this form?
Ensure all fields are completed accurately. Common mistakes include omitting required information, entering incorrect details, and failing to sign the form where needed.
How long does it take to process my submitted questionnaire?
Processing times may vary, but typically you can expect feedback regarding your application status within a few weeks. For specific inquiries, contact ICON Clinical Research directly.
Is notarization required for this form?
No, notarization is not required for the Investigator Registration Questionnaire, making the submission process simpler and more efficient.
Where can I find support if I have questions about the form?
For assistance, you can reach out to ICON Clinical Research’s support team or refer to their website for additional resources and FAQs related to the Investigator Registration Questionnaire.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.