Last updated on Sep 28, 2014

Get the free New Protocol Review Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Protocol Review Form
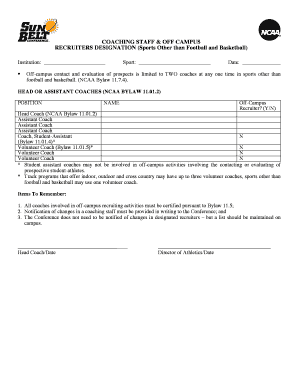
The New Protocol Review Form is a legal document used by Institutional Review Boards (IRBs) to evaluate and approve human subject research protocols.
pdfFiller scores top ratings on review platforms




Who needs Protocol Review Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Protocol Review Form
What is the New Protocol Review Form?
The New Protocol Review Form is a critical tool utilized by Institutional Review Boards (IRBs) to evaluate human subject research protocols. Its primary function is to assess areas such as risk assessment, subject selection, and informed consent to ensure ethical compliance. The form is designed to gather essential information, requiring a reviewer's signature for validation. By using this irb review form, researchers can facilitate the protocol approval process, ensuring that each project meets necessary ethical standards.
Why is the New Protocol Review Form Important?
This form plays a significant role in maintaining ethical oversight in research involving human subjects. By utilizing a dedicated research ethics form, researchers can establish a formal commitment to participant safety and adherence to ethical standards. Moreover, this protocol approval form is vital in protecting vulnerable populations, ensuring that appropriate safeguards are in place during the research process. Its use reinforces the ethical integrity of research projects and fosters trust between researchers and participants.
Who Needs the New Protocol Review Form?
The New Protocol Review Form is necessary for various individuals and organizations involved in research. This includes faculty, graduate students, and institutional researchers within universities and healthcare organizations. Researchers must meet specific eligibility criteria to access the form, typically based on their research topics that involve human subjects. By ensuring clarity and compliance, the irb signature form supports a well-structured review process for approved research endeavors.
How to Fill Out the New Protocol Review Form Online (Step-by-Step)
Filling out the New Protocol Review Form online can be efficient when following a structured approach. Here is a step-by-step guide to assist in the process:
-
Log in to the designated online platform for the form.
-
Gather all necessary documentation, including study protocols and informed consent materials.
-
Begin to fill out each section of the form carefully, ensuring accurate information.
-
Review each entry for completeness and clarity before proceeding.
-
Finalize the form by adding your digital signature if required.
By adhering to these steps, users can streamline their experience and ensure the proper filling of the research protocol evaluation.
Field-by-Field Instructions for the New Protocol Review Form
Each section of the New Protocol Review Form requires specific information. Below are the general field instructions to guide users:
-
Provide a brief overview of the research question and objectives.
-
Detail the risk assessment related to the study subjects.
-
Include information on how informed consent will be obtained.
-
Outline the selection criteria for study participants.
Common mistakes include leaving sections incomplete or using unclear terminology. Users should strive for clarity to avoid delays in the approval process.
How to Sign the New Protocol Review Form (Digital Signature vs. Wet Signature)
Signing the New Protocol Review Form can be done through either a digital signature or a wet signature. Digital signatures provide the convenience of eSigning, which is often faster and more secure. However, wet signatures may be required in some cases, depending on institutional policies. It's essential to be aware of the legal considerations for each type of signature to ensure compliance with the regulations governing research protocols.
Submission Methods and Deadlines for the New Protocol Review Form
The completed New Protocol Review Form can be submitted through various methods, including online portals and physical mail. Timely submission is crucial; researchers should be aware of specific deadlines and typical processing times to avoid delays. Adhering to these timelines ensures that the review process proceeds smoothly and efficiently.
Common Errors and How to Avoid Them When Filing the New Protocol Review Form
Many users encounter common errors when filling out the New Protocol Review Form. By identifing these issues, researchers can minimize mistakes. Here are some frequent pitfalls:
-
Incomplete sections or missing signatures.
-
Failing to adhere to formatting requirements.
To enhance accuracy, users should utilize a review and validation checklist before submission. This proactive approach helps catch errors early, improving the chances of a successful approval.
Security and Compliance Considerations for the New Protocol Review Form
Security is paramount when handling the New Protocol Review Form, especially given the sensitive nature of personal data involved. Researchers must comply with regulations such as HIPAA and GDPR to protect participant privacy. Ensuring that proper data monitoring protocols are in place can safeguard against breaches, thereby maintaining compliance with established ethical standards.
Experience Seamless Form Filling with pdfFiller
pdfFiller offers a convenient and efficient platform for filling out, signing, and submitting the New Protocol Review Form. Utilizing features such as cloud storage, secure signing, and editing capabilities, researchers can experience a streamlined workflow. This tool simplifies the process for users, making it easy to manage documents securely while focusing on what truly matters—the integrity of their research.
How to fill out the Protocol Review Form
-
1.To begin, visit pdfFiller and use the search bar to locate the New Protocol Review Form. Ensure you are logged in or create an account if necessary.
-
2.Once you find the form, click to open it in the pdfFiller interface. Review the layout to familiarize yourself with the sections provided.
-
3.Prior to completing the form, gather all necessary information, including project details, risk assessments, and any prior approvals.
-
4.Navigate through the form by clicking on fields to input your data. Use dropdowns for selections when applicable, and type responses in text boxes directly.
-
5.For fields requiring detailed descriptions, make sure to clearly outline the risks, benefits, and safeguards for human subjects involved in your research.
-
6.Stay attentive to the checkboxes, ensuring that you complete sections that assess subject selection and informed consent accurately.
-
7.After finishing all entries, review your responses carefully to verify that all information is complete and accurate, ensuring there are no omissions.
-
8.Finalize the document by signing where indicated. Use the 'Reviewer Signature' field for your signature and date it appropriately.
-
9.Once finalized, save your work directly in pdfFiller. You can download the completed form as a PDF or submit it electronically through the platform.
-
10.If submitting electronically, follow the prompts provided by pdfFiller to ensure your form is sent to the appropriate department or institution.
-
11.Remember to keep a copy of the submitted form for your records to ensure you have reference material.

Who is eligible to fill out the New Protocol Review Form?
This form is primarily for Institutional Review Board members, researchers, and administrators involved in human subject research. An understanding of research ethics and protocol standards is essential.
What is the submission procedure for this form?
The New Protocol Review Form must be submitted electronically through platforms like pdfFiller, where it can be filled out and sent directly to the IRB for evaluation. Ensure all fields are completed to avoid delays.
Are there any supporting documents required when submitting this form?
Yes, it is common to attach related documents such as research protocols, consent forms, and data monitoring plans that provide necessary context for the IRB's assessment.
What are some common mistakes to avoid when filling out this form?
Be thorough in filling out all required fields and avoid leaving items blank. Ensure accuracy in your descriptive responses, especially concerning risks and safeguards, to facilitate a smooth review process.
How long does it typically take to process the New Protocol Review Form?
Processing times can vary depending on the IRB’s workload. Typically, reviews can take from a few days to several weeks, so it is advisable to submit your form well in advance of your research timeline.
Is notarization required for this form?
No, notarization is not required for the New Protocol Review Form, allowing for quicker completion and submission through electronic platforms like pdfFiller.
What should I do if I encounter technical issues while filling out this form on pdfFiller?
If you face any technical difficulties, pdfFiller provides support options. You can access their help center or contact customer service for assistance regarding form completion.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.