Last updated on Oct 22, 2014
Get the free Vaccine Storage Temperature Log
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Temperature Log
The Vaccine Storage Temperature Log is a healthcare form used by medical facilities to monitor and record the temperatures of vaccine storage units.
pdfFiller scores top ratings on review platforms




Who needs Temperature Log?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Temperature Log
What is the Vaccine Storage Temperature Log?
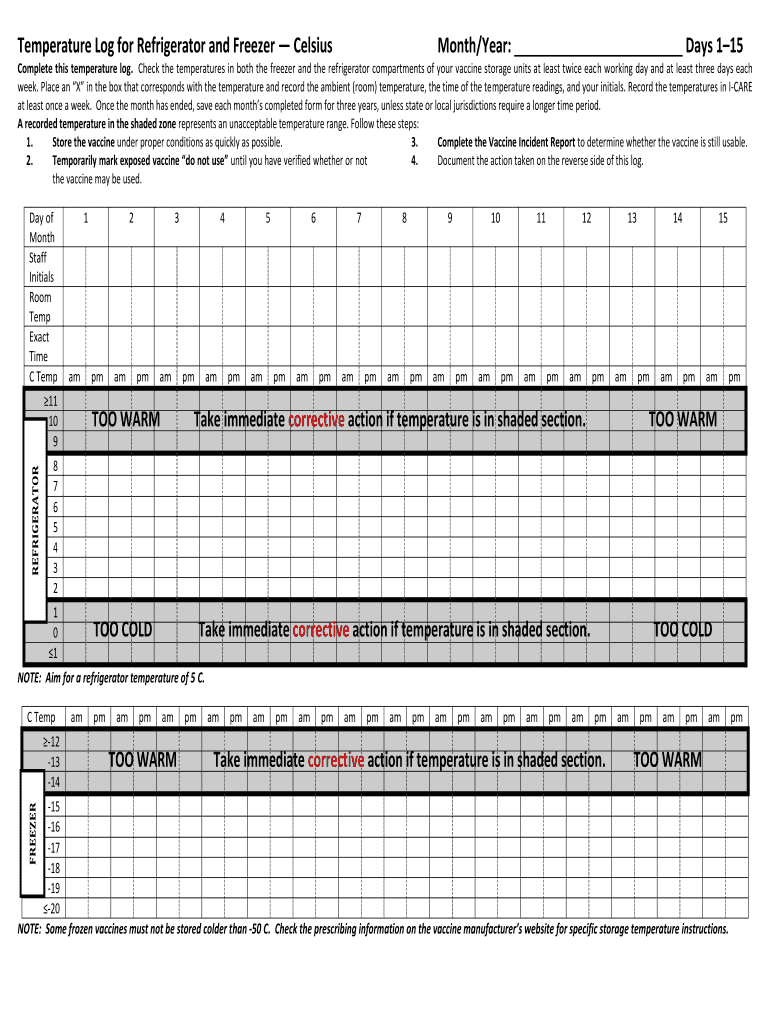
A Vaccine Storage Temperature Log is a crucial documentation tool in healthcare settings that records the specific temperatures of vaccine storage units. This log plays a vital role in ensuring the safety and efficacy of vaccines, as any deviation from the required temperature can compromise their effectiveness. Monitoring temperatures regularly is essential for maintaining compliance with health regulations and protecting patient health.
Purpose and Benefits of the Vaccine Storage Temperature Log
The primary purpose of the Vaccine Storage Temperature Log is to aid in vaccine storage monitoring. Proper usage of this log ensures compliance with regulations and safeguards the integrity of vaccines. By diligently maintaining accurate temperature records, healthcare facilities can significantly reduce the risk of vaccine spoilage and facilitate better patient health outcomes.
Key Features of the Vaccine Storage Temperature Log
This log includes several functional sections designed to streamline the recording process. Each form contains fields for:
-
Recording temperatures for both the freezer and refrigerator compartments
-
Initials of personnel checking and documenting temperatures
-
Incident reporting for any temperature excursions
Furthermore, built-in instructions guide users through the essential steps for temperature checks and how to document any issues effectively.
Who Needs the Vaccine Storage Temperature Log?
The Vaccine Storage Temperature Log is essential for a variety of healthcare providers, including clinics, hospitals, and pharmacies that manage vaccines. These organizations depend on accurate logs to ensure regulatory compliance and uphold the safety of patient care. Failing to maintain appropriate temperature records can have serious implications for vaccine efficacy and patient health.
How to Fill Out the Vaccine Storage Temperature Log Online
Filling out the Vaccine Storage Temperature Log online is straightforward. Follow these steps to ensure proper completion:
-
Access the digital form and locate the appropriate fields for entering temperature readings.
-
Input the temperatures recorded in both compartments, ensuring accuracy.
-
Initial each entry as verification by the responsible staff member.
-
Document any temperature excursions in the designated section, providing necessary details.
-
Complete the form by signing it digitally where required.
Attention to detail during this process is vital for maintaining accurate records.
Common Errors and How to Avoid Them
When completing the Vaccine Storage Temperature Log, several common errors can occur:
-
Neglecting to initial temperature entries, which can lead to unverified records.
-
Filling out records without checking device calibration, potentially leading to inaccurate readings.
-
Failing to report temperature excursions appropriately.
To avoid these mistakes, implement validation checks before submission and ensure that all entries are verified for completeness and correctness.
Security and Compliance for the Vaccine Storage Temperature Log
The handling of the Vaccine Storage Temperature Log requires stringent security measures to protect sensitive healthcare information. Key security protocols include:
-
Utilization of 256-bit encryption to secure data transmission.
-
Adherence to compliance standards such as HIPAA and GDPR.
-
Regular audits to ensure that patient information remains confidential.
These measures are essential to safeguard patient data associated with vaccine storage, building trust within healthcare environments.
How to Download and Save the Vaccine Storage Temperature Log
Accessing the Vaccine Storage Temperature Log digitally is simple. To download the form, follow these instructions:
-
Visit the designated website to locate the download option for the Vaccine Storage Temperature Log.
-
Select the format you prefer, commonly PDF, for easy storage.
-
Save the document to your device, ensuring it is organized for ease of access.
-
Store completed logs in a secure location for compliance tracking.
This process ensures that you maintain a readily accessible record of temperature logs.
Sample or Example of a Completed Vaccine Storage Temperature Log
Providing a visual reference for a completed Vaccine Storage Temperature Log can significantly aid users. A filled-out log typically includes:
-
Daily temperature entries for both refrigeration and freezing units
-
Staff initials for temperature checks
-
Documentation of any temperature excursions that occurred
Reviewing such samples can enhance understanding and accuracy in filling out your logs.
Experience Easier Form Management with pdfFiller
pdfFiller offers an intuitive platform for managing the Vaccine Storage Temperature Log and other healthcare documentation. Users benefit from features that simplify the filling, signing, and tracking of forms, ensuring compliance is easily maintained. The platform prioritizes security and compliance, providing peace of mind while handling sensitive information.
How to fill out the Temperature Log
-
1.Access pdfFiller and search for 'Vaccine Storage Temperature Log' in the template library.
-
2.Open the form to start filling it out. Familiarize yourself with the fields present in the template.
-
3.Before starting, gather necessary information, including the current temperature readings for the freezer and refrigerator compartments.
-
4.In the designated fields, record the time and corresponding temperature readings for both compartments accurately.
-
5.Utilize the fillable fields to enter your initials next to each temperature entry to authenticate the checks.
-
6.If any temperature excursions occur, follow the instructions provided on the form to document the incident properly.
-
7.After completing all fields, review the form for accuracy and completeness, ensuring all required entries are filled.
-
8.Once you are satisfied with the completed form, save your work by downloading it or submitting it directly through pdfFiller for your records.

Who is eligible to use the Vaccine Storage Temperature Log?
The Vaccine Storage Temperature Log is intended for use by any healthcare provider or organization that stores vaccines, including hospitals, clinics, and pharmacies. All personnel responsible for vaccine management should utilize this form.
How often should temperature readings be recorded?
Temperature readings should be taken and recorded at least twice each working day and for a minimum of three days each week to ensure vaccines are stored at the correct temperatures.
What should be done in case of a temperature excursion?
If a temperature excursion occurs, it's important to follow the provided guidelines on the form to document the incident, including the time, temperature, and any corrective actions taken.
Is notarization required for the Vaccine Storage Temperature Log?
No, notarization is not required for the Vaccine Storage Temperature Log. The form is designed to be filled out and used internally within healthcare facilities.
What are common mistakes to avoid when filling out this form?
Common mistakes include forgetting to record initial readings, not documenting temperature excursions correctly, and overlooking required entries. Always cross-check your entries before finalizing the form.
How should this form be submitted or stored after completion?
After completing the Vaccine Storage Temperature Log, it can be saved and stored electronically or printed for physical records. Ensure it is accessible for future audits and inspections.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.






















