Last updated on Apr 12, 2026

Get the free Research Study Consent Form Template
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is research study consent form
The Research Study Consent Form Template is a legal document used by researchers to obtain informed consent from participants in a research study.
pdfFiller scores top ratings on review platforms




Who needs research study consent form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to research study consent form
What is the Research Study Consent Form Template?
The Research Study Consent Form Template is an essential document used to obtain informed consent from individuals participating in a research study. It serves as a fundamental element in medical research by clearly outlining the purpose, procedures, risks, benefits, and confidentiality measures related to the study. Informed consent is a vital aspect of participant rights and ethical research practices, ensuring that participants fully understand what they are agreeing to before taking part in any clinical trial.
Purpose and Benefits of Using a Research Study Consent Form Template
This consent form template is crucial for both researchers and participants for several reasons. Primarily, it helps outline the procedures, potential risks, and benefits associated with the research, ensuring that participants make informed decisions. Additionally, the template effectively protects participants' rights while enhancing accountability on the part of the researchers.
-
Clearly defines study procedures and expectations.
-
Outlines potential risks and benefits for participants.
-
Safeguards participant rights throughout the research process.
-
Ensures ethical practices by documenting consent.
Key Features of the Research Study Consent Form Template
The Research Study Consent Form Template includes several key features that aid in the collection of informed consent. Each section is meticulously designed to capture essential information relevant to the study.
-
Purpose statement that explains the research impetus.
-
Sections outlining risks and benefits of participation.
-
Blank fields for participant information and consent details.
-
Instructions that guide participants on how to fill out the form correctly.
Who Needs the Research Study Consent Form Template?
This form is required to be filled out by research participants prior to their involvement in any study. It is essential in scenarios where informed consent is legally mandated, ensuring that participants are aware of the study's implications.
-
All individuals partaking in research activities.
-
Clinical trial participants requiring formal consent.
-
Anyone providing sensitive health information as part of research.
How to Fill Out the Research Study Consent Form Template Online
Completing the Research Study Consent Form Template using pdfFiller is straightforward. Here’s a step-by-step guide to assist participants in accurately filling out the form.
-
Access the template on pdfFiller.
-
Input required participant information in the designated fields.
-
Read and acknowledge the sections outlining risks and benefits.
-
Provide a digital signature to confirm your consent.
To avoid common mistakes, ensure all fields are fully completed before submission and double-check for accuracy.
Review and Validation Checklist for the Research Study Consent Form Template
Before submitting the completed Research Study Consent Form Template, use the following checklist to ensure accuracy and compliance.
-
Verify that all fields are filled out completely.
-
Ensure you have signed and dated the form.
-
Double-check that the purpose, risks, and benefits are clear.
-
Confirm that you have provided correct contact information.
This validation process helps to avoid common errors and ensures that the form meets all regulatory requirements.
Submission Methods and Delivery for the Research Study Consent Form Template
Once the form is completed, participants can submit it through various methods. Understanding these options is crucial for timely submission and participation in the study.
-
Online submission directly through the pdfFiller platform.
-
Emailing the completed form to the designated research team.
-
In-person drop-off at a specified location, if applicable.
Processing times can vary, so it’s essential to inquire about submission deadlines and tracking availability.
Security and Compliance When Using the Research Study Consent Form Template
Handling sensitive information responsibly is paramount when using the Research Study Consent Form Template. pdfFiller offers robust security features to protect participant data.
-
256-bit encryption safeguards all documents.
-
Compliance with HIPAA and GDPR ensures data protection.
-
Protocols in place for maintaining participant privacy.
Users can trust that their information is secure throughout the consent process and beyond.
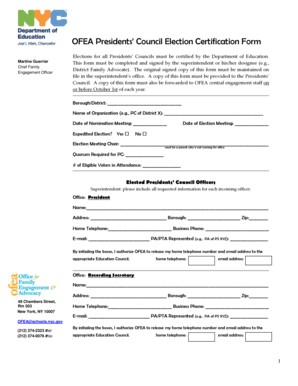
Sample of a Completed Research Study Consent Form Template
Seeing a completed research study consent form can provide valuable insights into how to fill out the template effectively. Below is a visual example.
-
A sample form demonstrating how to input participant information.
-
Explanatory notes for each section to clarify requirements.
This sample serves as a useful guide for users looking to understand what a fully executed consent form looks like.
Utilizing pdfFiller for the Research Study Consent Form Template
pdfFiller offers powerful capabilities for completing the Research Study Consent Form Template seamlessly. Using a cloud-based solution simplifies the entire process, enhancing user experience.
-
Ability to create fillable forms for various research purposes.
-
Secure eSignature options for legally binding consent.
-
Easy storage and retrieval of completed forms.
Leveraging pdfFiller's features ensures that participants can manage their documents efficiently and securely.
How to fill out the research study consent form
-
1.Access pdfFiller and search for the Research Study Consent Form Template in the search bar provided.
-
2.Once located, click on the form to open it in pdfFiller's editing interface.
-
3.Before filling out the form, gather necessary information such as participant details, risks, and benefits associated with the study.
-
4.Navigate through the form fields using the mouse or keyboard, placing the cursor where information is required.
-
5.Fill in personal details such as name, address, and contact information in the appropriate sections.
-
6.Provide a clear description of the research study's purpose and procedures in the designated fields.
-
7.Outline expected risks and benefits, ensuring participants understand what they are agreeing to.
-
8.Include confidentiality measures to assure participants that their personal information will be protected.
-
9.Review all completed information for accuracy and completeness, making necessary adjustments to ensure clarity.
-
10.Once satisfied with the entries, finalize the form by checking for any required signatures.
-
11.Save your work by clicking the save button or download the completed form directly to your device.
-
12.If needed, submit the form electronically through pdfFiller’s submission options or print it for physical submission.

Who is eligible to sign the Research Study Consent Form?
Individuals who are participating in the research study are eligible to sign the Research Study Consent Form. Typically, this includes adults who understand the study's purpose and agree to take part voluntarily.
Is there a deadline for submitting the consent form?
Yes, the consent form should be submitted before the participant begins their involvement in the research study. It's important to adhere to any specific deadlines set by the research team.
How do participants submit the consent form once completed?
Participants can submit the completed consent form electronically through pdfFiller or print it for physical submission to the research coordinator or designated person.
What supporting documents are required with the consent form?
Typically, no additional documents are needed with the consent form itself. However, participants may need to provide identification or other personal information as specified by researchers.
What are common mistakes to avoid when filling out the form?
Common mistakes include omitting required details, misunderstanding risks and benefits, and failing to sign the form where indicated. Always double-check all entries.
How long does it take to process the consent form?
Processing times can vary depending on the study. Generally, forms are reviewed quickly to ensure participants are adequately informed and can begin the study promptly.
What if a participant changes their mind after signing the form?
Participants have the right to withdraw their consent at any time without penalty. It's important to inform the research team of any change in participation status.
Related Catalogs
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.