Last updated on Mar 27, 2026
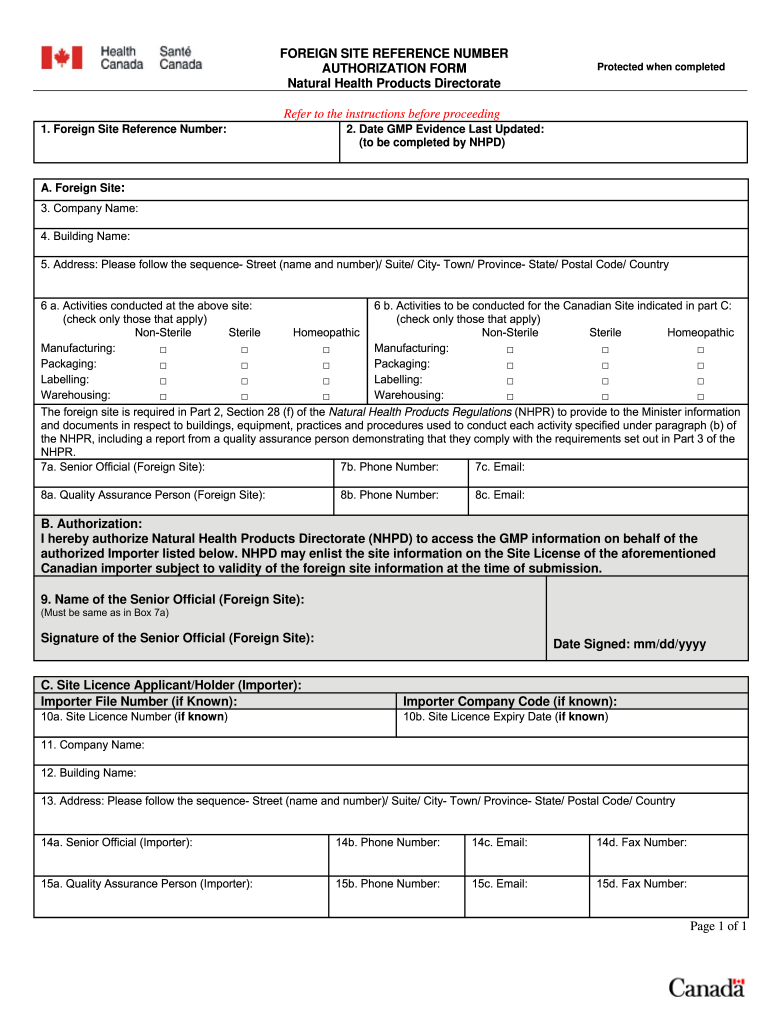
Canada Foreign Site Reference Number Authorization free printable template
pdfFiller is not affiliated with any government organization
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Canada Foreign Site Reference Number Authorization
The Foreign Site Reference Number Authorization Form is a document used by foreign sites registered with Health Canada to authorize Canadian importers to access their GMP information.
pdfFiller scores top ratings on review platforms




Who needs Canada Foreign Site Reference Number Authorization?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Canada Foreign Site Reference Number Authorization
What is the Foreign Site Reference Number Authorization Form?
The Foreign Site Reference Number Authorization Form serves a vital role in the healthcare sector, specifically for foreign sites registered with Health Canada. This form is essential for Canadian importers seeking access to good manufacturing practice (GMP) information, thereby ensuring compliance with regulations. It is crucial for any foreign site involved in the distribution of health products, facilitating the authorization process for Canadian importers.
Purpose and Benefits of the Foreign Site Reference Number Authorization Form
This form provides numerous benefits to both foreign sites and Canadian importers. Firstly, it enhances compliance with Health Canada regulations, streamlining the authorization process significantly. By properly documenting access to GMP information, the form helps ensure product quality and safety.
-
Facilitates quick authorization for foreign sites.
-
Streamlines the compliance process for Canadian importers.
-
Ensures the safety and quality of health products.
Who Needs the Foreign Site Reference Number Authorization Form?
Key stakeholders who must utilize this form include senior officials and quality assurance personnel from both foreign sites and Canadian importers. Each role plays a specific part in the authorization process, with responsibilities that are clearly outlined.
-
Senior Officials: Responsible for signing the form.
-
Quality Assurance Personnel: Ensure compliance with GMP standards.
To be eligible to complete this form, individuals must hold designated positions within their respective organizations.
How to Fill Out the Foreign Site Reference Number Authorization Form Online (Step-by-Step)
Filling out the Foreign Site Reference Number Authorization Form online can be easily achieved using pdfFiller. Here’s a simple guide to help you through the process:
-
Access the form on pdfFiller.
-
Complete the key sections: enter foreign site details and activities conducted.
-
Ensure all fields are accurately filled out before submission.
Double-checking your information will help avoid inaccuracies.
Field-by-Field Instructions for the Foreign Site Reference Number Authorization Form
Each fillable field in the form requires careful attention. Below are detailed instructions for filling out the critical sections:
-
Required information includes contact details for senior officials.
-
Checkboxes should be used appropriately to indicate activities conducted.
-
Avoid common mistakes such as missing signatures or incorrect dates.
Filling each field correctly is paramount to ensure a smooth authorization process.
Review and Validation Checklist for the Foreign Site Reference Number Authorization Form
Prior to submission, use the following checklist to validate the completion of your form:
-
Confirm all required fields are filled out correctly.
-
Check that signatures and dates are present.
-
Ensure all information complies with Health Canada requirements.
How to Sign the Foreign Site Reference Number Authorization Form
Signing the form correctly is essential. A senior official’s signature is a requirement for submission. You have options for signing: a digital signature or a wet signature. For an efficient process, pdfFiller can be utilized for eSigning the document.
Submission Methods and What Happens After You Submit the Form
Once your form is completed, you can submit it to Health Canada through designated methods. Be aware of possible fees and processing times. Ensure you track your submission for confirmation and expected feedback.
-
Available submission methods will be detailed on Health Canada’s website.
-
Processing times may vary depending on the form's completeness.
Security and Compliance in Handling the Foreign Site Reference Number Authorization Form
Data protection is imperative when handling healthcare forms. Utilizing pdfFiller ensures your sensitive information is safeguarded through robust security measures. The platform complies with both HIPAA and GDPR regulations, guaranteeing the privacy of medical documents.
Utilizing pdfFiller for the Foreign Site Reference Number Authorization Form
pdfFiller offers a comprehensive suite of tools for efficiently filling out, editing, and signing your forms. Experience the convenience and speed of managing healthcare forms with this cloud-based PDF editor. Start streamlining your authorization process with pdfFiller today.
How to fill out the Canada Foreign Site Reference Number Authorization
-
1.Access the Foreign Site Reference Number Authorization Form by visiting pdfFiller's website and searching for the form name in the search bar.
-
2.Once located, click on the form to open it in pdfFiller’s editing interface.
-
3.Begin by filling in the foreign site's details, including registration information and the specific activities conducted.
-
4.Next, provide the contact information for the senior official and quality assurance personnel, making sure the information is accurate and current.
-
5.Utilize the fillable fields efficiently—click on each field to enter data. Use checkboxes where applicable for options provided in the form.
-
6.Before proceeding, gather all necessary information, including previous correspondence, licenses, and other relevant documentation to ensure accuracy in completion.
-
7.Once all fields have been completed, thoroughly review the document for any errors or omissions to ensure compliance.
-
8.When satisfied with your entries, utilize pdfFiller's tools to save the document, or use the download option to obtain a PDF version.
-
9.If required, you can submit the form directly through the platform if the submission feature is enabled, or print it for mailing.

Who is eligible to use the Foreign Site Reference Number Authorization Form?
This form is designed for foreign sites registered with Health Canada and their Canadian importers needing access to GMP-related information. Eligibility includes senior officials and quality assurance personnel from both foreign sites and Canadian importers.
What information is required to complete this form?
To complete the form, you'll need details about the foreign site, activities being conducted, and contact information for senior officials and quality assurance personnel. Ensure that you have accurate and up-to-date information before starting the form.
How do I submit the completed Foreign Site Reference Number Authorization Form?
After completing the form, you can submit it through pdfFiller's platform if this option is available. Alternatively, you can save the form and print it to mail it directly to the required authority.
Are notarization or additional supporting documents needed for this form?
No notarization is required for this form. However, it may be beneficial to attach any relevant documents that support the authorization, such as previous communications and regulatory approvals.
What common mistakes should I avoid when filling out this form?
Avoid common mistakes such as leaving required fields blank, providing inaccurate contact information, or failing to obtain the necessary signatures from senior officials before submission.
Is there a deadline for submitting the Foreign Site Reference Number Authorization Form?
Deadlines may vary based on specific regulatory requirements or internal company policies. It's advisable to submit the form as soon as possible to prevent any delays in authorization.
How long does it take to process the Foreign Site Reference Number Authorization requests?
Processing times can vary depending on the complexity of the request and the regulatory authority involved. It is recommended to inquire directly for current processing times specific to your form submission.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.





















