Last updated on May 6, 2015

Get the free FDA Prior Notice Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is FDA Prior Notice
The FDA Prior Notice Form is a government document used by importers to provide the U.S. FDA with advance information about imported food shipments.
pdfFiller scores top ratings on review platforms




Who needs FDA Prior Notice?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to FDA Prior Notice
What is the FDA Prior Notice Form?
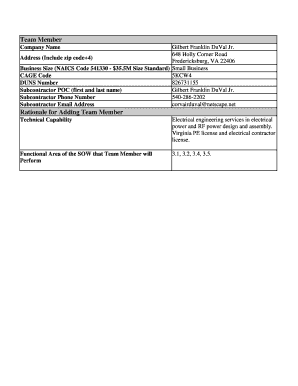
The FDA Prior Notice Form is an essential document for anyone importing food products into the United States. This form is designed to facilitate communication with the U.S. Food and Drug Administration (FDA) about incoming food shipments. Its purpose is not only regulatory but also critical in ensuring food safety standards are met. The submitter, often a representative of the importing entity, is responsible for completing and submitting this form to the FDA, making it a crucial component of the FDA import process.
Why is the FDA Prior Notice Form Necessary?
Filing the FDA Prior Notice Form is vital for maintaining compliance with food safety regulations enforced by the FDA. The FDA plays a key role in evaluating imported food products to ensure they meet safety standards. Failure to file this form can lead to serious consequences, including shipment delays at customs and potential penalties. Understanding the necessity of this form helps importers avoid complications and ensures that food products can flow smoothly into the U.S. market.
Who Needs to Complete the FDA Prior Notice Form?
Several key roles participate in the completion of the FDA Prior Notice Form. The primary signers include:
-
Submitter - the individual submitting the form.
-
Shipper - responsible for sending the food products.
Optional signers may include the ultimate consignee, who receives the shipment, and the manufacturer or producer of the imported goods. Understanding these roles is crucial for ensuring that the form is completed accurately and submitted in accordance with FDA requirements.
When to Submit the FDA Prior Notice Form
Timeliness is critical when filing the FDA Prior Notice Form. Importers should submit the form at least two hours before the food products arrive at the U.S. border. Additionally, some states may have specific regulations that affect the timing of submissions. It is advisable for importers to be aware of these state-specific rules to avoid any potential issues with their food shipments.
Essential Information Required for the FDA Prior Notice Form
Completing the FDA Prior Notice Form requires detailed information to ensure compliance. Key fields that must be filled out include:
-
Name
-
Firm Name
-
Address
-
Phone Number
-
E-mail
-
FDA Registration Number
-
Description of Item
-
FDA Product Code
Additionally, important documents such as the lot or can code, number of packages, and weight of packages (both in pounds and kilograms) should accompany the form to provide comprehensive product details.
How to Fill Out the FDA Prior Notice Form Online
Utilizing pdfFiller simplifies the process of completing the FDA Prior Notice Form. The platform offers a step-by-step guide to ensure users can fill out each field correctly. With user-friendly features, pdfFiller streamlines the submission process, allowing for efficient input of details such as the product description, FDA registration number, and more. Field-by-field guidance helps eliminate errors and ensures compliance with FDA regulations.
Common Mistakes to Avoid When Completing the FDA Prior Notice Form
Many users inadvertently make common mistakes while completing the FDA Prior Notice Form. To ensure accuracy, it is essential to:
-
Double-check all required fields for completeness.
-
Validate the accuracy of the FDA Registration Number and other critical details before submission.
By paying attention to these common errors, importers can prevent delays and penalties associated with incomplete or inaccurate submissions.
How to Submit the Completed FDA Prior Notice Form
Once the FDA Prior Notice Form is completed, it can be submitted through various methods. Importers may opt to submit the form online or via mail. It is crucial to confirm the receipt of the form by the FDA post-submission. This confirmation ensures that the form has been processed and prevents any issues with food shipments arriving in the U.S.
Safety and Security in Handling Your FDA Prior Notice Form
When submitting the FDA Prior Notice Form, data security must be a priority. pdfFiller employs robust security measures, including 256-bit encryption, to protect user information during the submission process. Compliance with regulations such as HIPAA and GDPR ensures that sensitive information remains secure both during and after the submission process.
Your Next Steps with pdfFiller to Complete the FDA Prior Notice Form
By choosing pdfFiller, users can experience the benefits of a secure and efficient platform for filling out the FDA Prior Notice Form. The features offered, such as eSigning and document management, enhance the overall user experience. Importers are encouraged to explore how pdfFiller can simplify the completion of the FDA Prior Notice Form, making the process seamless and compliant.
How to fill out the FDA Prior Notice
-
1.To access the FDA Prior Notice Form, go to pdfFiller and search for the form by typing its name in the search bar.
-
2.Once the form appears, click on it to open the interactive editing interface where you'll see fillable fields and instructions.
-
3.Before starting, gather necessary information such as shipment details, manufacturer information, product codes, and contact details for all parties involved.
-
4.Begin filling in the blank fields by entering required data like 'Name:', 'Firm Name:', 'Address:', and 'Phone:'. Remember to double-check the accuracy of each entry.
-
5.Make sure to fill in the 'FDA Reg#', 'Description of Item', 'FDA Product Code', 'Lot or Can Code#', 'Number of Packages', and 'Weight of Packages LB/KG'.
-
6.Indicate the 'Country of Origin' and ensure that all information reflects what is present on your product documentation.
-
7.Leave the signature line to sign later — check that the box confirming accuracy is checked.
-
8.Once completed, review all provided information for accuracy and completeness using pdfFiller's preview feature.
-
9.To finalize the form, click on the save option, and choose to download or submit the form directly through pdfFiller.
-
10.Consider signing electronically through the platform if applicable and ensure to keep a copy for your records.

Who is eligible to use the FDA Prior Notice Form?
Any individual or entity involved in the importation of food products into the U.S. is eligible to use the FDA Prior Notice Form, including food importers, shippers, and consignees.
What is the deadline for submitting the FDA Prior Notice Form?
The FDA Prior Notice Form must be submitted at least 2 hours before the food product arrives at the U.S. border to allow for processing and compliance checks.
How should I submit the completed FDA Prior Notice Form?
You can submit the FDA Prior Notice Form electronically through pdfFiller or directly through the FDA’s online portal. Ensure all fields are correctly filled before submission.
What supporting documents are required when submitting this form?
While the FDA Prior Notice Form itself does not require additional documents, it is advisable to have shipping invoices, product documentation, and registration details ready for reference.
What are common mistakes to avoid when filling out the form?
Common mistakes include omitting required fields, misspelling product details, and not checking the accuracy box. Always review your entries before submission to avoid these issues.
How long does it take for the FDA to process the Prior Notice Form?
The FDA typically processes the Prior Notice Form within 2 hours, provided the submission is complete and arriving during operational hours.
Is there a fee associated with submitting the FDA Prior Notice Form?
No, there is no fee required to submit the FDA Prior Notice Form. It is a free process for compliance with import regulations.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.