Last updated on Jul 28, 2014

Get the free Informed Consent for DNA Testing
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is DNA Testing Consent
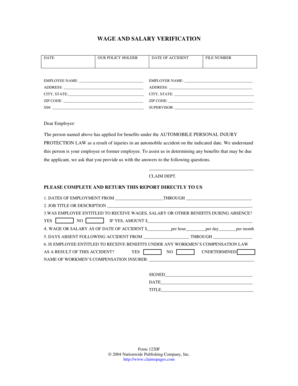
The Informed Consent for DNA Testing is a healthcare form used by patients in the US to authorize DNA-based testing for Multiple Endocrine Neoplasia 2B (MEN2B) / RET gene.
pdfFiller scores top ratings on review platforms




Who needs DNA Testing Consent?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to DNA Testing Consent
What is the Informed Consent for DNA Testing?
The Informed Consent for DNA Testing is a crucial document in medical settings, especially for patients undergoing testing related to Multiple Endocrine Neoplasia 2B (MEN2B). This form outlines the testing process and emphasizes the importance of obtaining patient consent before proceeding with DNA testing. Understanding the relevance of the informed consent DNA testing form is essential for the integrity of medical practices.
Purpose and Benefits of Informed Consent for DNA Testing
The purpose of the informed consent form is to ensure patients are fully aware of the DNA testing process, including its risks and limitations. By signing this document, patients can engage in informed decision-making regarding genetic testing, allowing them to weigh potential benefits and concerns. This process safeguards the rights of patients while providing necessary insights into their health.
-
Awareness of testing procedures and risks
-
Empowerment for informed decision-making
-
Clarification on genetic testing consent
Key Features of the Informed Consent for DNA Testing
Key features of the informed consent form include required signatures from the patient, physician, and a witness. Additionally, the form includes checkboxes for patients to select the specific type of DNA testing they are authorizing. These features are designed to facilitate transparency and ensure all parties understand the nature of the consent given.
-
Signatures required from patient, physician, and witness
-
Checkboxes for selecting types of DNA testing
Who Needs to Complete the Informed Consent for DNA Testing?
The completion of the informed consent for DNA testing requires the involvement of multiple parties: the patient, physician or counselor, and a witness. Each participant plays a specific role in the process, ensuring that consent is obtained responsibly and ethically. Patients must meet certain eligibility criteria based on their medical context.
How to Fill Out the Informed Consent for DNA Testing (Step-by-Step)
Filling out the Informed Consent for DNA Testing must be approached with careful attention to detail. The patient needs to provide necessary personal information, such as the names and dates of birth of any minor children if applicable. The following steps outline the process for completing the form electronically:
-
Enter personal details accurately, including the names and birthdays of any involved minors.
-
Read through the form carefully to understand all instructions and requirements.
-
Fill in necessary fields and ensure all checkboxes are marked appropriately.
-
Complete the signature requirements as outlined in the form.
Common Mistakes to Avoid When Completing the Informed Consent for DNA Testing
Common pitfalls during the completion of the informed consent form can lead to delays and complications. Missing signatures or unchecked boxes are frequent errors that should be avoided. It is vital for patients to review the document thoroughly before submission to ensure all necessary information is included and accurate.
-
Missing signatures from any required parties
-
Unchecked boxes for selected testing types
Security and Compliance for the Informed Consent for DNA Testing
The security of sensitive documents such as the informed consent for DNA testing is paramount. pdfFiller implements robust security measures, including 256-bit encryption, to protect patient data. Furthermore, the platform complies with HIPAA and GDPR regulations, ensuring that all personal information is handled with the utmost care and respect for privacy.
How to Submit the Informed Consent for DNA Testing
Once the informed consent form is completed, patients have several options for submission. They may submit the form directly to their healthcare provider or relevant agency. It is advisable to track submissions and confirm receipt to ensure that the information has been successfully processed.
-
Submit to healthcare providers or agencies directly
-
Track the submission for confirmation of receipt
What Happens After You Submit the Informed Consent for DNA Testing?
After submitting the informed consent form, patients can expect certain outcomes regarding the processing of their request. Typical processing times may vary, and it is important to be aware of the next steps following submission. Patients should follow up if necessary to check the status of their application.
-
Understanding typical processing times
-
Checking application status and taking necessary follow-up actions
Using pdfFiller to Manage Your Informed Consent for DNA Testing
pdfFiller offers valuable services for managing the informed consent form and other document-related tasks. Users can take advantage of features such as editing, eSigning, and securely sharing documents without the hassle of traditional methods. The platform is designed for ease of use and provides comprehensive support to users navigating the paperwork process.
How to fill out the DNA Testing Consent
-
1.Begin by accessing pdfFiller and entering the search term 'Informed Consent for DNA Testing' in the search bar.
-
2.Once located, click on the form to open it in the editor interface where you can fill in your details.
-
3.Before starting, gather necessary information such as the names and dates of birth of any minor children involved and understand the testing process and associated risks.
-
4.Use the available fields to input your personal information accurately, ensuring to fill out sections regarding the type of DNA testing you are requesting.
-
5.Carefully read the instructions provided in the document to ensure all required fields are addressed.
-
6.Identify the fields that necessitate signatures, ensuring they are correctly signed by you, a witness, and a physician or counselor.
-
7.Once all fields are completed, review the document for any omissions or inaccuracies to avoid common mistakes.
-
8.To save your progress, click on the 'Save' button. You can also download a copy for your records or submit it electronically through pdfFiller.

Who is eligible to use the Informed Consent for DNA Testing form?
Any individual seeking DNA testing for Multiple Endocrine Neoplasia 2B (MEN2B) or related genetic conditions can utilize this form, with particular relevance for patients, legal guardians of minors, and healthcare providers.
Is there a deadline for submitting the Informed Consent for DNA Testing form?
While there is no universal deadline, it is advisable to complete and submit the form prior to scheduled testing to ensure compliance with medical procedures and policies.
What submission methods are acceptable for the completed form?
The completed Informed Consent for DNA Testing form can typically be submitted electronically via healthcare provider systems or printed and handed directly to a physician or counselor during the consultation.
Are there any supporting documents required with the Informed Consent form?
Generally, additional supporting documents are not needed, though it may be beneficial to have previous medical history or identification available when filling out the form, particularly for minors.
What are common mistakes to avoid when filling out the form?
Ensure all required fields are filled in completely and accurately, particularly the names and signatures. Also, avoid leaving any sections blank that are crucial for processing your consent.
How long does processing take after submitting the form?
Processing times may vary by healthcare provider, but typically, you should expect information and follow-up within a few days to a week. Always confirm with your provider.
What specific concerns should I know about before signing the consent?
It is important to understand the implications of genetic testing, including potential emotional impacts, confidentiality of results, and how results may affect family members before signing the consent.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.