Last updated on Apr 14, 2026

Get the free Human Participants Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is human participants form
The Human Participants Form is an educational document used by student researchers to obtain consent for research involving human participants outside Regulated Research Institutions.
pdfFiller scores top ratings on review platforms




Who needs human participants form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to human participants form
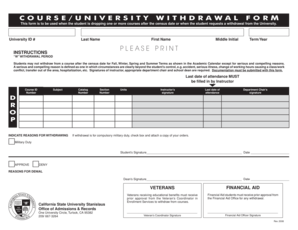
What is the Human Participants Form?
The Human Participants Form is a critical document in research involving human subjects. Its primary role is to facilitate necessary approvals, such as IRB approval, and to ensure the process of informed consent. This form encompasses essential elements including consent acquisition and risk assessment to uphold ethical research practices.
Researchers must use the Human Participants Form to clearly outline their project's purpose, methodology, and the potential risks involved to participants, ensuring transparency and ethical responsibility.
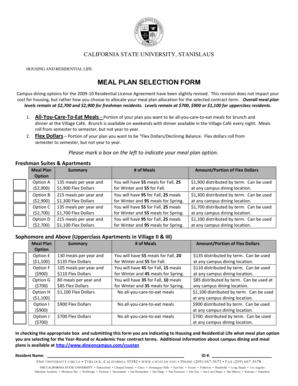
Purpose and Benefits of the Human Participants Form
The Human Participants Form serves several essential functions that enhance the ethical landscape of research. Primarily, it emphasizes the importance of informed consent, allowing participants to make educated choices regarding their involvement in research activities.
By using this form, researchers can streamline the approval process, making it easier to secure necessary permissions for diverse research initiatives. This efficiency not only benefits researchers but also protects participants’ rights and fosters trust in the research process.
Key Features of the Human Participants Form
This form includes various essential sections critical to the research workflow. Key features encompass role identification, where participants such as the student researcher and medical or mental health professionals are clearly defined, along with their respective signing requirements.
Additionally, the Human Participants Form includes sections dedicated to risk assessment and mandatory signatures from various stakeholders, such as educators and school administrators, strengthening the document's integrity.
Who Needs to Use the Human Participants Form?
The Human Participants Form is intended for a diverse audience involved in research projects. Roles that are required to sign this form include medical or mental health professionals, educators, and school administrators, all crucial for ensuring ethical compliance.
Moreover, eligibility criteria for research participants are carefully outlined within the form, making it an indispensable tool for anyone conducting research that involves human subjects.
How to Fill Out the Human Participants Form Online (Step-by-Step)
-
Begin by accessing the form through pdfFiller.
-
Carefully read each section to understand what information is required.
-
Gather necessary details, including names and contact information of all signatories.
-
Fill out the sections related to project details, risks, and informed consent accurately.
-
Complete the signature lines electronically with the help of pdfFiller's digital signature feature.
Following these steps can ensure a smooth submission process and help maintain compliance with necessary regulations.
Common Errors and How to Avoid Them
Identifying typical mistakes can significantly enhance the completion of the Human Participants Form. Some common errors include missing signatures and incomplete sections, which can delay the approval process.
To mitigate these issues, review the form thoroughly before submission. Create a checklist to validate that all required fields are filled, ensuring that no vital information is overlooked.
Submission Methods and Delivery for the Human Participants Form
There are multiple ways to submit the completed Human Participants Form. Options primarily include online submission through platforms like pdfFiller, or traditional methods such as mailing the document.
Be sure to check any state-specific rules regarding submission to ensure compliance with local regulations.
What Happens After You Submit the Human Participants Form?
After submitting the Human Participants Form, you can expect to receive confirmation of submission. This confirmation may include tracking options to monitor the progress of your form.
It's pertinent to be prepared for possible follow-ups or requests for additional documentation, ensuring an efficient response to any inquiries.
Security and Compliance of the Human Participants Form with pdfFiller
Using pdfFiller to complete the Human Participants Form guarantees security and compliance. The platform implements 256-bit encryption for data safety, ensuring user privacy throughout the form-filling process.
Moreover, pdfFiller is committed to maintaining compliance with industry standards, including HIPAA and GDPR, providing users with peace of mind when handling sensitive materials.
Get Started with Your Human Participants Form Today
Utilizing pdfFiller to fill out the Human Participants Form is a user-friendly experience. The platform simplifies the process, allowing you to create, edit, and secure your forms online effectively.
Engage with pdfFiller today to efficiently complete your Human Participants Form and enhance your research endeavors.
How to fill out the human participants form
-
1.Access the Human Participants Form on pdfFiller by searching for its title in the platform's search bar.
-
2.Open the form by clicking on its title in the search results to enable editing.
-
3.Familiarize yourself with the form’s layout, including checkboxes and signature lines for various roles.
-
4.Collect the necessary information regarding risk levels, participant details, and consent requirements before you begin filling out the form.
-
5.Start filling in the required fields by clicking on each textbox within pdfFiller and typing your information directly.
-
6.Utilize pdfFiller’s features, such as text editing and dropdowns, to efficiently complete checkboxes and selections based on the participants involved.
-
7.Once all sections are filled out, review the form for any missing information or errors using the review option on pdfFiller.
-
8.Finalize the form by ensuring all necessary signatures are completed. You can do this by using the e-signature feature within pdfFiller.
-
9.Save your changes by clicking on the save button and choose whether to download the completed form in PDF format.
-
10.Submit the form directly through pdfFiller if applicable, or download it for email sending or physical submission to the relevant authorities.

Who is eligible to fill out the Human Participants Form?
The form is designed for student researchers, adult sponsors, and other designated roles such as medical professionals, educators, and parents/guardians who support minor participants.
What is the deadline for submitting the Human Participants Form?
Deadlines vary based on specific research projects or institutional requirements. It is best to consult with your educational institution for specific submission timelines.
How do I submit the completed form after filling it out?
After completing the form on pdfFiller, you can download it or submit it directly through the platform if the option is available. Ensure all required signatures are included before submission.
Are there any supporting documents required with this form?
Typically, supporting documents may include consent letters from parents or guardians, and any approvals from medical or mental health professionals. Check specific requirements based on your research.
What are common mistakes to avoid when filling out this form?
Common mistakes include missing signatures, incomplete fields, and incorrect risk level assessments. Always double-check each section before finalizing to avoid these issues.
What is the processing time for the Human Participants Form?
Processing times can vary significantly depending on institutional review schedules. It's advisable to submit the form as early as possible to ensure timely approval for your research project.
What should I do if I have questions about the form while filling it out?
If you have questions, consult your institution's research office or a supervising educator. They can provide specific guidance on completing the form correctly.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.