Last updated on Dec 22, 2014

Get the free FDA Approved Investigational Device Exemption (IDE) Pre-Approval Data Submission Req...
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is FDA IDE Request
The FDA Approved Investigational Device Exemption (IDE) Pre-Approval Data Submission Request is a healthcare form used by providers to submit documentation for investigational device approval.
pdfFiller scores top ratings on review platforms




Who needs FDA IDE Request?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to FDA IDE Request
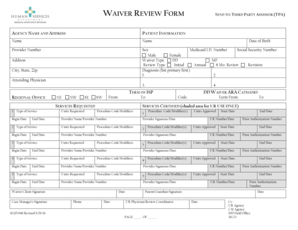
What is the FDA Approved Investigational Device Exemption (IDE) Pre-Approval Data Submission Request?
The FDA IDE form is a critical document used in the medical device approval process, specifically designed to facilitate the submission of important documentation to regulatory bodies. It serves as a formal request for an Investigational Device Exemption (IDE), allowing healthcare providers to obtain necessary approvals for investigational devices. The significance of the FDA IDE request form lies in its structured format, which ensures that all essential information is provided comprehensively.
This form plays an essential role within the healthcare industry as it standardizes documentation requirements and processes, ultimately contributing to successful device approvals. Moreover, it establishes a direct connection to regulatory entities like Noridian Healthcare Solutions, LLC, guiding healthcare providers through the complexities of device submissions.
Purpose and Importance of the FDA IDE Pre-Approval Data Submission Request
The FDA IDE Pre-Approval Data Submission Request serves several crucial purposes for healthcare providers seeking investigational device approval. Firstly, it streamlines the approval process by providing a consolidated approach to submitting all necessary documentation required by regulatory authorities. Utilizing this form can significantly enhance the chances of obtaining timely approvals, benefiting healthcare practices and patient care.
Another vital aspect of this form is the emphasis it places on compliance and proper documentation. Maintaining consistency in submissions fosters trust and facilitates smoother communication with regulatory bodies, ensuring that submissions are thoroughly reviewed and considered. Proper documentation is not only imperative for compliance but also directly impacts the overall success of the approval process.
Who Needs to Use the FDA IDE Pre-Approval Data Submission Request?
The FDA IDE Pre-Approval Data Submission Request is essential for various roles within the healthcare landscape, primarily involving providers and their contact personnel. The key roles required for completing this form include Providers, as well as both Primary and Secondary Contact Persons, each playing a vital role in the application process. These individuals must be adequately informed about the device and the specific requirements outlined in the form.
Healthcare professionals—including clinicians, researchers, and administrators—may also need to participate in the process to ensure all pertinent information is submitted. Eligibility extends to those who are directly involved in patient care and device management, reinforcing the need for interdisciplinary collaboration in the approval journey.
Required Documents and Supporting Materials for Submission
To successfully complete the FDA IDE Pre-Approval Data Submission Request, several mandatory documents must accompany the submission. These include:
-
FDA approval letters that confirm prior approvals.
-
Institutional Review Board (IRB) approval forms to assure ethical compliance.
-
Patient consent forms outlining participation rights and disclosure.
-
Billing codes relevant to the investigational device for insurance and billing purposes.
It is crucial for providers to gather these documents before starting the form to avoid delays and ensure a comprehensive and accurate submission process.
How to Fill Out the FDA IDE Pre-Approval Data Submission Request Online
Completing the FDA IDE Pre-Approval Data Submission Request online is straightforward when following specific steps. First, navigate to the fillable form accessible via pdfFiller. Users can easily upload the document without additional downloads.
Field-by-field instructions provide clarity, explaining complex terms or sections to reduce confusion. A review and validation checklist is highly recommended to catch common errors, ensuring a complete and accurate submission. The following steps can enhance the completion process:
-
Access the form and gather all necessary documents.
-
Fill in all required fields with precise and accurate information.
-
Review each section for completeness and accuracy.
-
Use the validation checklist to cross-check entries before submission.
Submission Methods and Delivery for the FDA IDE Pre-Approval Data Submission Request
Once the FDA IDE Pre-Approval Data Submission Request is completed, users have several submission methods available. Options typically include online submissions via the regulatory agency’s platform, mail, or in-person deliveries. It is essential to understand the preferred submission method for the relevant regulatory body.
Users should also be aware of submission address details and any deadlines for timely processing. Tracking submission status or confirming receipt is equally important, ensuring that documentation is accounted for without confusion. Make sure to follow specific instructions regarding submission requirements carefully.
Security and Compliance When Using the FDA IDE Pre-Approval Data Submission Request
Handling sensitive information during the FDA IDE Pre-Approval Data Submission Request submissions necessitates strict security measures. pdfFiller, as a document management platform, is committed to implementing robust data protection protocols to ensure compliance with HIPAA regulations. This underscores the importance of maintaining patient privacy while processing sensitive medical documentation.
Implementing privacy measures not only protects personal and medical information but also fortifies trust in the submission process. Security protocols are paramount in the context of healthcare documentation, safeguarding against potential data breaches while ensuring compliance with relevant regulations.
Common Issues and Solutions When Using the FDA IDE Pre-Approval Data Submission Request
Users may encounter various challenges while navigating the FDA IDE Pre-Approval Data Submission Request. Common rejection reasons include incomplete forms, missing documentation, or lack of proper signatures. To avoid these pitfalls, ensure all required information is thoroughly reviewed prior to submission.
In cases of rejected submissions, users have pathways to correct or amend their applications to rectify mistakes or provide additional information. Understanding the renewal or resubmission process can significantly alleviate stress, allowing users to track their application status methodically.
Utilizing pdfFiller for a Seamless FDA IDE Pre-Approval Data Submission Experience
pdfFiller enhances the experience of completing the FDA IDE Pre-Approval Data Submission Request through a variety of features. Users can benefit from the platform’s capabilities to create, edit, and securely eSign forms without difficulty. The user-friendly interface streamlines the documentation process, making it easier to manage submissions effectively.
By leveraging these features, healthcare providers can simplify the overall form management process, ensuring swift, secure, and efficient document handling throughout the FDA IDE submission experience.
How to fill out the FDA IDE Request
-
1.Begin by accessing the FDA Approved Investigational Device Exemption (IDE) Pre-Approval Data Submission Request form on pdfFiller. Use the search function to locate the specific form.
-
2.Once opened, navigate through the form using pdfFiller’s user-friendly interface. You will see various fillable fields where you can input data.
-
3.Before starting, gather all necessary information, including device information, FDA approval letters, IRB approval documentation, study protocols, patient consent forms, and relevant billing codes.
-
4.Carefully fill in each section of the form, ensuring you provide accurate information. Use the guide prompts, if available, to assist in completing each section.
-
5.Review your entries as you progress. Take advantage of the review highlights within pdfFiller to double-check for any incomplete fields or errors.
-
6.Once all information is filled out, go through the entire form one last time to finalize your entries and confirm their accuracy.
-
7.After ensuring everything is correct, you can save your work, download the completed form, or submit it directly through pdfFiller using the submit options available.

Who is eligible to use this form?
The FDA Approved IDE Pre-Approval Data Submission Request form is intended for healthcare providers and their designated contacts involved in submitting documentation for investigational devices.
Are there any deadlines for submitting the form?
Deadlines can vary depending on specific clinical protocols and regulatory timelines. It’s crucial to check with the FDA or relevant institutions for any submission deadlines relevant to your investigation.
How do I submit the completed form?
You can submit the completed form directly through pdfFiller or download it for submission via email or mail, depending on the submission guidelines of the FDA or your institution.
What supporting documents are required with this form?
Supporting documents typically include FDA approval letters, IRB approvals, study protocols, patient consent forms, and relevant medical billing codes. Ensure all required documents are gathered before submission.
What are common mistakes to avoid when filling out this form?
Common mistakes include incomplete fields, missing required signatures, and not including necessary supporting documents. Always double-check all entries before submission.
What are the processing times for submissions?
Processing times can vary based on the complexity of the submission and the review workload of the FDA. Plan for variability, and always follow up if there are urgent needs.
Can the form be notarized?
No, this form does not require notarization as stated in its metadata. Ensure you follow the signature and approval requirements outlined by your institution.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.