Last updated on Nov 18, 2014
Get the free Integrated Data Repository - Application for Translational Data
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is IDR Data Request
The Integrated Data Repository - Application for Translational Data is a form used by researchers to request access to clinical and demographic data for translational research purposes.
pdfFiller scores top ratings on review platforms




Who needs IDR Data Request?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to IDR Data Request
What is the Integrated Data Repository - Application for Translational Data?
The Integrated Data Repository - Application for Translational Data serves a crucial role in facilitating access to healthcare research data. This application allows researchers to request essential clinical and demographic data, supporting various translational research endeavors.
Typically, this application is utilized by researchers, clinicians, and academic institutions engaged in healthcare studies. Its purpose is to streamline data collection and enhance the quality of research insights. The integrated data repository application plays a pivotal role in connecting multiple data sources and providing a comprehensive view needed for effective research outcomes.
Purpose and Benefits of the Integrated Data Repository - Application for Translational Data
The main purpose of the Integrated Data Repository is to provide researchers with a methodical way to access clinical and demographic data. This access significantly enhances the ability to conduct thorough and impactful research. By leveraging this application, researchers can meet predefined criteria and gather valuable insights that foster informed decision-making in healthcare.
Key benefits of utilizing this form include improved research quality and an enhanced understanding of patient data. Accessing a structured database allows users to uncover trends and correlations that are essential for translational studies. Utilizing a clinical data request form maximizes the potential for groundbreaking research.
Key Features of the Integrated Data Repository - Application for Translational Data
The Integrated Data Repository offers a range of features designed to aid researchers in the data request process. These include:
-
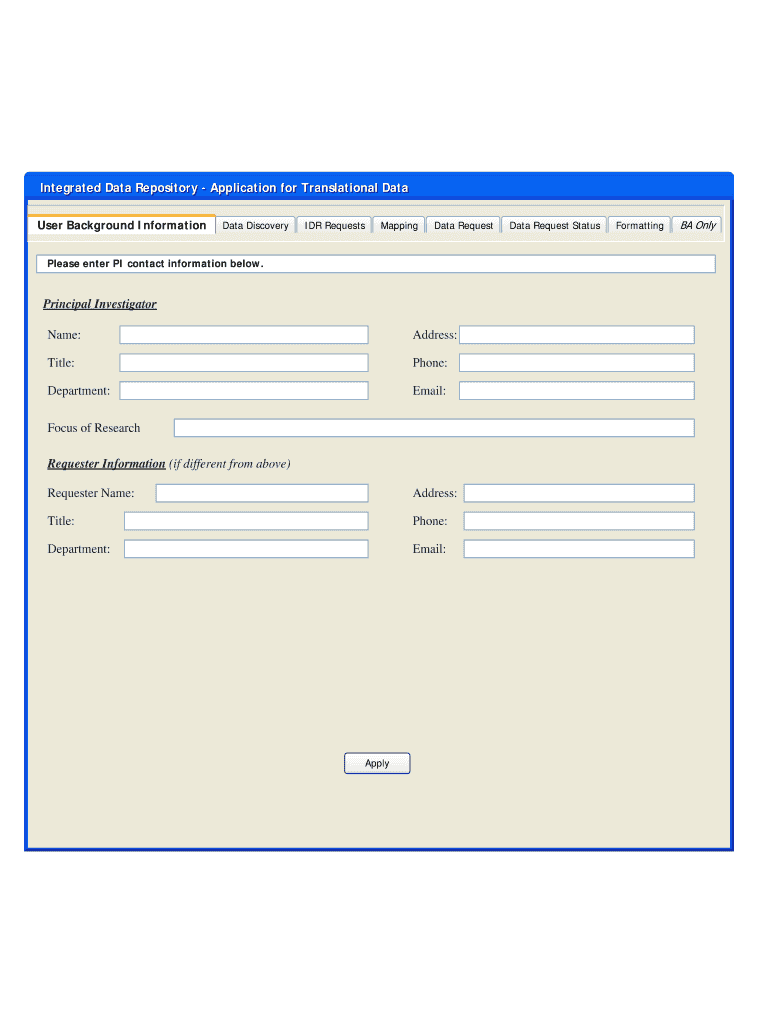
Fillable fields for personal information, including the Principal Investigator Name and Address.
-
Options for specifying the type of research focus needed for effective data gathering.
-
Selection options for various data systems and information types required.
-
IRB approval form integration to ensure compliance with research standards.
-
Check boxes for easy selection of demographic information and healthcare data requests.
Who Needs the Integrated Data Repository - Application for Translational Data?
The target users of the Integrated Data Repository application include principal investigators and research requesters. These individuals play a vital role in healthcare research by submitting requests for access to necessary data. Understanding the significance of IRB approval is essential, as it secures ethical standards and patient confidentiality in the data request process.
While primarily utilized in academic and clinical research settings, this application can also accommodate various research scenarios, encouraging a broader adoption among healthcare providers and researchers, including those focused on demographic information requests.
How to Fill Out the Integrated Data Repository - Application for Translational Data Online
Filling out the Integrated Data Repository application correctly involves several steps:
-
Access the application online through the designated platform.
-
Begin by entering the Principal Investigator Name in the specified field.
-
Input your Address, Title, Phone, Department, and Email as prompted.
-
Complete the Requester Information fields with accurate details.
-
Follow the instructions for selecting the necessary data types and systems.
Attention to detail is crucial during this process, ensuring all sections are accurately filled out and any required document uploads are included.
Common Errors and How to Avoid Them
Users can encounter several common errors when completing the Integrated Data Repository application. Typical mistakes include:
-
Entering incomplete information in required fields.
-
Forgetting to check IRB approval requirements before submission.
-
Misunderstanding the specific data types needed for the research.
To avoid these pitfalls, ensure that all necessary details are included, and review the application before submitting to confirm its accuracy. This diligence helps facilitate a smoother approval process.
Submission Methods and Delivery for the Integrated Data Repository - Application for Translational Data
Submitting the Integrated Data Repository application can be accomplished through various methods, ensuring flexibility and convenience:
-
Online submission via the application's dedicated portal.
-
Email submission of completed applications and supporting documents.
-
Postal mail for physical submissions when required.
After submitting the application, users can expect follow-up communications and processing times detailed in the submission guidelines.
Security and Compliance for the Integrated Data Repository - Application for Translational Data
The security of user data during the application process is a top priority. Data protection measures in place include advanced encryption and strict compliance with relevant regulations such as HIPAA and GDPR. Understanding these measures is critical for maintaining confidentiality while handling sensitive clinical data requests.
Research participants and data subjects can trust that their privacy is safeguarded throughout the submission and data retrieval processes.
Why Use pdfFiller for Your Integrated Data Repository Application?
pdfFiller provides a user-friendly platform to streamline the application process. By using pdfFiller, researchers can easily complete the Integrated Data Repository application while benefiting from features such as:
-
Editing capabilities for filling out forms and correcting entries.
-
eSigning options for secure document approval.
-
Conversion tools to change document formats as needed.
This seamless experience enables researchers to submit their applications efficiently, leveraging the comprehensive document management capabilities of pdfFiller.
Final Steps After Submission of the Integrated Data Repository - Application for Translational Data
After submitting the application, users should take specific steps to ensure their request is properly tracked and followed up:
-
Check the application status through the provided tracking link.
-
Prepare for any follow-up actions as instructed within the confirmation email.
Being proactive in these final steps can greatly enhance the chances of a timely review and approval of your application.
How to fill out the IDR Data Request
-
1.Start by accessing the Integrated Data Repository form on pdfFiller. Use the search bar to enter the form name or navigate through the healthcare forms category.
-
2.Once the form is open, familiarize yourself with the fillable fields. You will see sections labeled for 'Principal Investigator Name,' 'Address,' 'Title,' 'Phone,' 'Department,' and 'Email.'
-
3.Before filling the form, gather all required information, including contact details of the principal investigator and any relevant research protocols. Ensure you have all necessary demographic data and objectives stated.
-
4.As you fill in each field, click on the text boxes to input your information directly. Use the checkboxes to select areas of interest and data systems relevant to your request.
-
5.After completing all sections, carefully review your entries. Ensure that all information is accurate and corresponds with the requirements set by the IRB.
-
6.To finalize the form on pdfFiller, utilize the preview option to check how the completed document looks. Make any adjustments if necessary.
-
7.Once satisfied, either download a copy of the filled form for your records or submit it directly through pdfFiller's submission options, following any additional guidelines for your institution.

Who is eligible to fill out the Integrated Data Repository form?
Eligible users include principal investigators and researchers conducting studies that require access to clinical and demographic data. Confirming IRB approval is typically necessary before submission.
What supporting documents are required when submitting the form?
You typically need to include a copy of your IRB approval letter. Additional documents may be required based on the specifics of your request and institutional policies.
What is the processing time for form requests?
Processing times can vary, but generally, you should expect a response within a few weeks. It's best to check directly with the data repository for specific timelines.
How do I submit the Integrated Data Repository form?
You can submit the form directly through pdfFiller after completion or download it and submit it via email or mailing as instructed by your institution's guidelines.
What common mistakes should I avoid when filling out the form?
Common mistakes include leaving required fields blank, providing inaccurate contact information, or failing to attach necessary supporting documents. Double-check all entries for completeness.
Can I edit the form after submitting?
Once submitted, changes might not be possible unless you reach out directly for amendments. Always review your form before submission.
Is there a fee for processing this application?
There may be processing fees associated with data requests, depending on your institution’s policies. It is advisable to inquire about any fees when submitting your form.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.






















