Last updated on Apr 12, 2026

Get the free PAP Device Compliance Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

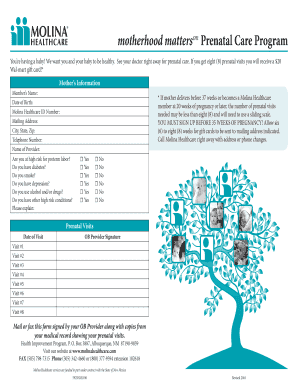
What is pap device compliance form
The PAP Device Compliance Form is a healthcare document used by physicians to confirm a patient's compliance with PAP therapy and validate the treatment's effectiveness.
pdfFiller scores top ratings on review platforms




Who needs pap device compliance form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to pap device compliance form
What is the PAP Device Compliance Form?
The PAP Device Compliance Form is a crucial document that verifies patient compliance with PAP therapy. Its primary function is to confirm the use of the PAP device, which is essential for effective sleep apnea treatment. This documentation is vital for healthcare providers to track the effectiveness of therapy and ensure patient safety. The form requires signing by the physician, which adds a layer of accountability and legitimacy to the compliance process.
Purpose and Benefits of Using the PAP Device Compliance Form
Accurate documentation is necessary in the treatment of sleep apnea, where the PAP Device Compliance Form plays a significant role. By thoroughly recording compliance, physicians can better evaluate treatment effectiveness and make necessary adjustments. This form benefits both healthcare providers and patients by fostering improved outcomes and potentially decreasing health risks associated with untreated sleep apnea.
-
Enhances patient outcomes through effective monitoring.
-
Promotes a systematic approach to managing sleep apnea treatment.
Key Features of the PAP Device Compliance Form
The design and structure of the PAP Device Compliance Form are user-friendly, incorporating blank fields and checkboxes for comprehensive input. Key sections are dedicated specifically for physician input, enabling clear communication of patient compliance. Security features, such as pdfFiller’s encryption, ensure that sensitive information complies with HIPAA standards.
Who Needs the PAP Device Compliance Form?
This compliance form is primarily aimed at healthcare providers, including physicians responsible for treating patients with sleep apnea. Additionally, patients may also find it beneficial as part of their treatment protocols, particularly in terms of understanding their compliance obligations. By ensuring all relevant parties utilize the form, treatment can be managed more efficiently.
How to Fill Out the PAP Device Compliance Form Online
Filling out the PAP Device Compliance Form online is a straightforward process. Using pdfFiller, users can easily navigate through the form’s sections, which are designed for clarity and efficiency. Before starting, it is advisable to gather all necessary patient information to streamline the process.
-
Access the form and begin with basic patient details.
-
Complete critical fields with accurate data from clinical visits.
How to Sign the PAP Device Compliance Form
Signing the PAP Device Compliance Form can be accomplished through different methods, including both digital and wet signatures. Digital signatures offer convenience, allowing for quick eSigning via pdfFiller's platform, ensuring compliance with legal standards. Understanding the nuances between digital signature and wet signature requirements can help streamline the process.
Submission Methods for the PAP Device Compliance Form
After completing the PAP Device Compliance Form, there are several options for submission. The form can be submitted electronically through secure channels or sent via traditional mail. Maintaining records of the submission is critical for compliance tracking and accountability.
What Happens After You Submit the PAP Device Compliance Form?
Post-submission, the confirmation process provides assurance that the form has been received successfully. In cases where the form is incomplete or rejected, follow-up actions are necessary to rectify issues, ensuring consistent compliance in patient care.
Security and Compliance for the PAP Device Compliance Form
Protecting patient data is paramount, and pdfFiller prioritizes security and regulatory compliance in handling the PAP Device Compliance Form. Best practices involve meticulous handling of the form, focusing on data retention and privacy considerations to ensure patient information remains secure.
Get Started with the PAP Device Compliance Form through pdfFiller
Utilizing pdfFiller for managing the PAP Device Compliance Form streamlines the process, offering an efficient platform for document management. With capabilities like easy editing and eSigning, pdfFiller enhances the overall user experience of handling compliance forms digitally.
How to fill out the pap device compliance form
-
1.Access the PAP Device Compliance Form through the pdfFiller platform by logging into your account or creating a new one.
-
2.Once logged in, use the search bar to find 'PAP Device Compliance Form' and select it from the results to open it in the editor.
-
3.Gather all necessary patient information prior to filling out the form, including patient name, date of visit, and compliance details on the PAP device usage.
-
4.Navigate the form by clicking on each blank field to enter the corresponding patient data.
-
5.Utilize pdfFiller's toolbar to incorporate checkboxes or additional comments in the designated areas if necessary.
-
6.Once all fields are filled, carefully review the information you provided for accuracy and completeness.
-
7.After finalizing, use the 'Save' feature in pdfFiller to store your completed form securely.
-
8.You can download the completed form as a PDF or submit it electronically via pdfFiller's submission options available within the platform.

Who needs to fill out the PAP Device Compliance Form?
The PAP Device Compliance Form must be filled out by physicians managing patients who utilize PAP devices for sleep apnea treatment, ensuring compliance with therapy.
Are there any deadlines for submitting this form?
While there is no strict deadline, it's recommended to submit the PAP Device Compliance Form promptly following the patient's appointment to ensure timely documentation and processing by insurance, if applicable.
How do I submit the PAP Device Compliance Form?
You can submit the PAP Device Compliance Form through pdfFiller's electronic submission feature or choose to download it and submit via fax or mail, depending on your clinic's policies.
What supporting documents are required with this form?
Typically, no additional documents are required with the PAP Device Compliance Form unless specified by your facility. However, it's advisable to have patient treatment records available for reference.
What are common mistakes to avoid when filling the form?
Common mistakes include failing to provide accurate patient details, overlooking the physician's signature, and leaving blank fields that could compromise the form’s validity.
How long does it take to process the PAP Device Compliance Form?
Processing times may vary based on the method of submission and the healthcare provider's policies, but typically expect a timeframe of a few days to two weeks.
Can this form be notarized?
The PAP Device Compliance Form does not require notarization unless specifically requested by a physician's office, so it is often sufficient with a physician's signature.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.