Last updated on May 21, 2015

Get the free Quality Control Inspector Job Description
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Quality Inspector Job
The Quality Control Inspector Job Description is a document used by employers in the medical device manufacturing industry to outline the responsibilities and qualifications required for the role of a Quality Control Inspector.
pdfFiller scores top ratings on review platforms




Who needs Quality Inspector Job?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Quality Inspector Job
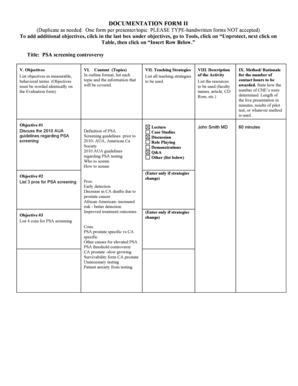
What is the Quality Control Inspector Job Description?
The Quality Control Inspector plays a crucial role in ensuring the standards within the medical device manufacturing industry. This position entails key responsibilities such as conducting inspections, performing audits, and maintaining accurate documentation. The job description serves as a vital tool in the overall quality assurance process, clearly outlining the expectations for both employers and employees.
Purpose and Benefits of the Quality Control Inspector Job Description
The Quality Control Inspector job description is essential for establishing clear expectations within the workplace. Its advantages include:
-
Defining the role to ensure alignment with company standards.
-
Helping to maintain compliance with industry regulations.
-
Streamlining the hiring process and enhancing training programs.
Key Features of the Quality Control Inspector Job Description
This document contains several essential components that contribute to its effectiveness:
-
A comprehensive list of typical duties, including inspections, audits, and documentation.
-
Specific skills and qualifications necessary for the role.
-
Signature and date fields, which indicate acknowledgment and compliance.
Who Needs the Quality Control Inspector Job Description?
The target audience for this document includes various stakeholders within the industry:
-
Employers operating in the medical device and manufacturing sectors.
-
HR departments and hiring managers responsible for recruitment.
-
New employees being onboarded into quality assurance roles.
How to Fill Out the Quality Control Inspector Job Description Online
Completing the Quality Control Inspector job description form can be done effectively by following these steps:
-
Access the form on pdfFiller.
-
Fill in the required fields with accurate information.
-
Review for completeness to avoid common errors.
-
Submit the completed form securely.
Digital Signature Requirements for the Quality Control Inspector Job Description
Signature requirements for this document are important for validation. Key points include:
-
Understanding the differences between digital and wet signatures.
-
Utilizing pdfFiller to securely eSign the job description.
-
Recognizing the importance of signatures for compliance.
Common Errors and How to Avoid Them
Users should be aware of frequent mistakes when completing the form. Common errors include:
-
Inaccurate information entry.
-
Omitting required fields.
-
Failure to review before submission, which can lead to processing delays.
How pdfFiller Can Help with the Quality Control Inspector Job Description
pdfFiller offers various features to streamline the completion of the Quality Control Inspector job description:
-
Edit text and images for precise documentation.
-
eSigning capabilities simplify the signature process.
-
User-friendly interface enhances document management.
Where to Submit the Quality Control Inspector Job Description
Guidance on submission methods should include:
-
Recommended submission methods, such as online options or in-person deliveries.
-
Relevant addresses and platforms for submission.
-
Awareness of any specific deadlines or processing times.
Maximizing Your Efficiency with the Quality Control Inspector Job Description
Engaging with pdfFiller can significantly enhance your efficiency in managing forms:
-
Easily access templates and tools tailored for form adjustments.
-
Experience the convenience of digital document management.
-
Realize the benefits of quick and straightforward form processing.
How to fill out the Quality Inspector Job
-
1.Begin by accessing the pdfFiller website and logging in or creating an account if you do not have one. Use the search bar to find the Quality Control Inspector Job Description form.
-
2.Once you have located the form, click on it to open. Familiarize yourself with the pdfFiller interface, noting the toolbar options available for editing.
-
3.Before filling out the form, gather all necessary information about the job requirements, responsibilities, and qualifications necessary for the Quality Control Inspector position.
-
4.Start completing the form by clicking on each field and entering the relevant information. Be thorough in detailing the expectations and duties associated with the role of the Quality Control Inspector.
-
5.After you have filled out all fields, review the completed document for any errors or missing information. Double-check the signature and date fields since the document requires the employee to sign.
-
6.Once confirmed and accurate, finalize your document by selecting the 'Save' option in the toolbar. You may also choose to download it as a PDF for offline access.
-
7.Finally, submit the completed form through your company’s designated submission protocol, or save it for internal records using pdfFiller’s cloud storage feature.

What are the eligibility requirements for filling out this form?
To fill out the Quality Control Inspector Job Description form, you should either be an HR manager or employee responsible for hiring in the medical device industry, ensuring you understand the job role.
Is there a deadline for submitting this job description form?
Typically, submission deadlines depend on the hiring schedule of your organization. Ensure you check with your HR department for any specific timelines related to job postings.
How should I submit the completed Quality Control Inspector Job Description form?
The form can be submitted through your company's HR portal or by emailing it to the relevant hiring manager. Check with your organization for precise submission methods.
What supporting documents are required along with this form?
Generally, no additional supporting documents are needed for the job description form itself. However, it might be beneficial to provide related organizational information or qualifications to aid in comprehension.
What common mistakes should I avoid while filling out the form?
Avoid overlooking the signature and date fields, as these are mandatory. Also, ensure all job responsibilities are clearly written to avoid any vague descriptions that could cause confusion.
How long does it take for the form to be processed?
Processing time can vary based on your organization's internal procedures. It might take anywhere from a few days to a couple of weeks for HR to review the document.
Can this job description be modified after submission?
Yes, job descriptions can be modified as necessary. If changes are required, consult with HR and ensure the revised description remains accurate and up to date.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.