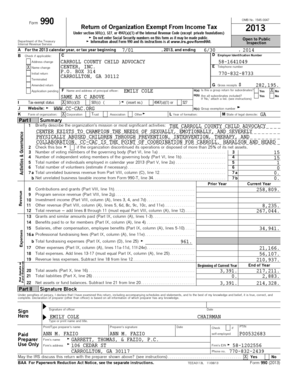Last updated on May 28, 2015

Get the free Registration Certificate for In Vitro Testing with Byproduct Material
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is In Vitro Testing Registration
The Registration Certificate for In Vitro Testing with Byproduct Material is a government form used by healthcare providers to register for the possession and use of byproduct material in clinical tests.
pdfFiller scores top ratings on review platforms




Who needs In Vitro Testing Registration?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to In Vitro Testing Registration
What is the Registration Certificate for In Vitro Testing with Byproduct Material?
The Registration Certificate for In Vitro Testing with Byproduct Material serves as a crucial document for healthcare professionals and institutions. This certificate defines the legal framework allowing physicians, clinical laboratories, and hospitals to possess and use specific byproduct materials for lab testing.
Its significance lies in adhering to the regulations set forth by the US Atomic Energy Commission, ensuring that all involved parties operate within compliant parameters for safety and accountability.
Purpose and Benefits of Using the Registration Certificate for In Vitro Testing
Utilizing the Registration Certificate for In Vitro Testing offers multiple advantages that enhance operational efficiency for healthcare providers. By completing this registration, professionals gain legal possession and usage rights needed for byproduct materials essential for laboratory tests.
Moreover, the registration ensures compliance with relevant regulatory frameworks, streamlining processes associated with clinical applications and improving overall patient safety.
Who Needs the Registration Certificate for In Vitro Testing?
This registration is essential for various stakeholders within the medical community. The primary target audience includes:
-
Physicians: Responsible for ordering tests that require byproduct materials.
-
Clinical Laboratories: Often perform the tests and must comply with safety and regulatory standards.
-
Hospitals: Need to ensure access to necessary materials for testing and patient diagnostics.
Specific instances when registration is required typically occur during the initial setup of facilities or when new byproduct materials are introduced.
Eligibility Criteria for the Registration Certificate for In Vitro Testing
Applicants seeking the Registration Certificate must meet specific qualifications and compliance requirements. It is essential to adhere to 10 CFR 31.11 standards, which outline vital safety regulations pertaining to the use of byproduct materials.
Ensuring adherence to these eligibility criteria is critical for maintaining lawfulness and safety in all laboratory operations.
How to Fill Out the Registration Certificate for In Vitro Testing Online
Filling out the Registration Certificate online can be accomplished effectively with tools such as pdfFiller. To complete the form successfully, follow these steps:
-
Access the online registration form through pdfFiller.
-
Carefully fill in required fields including your name and address.
-
Provide necessary supporting documentation as indicated.
Ensuring accuracy in your submission is paramount; any discrepancies may delay processing and lead to complications in validation.
Common Errors and How to Avoid Them
Avoiding common pitfalls during the registration process is essential for smooth submission. Frequent mistakes include:
-
Filling out forms inadequately or incorrectly.
-
Failing to review documentation before submission.
Confirming accuracy through a thorough review checklist is advised to help ensure the application's success and completeness.
Submission Methods and Tracking Your Registration Certificate
Once the registration form is completed, there are multiple submission methods available. You can choose between online submission through pdfFiller or traditional mail. Regardless of the method, it is crucial to retain confirmation of submission and track your application's status post-submission.
Being diligent about these details will facilitate smoother interactions with the U.S. Atomic Energy Commission throughout the processing period.
What Happens After You Submit the Registration Certificate
After a submission is made, individuals can expect a processing timeline regarding the registration certificate. Typically, notifications are provided from the U.S. Atomic Energy Commission concerning the status of their application.
If adjustments are necessary, it is essential to respond promptly to any requirements for correction or amendment.
Renewal or Resubmission of the Registration Certificate for In Vitro Testing
For those needing to renew or resubmit their Registration Certificate, specific guidelines are set in place. Renewal of the registration certificate should happen within the defined timelines to maintain compliance. Familiarizing yourself with the renewal process can significantly aid in preventing lapses in legal possession and use of byproduct materials.
Failure to renew on time may lead to potential legal complications or interruptions in your testing capabilities.
Experience the Convenience of Using pdfFiller for Your Registration Certificate
Utilizing pdfFiller can greatly simplify the process of completing and submitting your Registration Certificate for In Vitro Testing. The platform offers a host of features designed for ease of use, including the ability to edit, fill, and securely manage your documents.
With stringent security measures in place, you can trust that your sensitive information is handled safely, allowing you to concentrate on filling, modifying, and submitting forms seamlessly.
How to fill out the In Vitro Testing Registration
-
1.Begin by visiting pdfFiller and log into your account or create a new one if needed.
-
2.Use the search bar to locate the Registration Certificate for In Vitro Testing with Byproduct Material.
-
3.Once the form appears, click on it to open within the pdfFiller interface.
-
4.Carefully read the instructions included in the form to familiarize yourself with required information.
-
5.Prepare necessary details, such as the name and address of your practice or institution, and any specific compliance information.
-
6.Start filling in the blank fields within the form, ensuring all required items are accurately completed.
-
7.Utilize pdfFiller's tools to insert your information smoothly, employing drop-down menus and checkboxes where applicable.
-
8.Double-check that all entries are correct and match your supporting documents.
-
9.Once completed, review the entire form to ensure compliance with the necessary requirements.
-
10.Save your progress frequently by clicking the ‘Save’ button at the top right corner.
-
11.When satisfied, download the completed form for your records or directly submit it electronically through pdfFiller's submit feature.
-
12.If needed, make any necessary adjustments to the saved copy before final submission.

Who is eligible to use the Registration Certificate for In Vitro Testing?
Eligibility for this form includes physicians, clinical laboratories, and hospitals in the US that need to register to use byproduct material for in vitro testing. It is crucial to comply with local regulatory requirements.
What documents do I need to submit with the form?
Typically, supporting documents may include proof of your facility's address, compliance certifications, and any additional information that verifies your intent to use byproduct materials for testing.
What is the submission process for this registration certificate?
After filling out the form on pdfFiller, you can submit it electronically through their platform. Ensure all sections are completed properly before finalizing your submission.
Are there deadlines for submitting the Registration Certificate?
There are no specific deadlines mentioned for this form; however, timely submission is advised to maintain compliance with regulatory standards regarding the use of byproduct materials.
What are common mistakes to avoid when filling out this form?
Common mistakes include providing inaccurate information, missing required signatures, and failing to check for compliance with regulations. Always double-check entries before submission.
How long does it take to process the registration certificate?
Processing times can vary, but typically you can expect a response within several weeks after submission. It's important to follow up if you do not receive confirmation.
Do I need to notarize the Registration Certificate before submission?
No, notarization is not required for this form. However, ensure that all information is accurately presented to comply with regulatory standards.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.