Last updated on Apr 15, 2015

Get the free Gel Documentation Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Gel Form
The Gel Documentation Form is a medical records release document used by healthcare professionals to record and track gel electrophoresis results.
pdfFiller scores top ratings on review platforms




Who needs Gel Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Gel Form
What is the Gel Documentation Form?
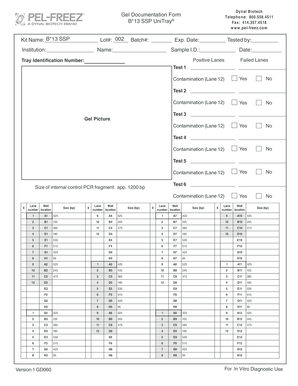
The Gel Documentation Form is an essential tool in laboratory environments, serving to accurately record results from gel electrophoresis procedures. Its primary purpose lies in ensuring that data from these tests is documented correctly to facilitate both immediate results review and long-term analysis.
This form is widely utilized in medical research settings, including for in vitro diagnostic applications, where tracking the specifics of gel electrophoresis results is critical. By maintaining accurate records of these experiments, practitioners can enhance the reliability of their findings.
Purpose and Benefits of the Gel Documentation Form
Utilizing the Gel Documentation Form offers numerous advantages within lab settings. Accurate documentation is crucial not only for validating results but also for maintaining integrity throughout the testing process.
-
Helps track vital information regarding kit details and sample identification.
-
Assists in ensuring compliance with regulatory standards.
-
Enhances record-keeping for future reference, making data easily retrievable.
Overall, employing this form contributes significantly to the organization and analysis of experimental data.
Key Features of the Gel Documentation Form
The Gel Documentation Form is structured to ensure ease of use, featuring several critical fields necessary for comprehensive documentation of gel electrophoresis tests. Key fields include Kit Name, Lot Number, Batch Number, Expiration Date, and Tested By.
In addition to these essential fields, the form includes sections for recording positive lanes, failed lanes, and contamination checks, thereby providing a robust framework for documenting the testing process.
Who Needs the Gel Documentation Form?
Various professionals within the medical and research fields rely on the Gel Documentation Form for their work. Laboratory technicians play a key role in filling out this form, alongside researchers and healthcare providers conducting gel electrophoresis tests.
Institutions engaged in detailed molecular biology work, such as clinical laboratories and research facilities, find this form indispensable for tracking and documenting their gel electrophoresis activities.
How to Fill Out the Gel Documentation Form Online (Step-by-Step)
Filling out the Gel Documentation Form using pdfFiller is straightforward. Start by accessing the form online, and ensure that you have created a pdfFiller account to facilitate your editing process.
-
Log in to your pdfFiller account.
-
Access the Gel Documentation Form from the template library.
-
Fill in essential information such as Kit Name, Sample ID, and Tested By.
-
Review each entry for accuracy before submitting the form.
Submission Methods and Delivery of the Gel Documentation Form
Once the Gel Documentation Form is completed, several submission options are available for sending it to relevant parties. Users can choose to submit the form online, print it for mailing, or email it directly depending on their institutional requirements.
It is crucial to follow guidelines pertaining to where and how to submit the completed form, which may vary based on institutional protocols and the nature of the documentation.
Common Errors and How to Avoid Them When Using the Gel Documentation Form
Awareness of potential errors when completing the Gel Documentation Form can save time and resources. Common mistakes include mislabeling samples or leaving fields incomplete, which undermine the integrity of test results.
To prevent these errors, ensure that you review all entries carefully. Utilizing resources available on pdfFiller for additional support can further enhance your accuracy.
Security and Compliance Considerations for the Gel Documentation Form
Protecting sensitive information is paramount when handling the Gel Documentation Form. pdfFiller implements security measures such as 256-bit encryption and complies with HIPAA and GDPR regulations to secure user data.
Healthcare professionals must adopt best practices for managing medical records, ensuring that all documentation is submitted and stored securely to maintain patient confidentiality.
Experience the Ease of Completing the Gel Documentation Form with pdfFiller
Completing the Gel Documentation Form using pdfFiller simplifies the documentation process, offering features such as fillable forms and e-signing capabilities. Users report significant time savings and improved organization in their documentation practices.
By utilizing pdfFiller, users can streamline their workflow while ensuring that they adhere to stringent security protocols, thus enhancing overall efficiency in managing their gel electrophoresis documentation.
How to fill out the Gel Form
-
1.To access the Gel Documentation Form on pdfFiller, visit the pdfFiller website and log into your account.
-
2.In the search bar, enter the form name 'Gel Documentation Form' and click to open it.
-
3.Familiarize yourself with the structure of the form, including sections for kit information, sample identification, lanes for positive and failed tests, and contamination checks.
-
4.Gather all necessary information before filling out the form. This should include the Kit Name, Lot #, Batch#, Exp. Date, name of the technician, the institution, Sample I.D., and the date of testing.
-
5.Start filling in the Kit Name, Lot #, and Batch# in the respective fields. Follow through by entering the Exp. Date and the name of the technician responsible for the tests.
-
6.Next, provide specific details in the Sample I.D. field and indicate the date of testing.
-
7.Complete the fields for well locations and sizes as required for your test results, ensuring all sections are accurately filled out to prevent errors.
-
8.After filling in all the required fields, review the form for accuracy. Make sure all entered data is correct and all sections are complete.
-
9.Once confirmed, you can save or download the form in PDF format. Alternatively, you can submit directly through pdfFiller if required.

Who is eligible to use the Gel Documentation Form?
The Gel Documentation Form can be used by laboratory technicians, medical researchers, and healthcare providers conducting gel electrophoresis and in vitro diagnostics. It is essential for those who need to document test results accurately.
What supporting documents are required to fill out this form?
Typically, no additional supporting documents are necessary to complete the Gel Documentation Form. However, having the kit information, patient sample identification, and results data handy will ensure accuracy during completion.
How should I submit the Gel Documentation Form once completed?
After completing the Gel Documentation Form, you can usually submit it via your institution’s standard reporting procedures. If using pdfFiller, you can also use their submission features after saving your completed form.
What common mistakes should I avoid when filling out this form?
Common mistakes include omitting required fields, such as Kit Name or Sample I.D., and misspelling important information that could affect test results. Always double-check your entries before submission.
How long will it take to process the information from this form?
Processing times can vary by institution, but generally, it should take a few days for the data to be reviewed and processed. For time-sensitive results, check with your lab for specific timelines.
Can the Gel Documentation Form be filled out electronically?
Yes, the Gel Documentation Form can be filled out electronically using pdfFiller. This allows for easy editing, saving, and sharing of completed forms.
Is notarization required for the Gel Documentation Form?
No, notarization is not required for the Gel Documentation Form. It is primarily an internal document for recording test results.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.