Last updated on Oct 5, 2014

Get the free KQ4 Quality Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is KQ4 Form
The KQ4 Quality Form is a medical document used by researchers to evaluate studies on labor and postpartum management of gestational diabetes mellitus.
pdfFiller scores top ratings on review platforms




Who needs KQ4 Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to KQ4 Form
1. Overview of the KQ4 Quality Form
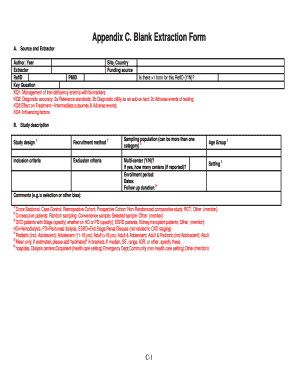
The KQ4 Quality Form is a medical research evaluation form specifically designed for studies focusing on gestational diabetes mellitus. This form aids in evaluating the quality of research related to labor and postpartum management, highlighting the significance of thorough medical assessments in these areas. By providing an organized structure, the KQ4 Quality Form assists researchers in making informed conclusions based on the analyzed data.
2. Purpose and Benefits of the KQ4 Quality Form
The KQ4 Quality Form serves multiple key objectives in enhancing healthcare outcomes through rigorous research evaluations. By utilizing this gestational diabetes study form, researchers can better assess their findings, thereby contributing to improvements in clinical practices. Healthcare professionals and institutions benefit significantly from the form as it streamlines the research process and enhances the ability to track and assess research quality.
-
Improved evaluation of clinical studies.
-
Enhanced collaboration among research teams.
-
Facilitation of informed decision-making in healthcare practices.
3. Key Features of the KQ4 Quality Form
The KQ4 Quality Form includes several critical elements that support efficient research evaluations. This clinical study assessment form is structured with various sections for detailing study design and sampling methods, making it a practical tool for researchers. The form includes fillable fields and checkboxes that allow for streamlined data entry, ensuring an organized and thorough completion process.
-
Sections for reporting study design and methodologies.
-
Fillable fields for easy data input.
-
Checkboxes to facilitate quick responses.
4. Who Needs the KQ4 Quality Form?
The KQ4 Quality Form is indispensable for various stakeholders in the healthcare and medical research fields. Researchers conducting studies specifically related to gestational diabetes mellitus and similar health concerns stand to gain the most from this tool. Additionally, institutions working on labor and postpartum management can efficiently utilize this form to enhance their research quality.
-
Healthcare researchers involved in diabetes studies.
-
Medical professionals focusing on postpartum management.
-
Institutions conducting health-related research evaluations.
5. When and How to Submit the KQ4 Quality Form
Understanding when and how to submit the KQ4 Quality Form is crucial for effective research documentation. It is recommended to submit the form as early as possible in the research process, ensuring timely evaluations. The submission can typically be completed online, following a clear set of steps designed to be user-friendly.
-
Complete all required fields in the form accurately.
-
Review the completed form for any errors or omissions.
-
Submit the form through the designated online platform.
6. Instructions for Filling Out the KQ4 Quality Form
Filling out the KQ4 Quality Form accurately ensures the integrity of the research conducted. A detailed breakdown of each field provides clarity on the specific information required for successful completion. It is also essential to be aware of common errors that may occur during the filling process to maintain accuracy in submissions.
-
Field-by-field instructions clarify data entry requirements.
-
Double-check for any potential inaccuracies before submission.
7. Ensuring Security and Compliance with the KQ4 Quality Form
Data security and compliance are paramount when utilizing the KQ4 Quality Form in sensitive healthcare research. The form incorporates robust security measures such as 256-bit encryption to protect submissions. Compliance with HIPAA regulations ensures that patient data remains confidential during the research process.
-
Ensure submissions are secured with data encryption.
-
Maintain HIPAA compliance for sensitive information.
-
Implement best practices for data protection during storage and handling.
8. How to Access and Utilize the KQ4 Quality Form with pdfFiller
pdfFiller offers essential tools for users looking to work with the KQ4 Quality Form efficiently. Features such as editing, eSigning, and sharing facilitate a smooth experience when filling out the form. A step-by-step guidance through pdfFiller simplifies the process of downloading and completing the KQ4 Quality Form.
-
Edit the form directly in pdfFiller's platform.
-
Use eSigning for prompt approvals to streamline the process.
-
Share the completed form securely with other stakeholders.
9. Review and Track Your Submission
After submitting the KQ4 Quality Form, understanding the tracking processes can enhance transparency. Users can expect a confirmation once the form has been received, allowing for subsequent checks on its submission status. Being aware of what happens post-submission aids in maintaining an organized research timeline.
-
Receive a confirmation email immediately upon submission.
-
Access submission status updates through the designated platform.
10. Take Action with Your KQ4 Quality Form Today
With the user-friendly tools provided by pdfFiller, getting started on the KQ4 Quality Form has never been easier. The platform allows prompt completion and submission of the form, ensuring seamless integration into ongoing research projects. Timely action in form filling can significantly impact the effectiveness of research outcomes.
How to fill out the KQ4 Form
-
1.Access the KQ4 Quality Form by visiting pdfFiller's website and searching for the form by its name in the search bar.
-
2.Once you locate the KQ4 Quality Form, click on it to open the document in the pdfFiller interface.
-
3.Begin by reviewing the form fields. Ensure you have all necessary information gathered beforehand, such as study design details and diagnostic accuracy data.
-
4.Use your mouse or touchpad to navigate through the fields. Click on any blank area to type in your relevant information, or select checkboxes where applicable.
-
5.If you need to revisit any section, utilize the scroll feature to move up and down the form easily. This allows for efficient editing.
-
6.After completing all fields, carefully double-check your entries for any errors or missing information to ensure accuracy.
-
7.When you're satisfied with the completed form, look for the options at the top of the page. Choose to save your work to your pdfFiller account, or download it directly to your device.
-
8.If necessary, submit the form via the provided submission options, ensuring to follow any additional instructions that may have been outlined.

Who should complete the KQ4 Quality Form?
The KQ4 Quality Form should be completed by researchers conducting studies on gestational diabetes mellitus and reviewing its postpartum management.
Are there deadlines for submitting the KQ4 Quality Form?
Deadlines may vary based on the specific study or institutional guidelines. It's crucial to check with your organization for any submission timelines.
How can I submit the KQ4 Quality Form once completed?
After filling out the form on pdfFiller, you can save it to your device or directly submit it via the submission methods outlined by your organization or study protocol.
What supporting documents are needed with the KQ4 Quality Form?
Typically, you may need to include study protocols, consent forms, or ethical review approvals along with the KQ4 Quality Form, depending on your institution’s requirements.
What common mistakes should I avoid when filling out the form?
Common mistakes include overlooking sections, misspelling study details, or failing to review the form before submission. Double-check all inputs carefully.
How long does it take to process the KQ4 Quality Form?
Processing times can vary significantly based on the reviewing body. It may take several weeks for review and feedback; check with your organization for specifics.
Can I edit the KQ4 Quality Form after initial submission?
Usually, once submitted, changes cannot be made. However, check with the receiving entity to confirm if revisions are allowed.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.