Last updated on Apr 3, 2026
Get the free CDC Form 0.753 Continuation Page
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is cdc form 0753 continuation
The CDC Form 0.753 Continuation Page is a government form used by individuals and organizations to apply for permission to import infectious biological agents into the United States.
pdfFiller scores top ratings on review platforms




Who needs cdc form 0753 continuation?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to cdc form 0753 continuation
What is the CDC Form 0.753 Continuation Page?
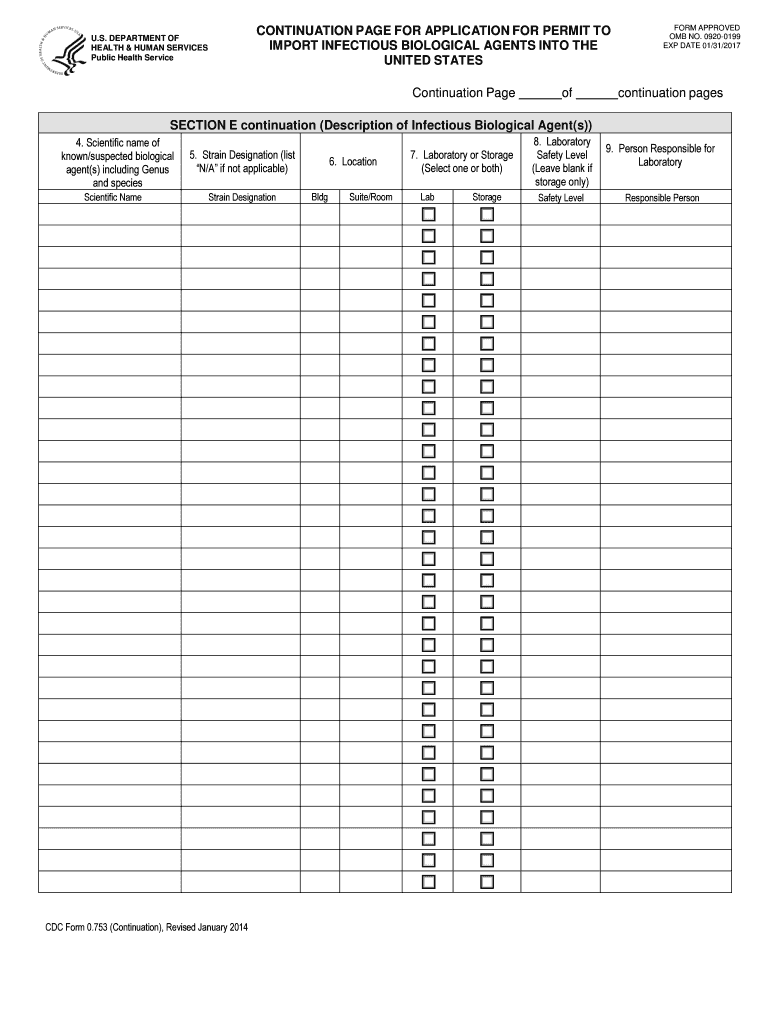
The CDC Form 0.753 Continuation Page is a critical document in the application process for importing infectious biological agents into the United States. This form enables applicants to provide detailed information about the agents they intend to import, facilitating compliance with regulatory requirements. It is important to note that this form has an expiration date of January 31, 2017, indicating that it may no longer be valid for current applications.
-
The form plays a significant role in the importation procedure of infectious biological agents.
-
It serves as a continuation page to include exhaustive descriptions of the agents.
-
Due to its expiration, applicants should check for updated forms relevant to their needs.
Purpose and Benefits of the CDC Form 0.753 Continuation Page
Understanding the purpose of the CDC Form 0.753 is essential for entities involved in importing biological agents. This form not only aids in maintaining compliance with CDC regulations but also offers several benefits that enhance the clarity of the application process.
-
It ensures that all necessary details about the biological agents are transparent and well-documented.
-
Having precise descriptions minimizes the risk of misunderstandings with regulatory authorities.
-
Non-compliance with CDC guidelines can lead to serious legal ramifications, making this form crucial.
Who Needs the CDC Form 0.753 Continuation Page?
The target audience for the CDC Form 0.753 includes a variety of entities engaged in research or commercial activities involving biological agents. Understanding who requires this form is vital for ensuring that the right parties comply with import regulations.
-
Research institutions handling infectious agents must complete this form.
-
Corporations importing biological samples for testing or development also fall under this requirement.
-
Both governmental and private sector entities should be aware of their responsibilities regarding this form.
How to Fill Out the CDC Form 0.753 Continuation Page Online (Step-by-Step)
Completing the CDC Form 0.753 online involves a few essential steps to ensure all information is accurately captured. A systematic approach will enhance the likelihood of a successful submission without delays.
-
Gather necessary information about the infectious biological agents, including scientific names and strain designations.
-
Access the form on the designated platform and enter the required information carefully.
-
Double-check all entries for accuracy before finalizing the form.
Accuracy is paramount when reporting scientific designs as inaccuracies could lead to processing issues.
Field-by-Field Instructions for the CDC Form 0.753 Continuation Page
Breaking down the fields in the CDC Form 0.753 can help applicants understand what is required, thus minimizing errors during completion.
-
Key fields include scientific names, which must be accurately reported.
-
Strain designations are critical for identifying specific biological entities.
-
Laboratory details must be filled out with precise information regarding handling and storage capabilities.
Be aware of potential pitfalls, especially in critical fields where incorrect entries can lead to form rejections.
Review and Validation Checklist for the CDC Form 0.753 Continuation Page
Before submitting the CDC Form 0.753, conducting a thorough review is essential to reduce the chances of delays or rejections.
-
Check the accuracy of all information provided in the form.
-
Ensure all required signatures are present before submission.
-
Verify that all relevant fields are completed correctly to avoid potential complications.
Neglecting the validation checklist could result in significant setbacks during the application process.
How to Submit the CDC Form 0.753 Continuation Page
Upon completing the CDC Form 0.753, understanding the submission methods will facilitate a smooth application process.
-
Forms can typically be submitted online or via mail, depending on the guidelines provided.
-
Processing times may vary, so it's crucial to remain informed about current timelines.
-
Common reasons for submission rejections include missing information or inaccuracies, so careful review is necessary.
Security and Compliance When Filling Out the CDC Form 0.753 Continuation Page
Ensuring security during the process of filling out sensitive forms like the CDC Form 0.753 is paramount. Users should be aware of the various protections in place.
-
Data protection and privacy measures are implemented for all form submissions.
-
Compliance with regulations such as HIPAA and GDPR is maintained rigorously.
-
The platform utilized should have robust security features for handling forms and sensitive data.
Why Use pdfFiller for Completing Your CDC Form 0.753 Continuation Page
Opting for pdfFiller when completing the CDC Form 0.753 offers several advantages that simplify the document management process.
-
Key features of pdfFiller allow for easy editing and eSigning of documents.
-
The platform is recognized for its strong security credentials, ensuring compliance with necessary regulations.
-
Using pdfFiller enhances the efficiency and organization of form completion and storage.
Get Started with Your CDC Form 0.753 Continuation Page Today!
Starting your application with the CDC Form 0.753 through pdfFiller is a straightforward process, equipped with user-friendly tools designed for efficiency. Embracing this platform will streamline your document management tasks.
-
Begin by accessing the pdfFiller platform to start filling out the CDC Form 0.753.
-
Utilize the secure and efficient tools provided by pdfFiller to manage your documentation process seamlessly.
How to fill out the cdc form 0753 continuation
-
1.Access pdfFiller and search for 'CDC Form 0.753 Continuation Page' to locate the form.
-
2.Once open, review the form's sections to familiarize yourself with the required fields.
-
3.Collect all necessary information, including scientific names and strain designations, prior to filling in the form.
-
4.Utilize pdfFiller’s text fields to enter details for biological agents, ensuring accuracy and completeness.
-
5.If needed, use any provided guidelines or tooltips within pdfFiller to assist with specific sections.
-
6.After filling in all necessary information, take a moment to review the form to check for any missing details or errors.
-
7.Once satisfied with the completed form, navigate to the option to save or download your form.
-
8.You can submit the form later by uploading it to the relevant government portal or email as instructed.

Who is eligible to submit the CDC Form 0.753 Continuation Page?
Any individual or organization looking to legally import infectious biological agents into the U.S. may fill out and submit this form, provided they adhere to relevant regulations.
Is there a deadline for submitting the CDC Form 0.753?
While there is no specific deadline, it should be submitted well in advance of any planned import to allow for processing and approval by the CDC.
How do I submit the CDC Form 0.753 after filling it out?
Completed forms can be submitted via mail or electronically depending on CDC guidelines. Check the CDC's official site for the most current submission procedures.
What supporting documents are required with the form?
Typically, supporting documentation may include evidence of the laboratory's capability to handle the agents and any additional permits based on the nature of the biological agents.
What common mistakes should I avoid when filling out the form?
Ensure all fields are completed, double-check for accuracy in scientific names, and confirm that you are using the most recent version of the form to avoid delays.
What is the processing time for this form?
Processing times can vary based on the complexity of the application and the current workload of the CDC. It’s advisable to allow several weeks for review.
What specific concerns should I have regarding the CDC Form 0.753?
Ensure you are aware that this form is outdated with an expiration date of 01/31/2017 and check for any updated requirements or versions from the CDC’s official resources.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.





















