Last updated on Apr 25, 2026

Get the free EU Cosmetic Product Serious Undesirable Effect Notification
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is SUE Form A
The EU Cosmetic Product Serious Undesirable Effect Notification is a healthcare form used by responsible persons and distributors to report serious undesirable effects (SUE) related to cosmetic products in the EU.
pdfFiller scores top ratings on review platforms




Who needs SUE Form A?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to SUE Form A
Understanding the EU Cosmetic Product Serious Undesirable Effect Notification
The EU Cosmetic Product Serious Undesirable Effect Notification is crucial for ensuring compliance with regulations surrounding cosmetics. A serious undesirable effect (SUE) refers to significant adverse reactions caused by cosmetic products, which can have serious implications for consumer safety. Regulation (EC) No 1223/2009 underscores the importance of safety in cosmetic products, mandating reporting to safeguard users.
SUE Form A serves as a vital tool for Responsible Persons and Distributors, enabling them to document and report these serious undesirable effects succinctly and methodically.
Benefits of the EU Cosmetic Product Serious Undesirable Effect Notification
Utilizing the SUE Form A is not merely compliance; it reflects a dedication to consumer safety. Reporting serious undesirable effects is a legal obligation and serves to enhance overall product safety. Furthermore, it plays a pivotal role in protecting brand reputation and trust, contributing to a transparent cosmetic market.
-
Fulfills legal reporting obligations under EU law.
-
Enhances consumer safety by addressing potential health risks promptly.
-
Timely reporting can prevent serious health consequences.
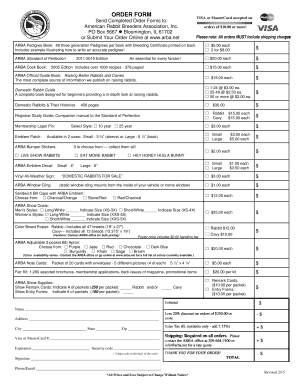
Key Features of the SUE Form A Template
The SUE Form A template is equipped with several essential components for effective reporting. It includes fillable sections where users must provide company details, event descriptions, and evidence surrounding the reported SUE.
-
Fields for company information and specific event details.
-
Checkboxes for selecting the type of report: Initial, Follow-up, or Final.
-
Evidence requirements that guide the thorough documentation of the adverse effects.
Who Should Use the EU Cosmetic Product Serious Undesirable Effect Notification?
The primary users of the SUE Form A are Responsible Persons and Distributors, essential stakeholders in the cosmetic industry. Competent Authorities also play a significant role, overseeing the integrity and accuracy of the reported submissions.
Various stakeholders share obligations under EU regulations, ensuring that the issue of serious undesirable effects is communicated effectively through proper reporting channels.
How to Complete the EU Cosmetic Product Serious Undesirable Effect Notification Online
Filling out the SUE Form A online is a straightforward process when attention is given to each section. Users should break down the form sections, clearly understanding the information required for accurate completion.
-
Identify necessary details for each form section carefully.
-
To avoid common mistakes, review guidance on each field provided within the form.
-
Consider using pdfFiller for assistance in filling and submitting forms smoothly.
Submission Process for the EU Cosmetic Product Serious Undesirable Effect Notification
Submitting the SUE Form A is crucial for compliance, and various methods are available to do so. Users can choose online submission, email, or traditional mailing based on their preference.
-
Clarify submission deadlines to ensure timely reporting.
-
Be aware of any potential fees that might be associated with submission.
-
Track submission status through confirmation options provided post-submission.
What Happens After You Submit the SUE Form A?
After submission, the processing of the SUE Form A adheres to established timelines. Users can expect structured follow-ups concerning the status of their reports, which helps maintain communication with identified authorities.
-
Regular updates regarding the processing timeline are typical.
-
Check the status of submitted forms as needed to stay informed.
-
Follow-up actions may be required depending on feedback from the authorities.
Security and Privacy Considerations for the EU Cosmetic Product Serious Undesirable Effect Notification
Security and privacy are paramount when submitting sensitive information regarding serious undesirable effects. pdfFiller incorporates robust security measures to protect users' data throughout the submission process. Compliance with GDPR and relevant regulations ensures user confidentiality and data integrity.
Utilizing pdfFiller for Your EU Cosmetic Product Serious Undesirable Effect Notification Needs
To efficiently create and manage your SUE Form A submissions, pdfFiller offers an invaluable resource. The platform provides users with easy access to form creation, editing, and submission features from any internet-enabled device.
-
Experience secure document handling with advanced encryption.
-
Utilize cloud-based features for easy access and collaboration.
-
Enjoy a user-friendly interface that simplifies the form-filling experience.
How to fill out the SUE Form A
-
1.Access pdfFiller and search for 'EU Cosmetic Product Serious Undesirable Effect Notification' in the template library.
-
2.Open the document to view the fillable form interface. Familiarize yourself with the sections provided.
-
3.Before starting, gather all necessary information regarding the serious undesirable effect, including product details and company contact information.
-
4.Begin by filling out the 'Company report number' and 'Type of report' fields; use clear and accurate information.
-
5.Proceed to the 'Date received by company’ section, ensuring to input the exact date you first learned about the SUE.
-
6.Utilize the checkboxes to indicate whether the report is 'Initial', 'Follow-up', or 'Final' appropriately.
-
7.Complete the sections detailing the serious undesirable effect, including symptoms, dates, and user complaints.
-
8.Once all fields are filled, review your entries for accuracy using pdfFiller's preview function to avoid errors.
-
9.Finalize the form by saving it and selecting the appropriate download option. You can also submit the form directly through pdfFiller if applicable.

Who is eligible to submit the SUE form?
The SUE form is primarily for responsible persons, distributors, and any entities involved in the cosmetic product supply chain that have knowledge of a serious undesirable effect.
Are there deadlines for submitting the SUE form?
Typically, the SUE form should be submitted promptly upon awareness of the serious undesirable effect, ideally within a few days to comply with regulatory requirements.
How can I submit the completed SUE form?
You can submit the completed SUE form via email or through the relevant online portal designated by your Competent Authority, as outlined in local regulations.
What supporting documents are required with the SUE form?
Supporting documents may include product safety assessments, consumer complaints, or additional scientific evidence of the reported undesirable effect.
What common mistakes should I avoid when completing the SUE form?
Common mistakes include incomplete sections, incorrect dates, and failing to include all necessary supporting information. Always double-check your entries.
How long does it take to process the SUE form after submission?
Processing times can vary among Competent Authorities, but expect it to take several weeks for review and feedback once submitted.
What should I do if I encounter issues while filling out the form?
If you experience issues, consider consulting the pdfFiller help section or reach out to regulatory bodies for guidance on completing the SUE form accurately.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.