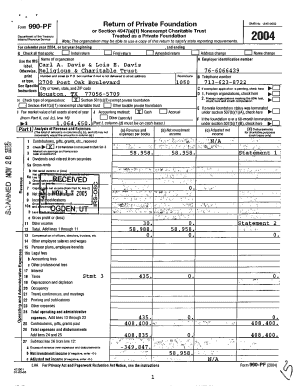
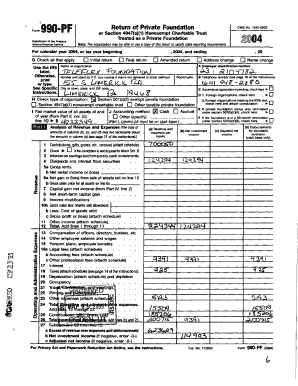
Last updated on Jan 18, 2015

Get the free Step by Step Guide to SPE Method Development
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is SPE Method Guide
The Step by Step Guide to SPE Method Development is a service agreement form used by laboratories to develop solid-phase extraction methods.
pdfFiller scores top ratings on review platforms




Who needs SPE Method Guide?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to SPE Method Guide
What is the Step by Step Guide to SPE Method Development?
The Step by Step Guide to SPE Method Development is a crucial resource for laboratories and researchers involved in sample preparation using solid-phase extraction (SPE). This guide highlights the importance of a structured approach in developing SPE methods, ensuring that researchers can optimize their analyte extraction processes effectively. By providing a comprehensive framework, the guide facilitates accurate method development and enhances laboratory efficiency.
Solid-phase extraction plays a pivotal role in simplifying the sample preparation task, enabling researchers to focus on critical aspects of their analysis. This guide serves as an essential tool, equipping users with the necessary techniques to refine their SPE methodologies.
Purpose and Benefits of Using the SPE Method Development Guide
The SPE Method Development Guide is designed to address the needs of laboratories and researchers aiming for precision in method development. By employing a structured approach outlined in this guide, users can enjoy numerous advantages that enhance their workflow.
Among the key benefits is the simplification of the analyte extraction process, which not only improves accuracy but also streamlines the overall methodology. This organized format allows users to focus on critical parameters while minimizing potential errors in sample preparation.
Key Features of the Step by Step Guide to SPE Method Development
The guide provides several key features that stand out as beneficial for users. It includes:
-
Fillable fields for essential information such as 'Sample name' and 'Analyte of interest.'
-
Checkboxes for confirming critical steps in the process.
-
Charts that assist with column conditioning and sample loading.
-
An overview of steps crucial to method development, fostering a thorough understanding of SPE.
Overall, these features facilitate efficient analyte extraction and contribute to the development of SOPs within laboratory settings.
Who Needs the Step by Step Guide to SPE Method Development?
This guide targets a diverse audience within the scientific community. Professions that would significantly benefit from utilizing the SPE Method Development Guide include laboratory technicians, researchers, and academic professionals.
These individuals, working across various sectors, will find this guide invaluable in achieving high standards in sample preparation, thereby ensuring reliable results in their analytical processes.
Field-by-Field Instructions for Completing the SPE Method Development Guide
To utilize the SPE Method Development Guide effectively, users must adhere to detailed instructions while filling out the form. Key fields include:
-
Sample name: Ensure the correct sample name is entered for identification.
-
Analyte of interest: Clearly specify the analyte to avoid ambiguity.
Before filling out the form, users should gather all necessary documentation and information to facilitate a smooth completion process, ensuring accuracy and efficiency.
Digital Completion of the SPE Method Development Guide using pdfFiller
pdfFiller offers a user-friendly platform for completing the SPE Method Development Guide efficiently. This tool streamlines the form-filling process with features that allow for editing and eSigning, catering to users' needs seamlessly.
Additionally, pdfFiller prioritizes the security of sensitive laboratory documents through industry-standard measures, such as 256-bit encryption, to safeguard users' data and comply with applicable privacy regulations.
Review and Validation Checklist for Your Completed Form
Before submitting the completed SPE Method Development Guide, users should conduct a thorough review to ensure all information is accurate. It is important to avoid common errors that may compromise data quality.
A useful checklist includes:
-
Ensure all fillable fields are completed with accurate information.
-
Cross-verify filled fields with original documents to confirm accuracy.
-
Attach any necessary supplemental documentation as required.
Following this checklist can help users reinforce the integrity of their submissions.
Submission Methods and Next Steps After Completing the Form
Once the SPE Method Development Guide is complete, users have multiple submission options available. These options include online submissions and in-person deliverance of the completed form.
Users should also familiarize themselves with required documentation for submission and anticipate possible follow-up actions, ensuring they can track their submissions effectively.
Ensuring Security and Compliance with SPE Method Development Documentation
Maintaining data security and compliance is paramount when handling the SPE Method Development Guide. pdfFiller adheres to rigorous encryption standards and compliance with laws such as HIPAA and GDPR, assuring users that their sensitive data is well-protected.
Best practices for handling sensitive information should always be considered, particularly during the form-filling process. This focus on security fosters trust and compliance in laboratory documentation practices.
Maximize Your Efficiency with the Step by Step Guide to SPE Method Development
The Step by Step Guide to SPE Method Development, when combined with the capabilities of pdfFiller, maximizes user efficiency in laboratory practices. This digital platform offers ease of access and effective document management, catering to the specific needs of researchers and technicians alike.
By leveraging this guide alongside pdfFiller, users can elevate their productivity and ensure a streamlined approach toward method development, ultimately contributing to enhanced outcomes in the laboratory setting.
How to fill out the SPE Method Guide
-
1.Access pdfFiller and search for 'Step by Step Guide to SPE Method Development'. Click to open the form in the editing interface.
-
2.Familiarize yourself with the fillable fields and checkboxes provided, including sections like 'Sample name' and 'Analyte of interest'.
-
3.Gather all necessary information, including sample details, types of analytes, sorbents to be used, and any previous method information relevant to your experiment.
-
4.Start filling in the form by clicking on the appropriate fields to enter your details. Use the guidance provided for each section to ensure accuracy.
-
5.Once all fields are filled, review the form thoroughly to verify that all information is correct and complete.
-
6.Utilize pdfFiller's tools to make any necessary edits or adjustments before finalizing the document.
-
7.After completing your review, save your work by clicking the save button. You may also download the file or submit it directly through pdfFiller.

Who can use the Step by Step Guide to SPE Method Development?
This form is designed for laboratory researchers and professionals involved in method development, including those in environmental science, pharmaceuticals, and academia.
Is there a deadline for submitting this guide?
While there is typically no formal deadline for internal documents like this, it is advisable to complete your method development promptly to maintain research timelines.
How do I submit the completed form?
You can submit the completed form directly through pdfFiller, or download it for manual submission to your supervisor or regulatory body.
Are there supporting documents required with this form?
Commonly, additional data on sample preparation and previous method performance may be helpful when using this guide, although it typically does not require formal attachments.
What are some common mistakes to avoid when filling this form?
Ensure all sections are completed fully, avoid vague descriptions, and double-check sample names and analyte details to prevent errors in method execution.
How long does it take to process this method development?
Processing time varies but typically includes some preliminary testing and adjustments; allow at least a few days to finalize your method depending on complexity.
Can I edit the form after initially filling it in?
Yes, pdfFiller allows you to edit the form as needed until you are satisfied with the information and ready to finalize it.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.