Last updated on Jan 3, 2015

Get the free Clinical Research Protocol Analysis Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is clinical research protocol analysis
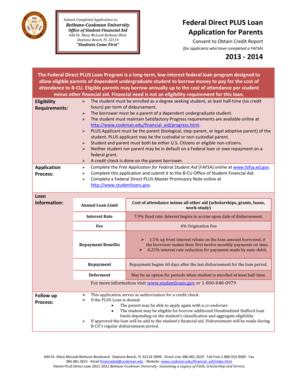
The Clinical Research Protocol Analysis Form is a medical document used by clinical researchers to analyze financial and procedural aspects of research protocols.
pdfFiller scores top ratings on review platforms




Who needs clinical research protocol analysis?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to clinical research protocol analysis
What is the Clinical Research Protocol Analysis Form?
The Clinical Research Protocol Analysis Form is an essential document used in clinical research to effectively document and analyze research protocols. This form plays a pivotal role in capturing critical information that influences the research process, including financial and procedural components. The form typically includes key sections such as sponsor information, financial details, and personnel involved in the study.
A crucial aspect of validation is the Principal Investigator's signature, which underscores the form's legitimacy and adherence to regulatory requirements. Researchers rely on this document to ensure transparency and accuracy in their protocols, thereby enhancing the integrity of clinical studies.
Purpose and Benefits of the Clinical Research Protocol Analysis Form
This form is vital for research studies as it provides numerous benefits during the financial planning phase of clinical trials. By documenting necessary financial aspects, the form aids in reimbursement processes and ensures that all protocol-induced procedures and tests are appropriately recorded.
-
Provides a systematic approach to financial analysis for research activities.
-
Streamlines documentation requirements, facilitating compliance and accountability.
-
Ensures clarity around budget allocation for various study components.
Key Features of the Clinical Research Protocol Analysis Form
The Clinical Research Protocol Analysis Form is designed with features that enhance usability and compliance for researchers. Among its notable attributes, the form includes fillable fields for essential information such as the 'Principal Investigator Name', 'Contact Person', and 'Protocol Title'.
Moreover, the form's digital capabilities allow for easy editing, eSigning, and sharing through platforms like pdfFiller. This ensures that sensitive information remains secure while simplifying the documentation process for researchers.
-
Compliance and security features to protect sensitive data.
-
User-friendly interface that supports quick data entry and retrieval.
Who Needs the Clinical Research Protocol Analysis Form?
The Clinical Research Protocol Analysis Form is primarily targeted towards a variety of stakeholders in the research community. Key users include Principal Investigators, research coordinators, and financial sponsors involved in clinical studies.
Understanding the roles and responsibilities associated with this form is essential. Each of these stakeholders must comply with specific regulations and documentation standards required for conducting research effectively.
How to Fill Out the Clinical Research Protocol Analysis Form Online
Completing the Clinical Research Protocol Analysis Form using pdfFiller is a straightforward process. Follow these steps to ensure accurate submission:
-
Access the form through the pdfFiller platform.
-
Fill out each relevant section, paying close attention to required fields.
-
Review all entered information before submission to avoid errors.
The clarity of instructions within the form will guide users on what fields are necessary to complete, ensuring an efficient filling experience.
Common Errors and How to Avoid Them When Filling the Form
When filling out the Clinical Research Protocol Analysis Form, it is critical to be aware of common mistakes that can jeopardize the completion process. Users often overlook sections requiring the Principal Investigator’s signature or fail to input financial details correctly.
-
Double-check all entries, especially financial and personnel information.
-
Stay informed about compliance requirements to ensure proper submissions.
Submission Methods for the Clinical Research Protocol Analysis Form
Upon completion, there are various methods available to submit the Clinical Research Protocol Analysis Form. Researchers can choose to submit their forms online using pdfFiller, via email, or as a hard copy, depending on institutional protocols.
It is crucial to follow the specific submission guidelines provided by different institutions or sponsors and to adhere to deadlines for timely processing. Tracking submissions can also aid in confirming receipt of the documents.
Security and Compliance for the Clinical Research Protocol Analysis Form
When handling sensitive information, security and compliance become paramount. PdfFiller offers robust security measures, including 256-bit encryption, to protect data during the submission process. The platform also adheres to strict compliance standards such as HIPAA and GDPR, ensuring that sensitive patient and research information is safeguarded.
This focus on security helps researchers manage and share documents with confidence, highlighting the platform's commitment to privacy and data protection.
Engage with pdfFiller to Complete Your Clinical Research Protocol Analysis Form
Using pdfFiller for completing the Clinical Research Protocol Analysis Form can significantly enhance the form-filling experience. The platform provides ease of use, accessibility, and a range of professional features designed to streamline the process.
By ensuring that sensitive documents are handled securely, users can focus on completing their research protocols effectively. Exploring additional resources and tools offered by pdfFiller can further facilitate the clinical research process.
How to fill out the clinical research protocol analysis
-
1.Access the Clinical Research Protocol Analysis Form on pdfFiller by searching for the form name or using the link provided by your organization.
-
2.Once opened, familiarize yourself with the layout of the form including header details, sections for input, and signature areas.
-
3.Prepare the necessary information before completing the form. Gather details like your name, contact information, protocol title, and any financial details related to the research.
-
4.Start filling out the form by clicking on each field, and type in the corresponding information. Use the fillable fields such as 'P.I. Name', 'Contact Person', and 'Protocol Title' to fill in required data.
-
5.Ensure to double-check the accuracy of all entries, particularly in financial sections and personnel information. Use pdfFiller's reviewing tools to highlight any sections that need further verification.
-
6.Once all information is entered, locate the 'P.I. Signature' box and ensure you sign where prompted. Validate if electronic signatures are accepted based on your institution's guidelines.
-
7.After completing all fields and signing, click the Save button to keep a record of your filled form, or select Download for a copy on your device.
-
8.To submit the form, check for an upload button or instructions that align with your institutional needs. Follow the submission protocols to ensure proper processing.

Who is eligible to use the Clinical Research Protocol Analysis Form?
The Clinical Research Protocol Analysis Form is primarily for Principal Investigators and their teams who are managing clinical trials or research studies and need to document associated financial or procedural details.
Are there any deadlines associated with submitting this form?
Deadlines for submitting the Clinical Research Protocol Analysis Form depend on your research protocol timeline. Always confirm with your institution or sponsor about specific date requirements that may apply.
How do I submit the Clinical Research Protocol Analysis Form?
You can submit the completed Clinical Research Protocol Analysis Form through pdfFiller's submission interface. Ensure all fields are complete before following your institution's guidelines for documentation submission.
What supporting documents are needed with this form?
Typically, additional documents might include financial agreements and study protocols. Ensure you confirm the specific requirements for your submission with your institutional review board.
What common mistakes should I avoid when completing this form?
Common mistakes include leaving fields blank, entering inaccurate financial details, and failing to sign the form. Always double-check your entries and ensure completion of all required sections.
How long does it take to process the Clinical Research Protocol Analysis Form?
Processing times vary depending on the reviewing body. Generally, it may take a few days to several weeks, so it's advisable to submit well in advance of any deadlines.
What should I do if I encounter issues while filling out the form on pdfFiller?
If you encounter any issues, refer to pdfFiller's help section or customer support for assistance. They can guide you through technical difficulties or usage questions.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.