Last updated on Apr 10, 2026
Get the free Laboratory Animal Clinical Record Sheet
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is laboratory animal clinical record
The Laboratory Animal Clinical Record Sheet is a medical record used by researchers to monitor and assess the health and welfare of sheep in research settings.
pdfFiller scores top ratings on review platforms




Who needs laboratory animal clinical record?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to laboratory animal clinical record
What is the Laboratory Animal Clinical Record Sheet?
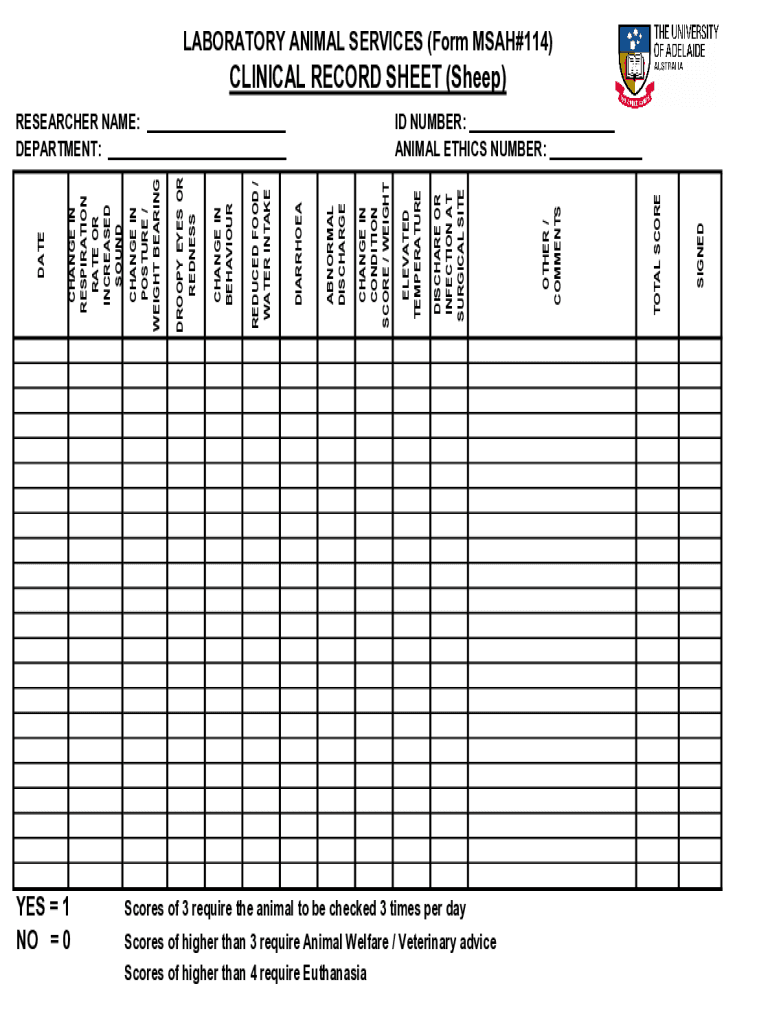
The Laboratory Animal Clinical Record Sheet (Form MSAH#114) is a crucial tool for monitoring the health of sheep in research environments. This form emphasizes the significance of systematic health observation in animal welfare, particularly in research settings. It provides a comprehensive overview of daily health observations required for each subject, along with effective scoring methods to assess the overall condition of the animals.
Utilizing this record sheet enables researchers to maintain accurate documentation, which is essential for ethical compliance and effective veterinary interventions.
Purpose and Benefits of Using the Laboratory Animal Clinical Record Sheet
This record sheet enhances animal welfare by facilitating regular health monitoring. By documenting health parameters, researchers can ensure compliance with ethical standards required in animal research. The form's structured approach aids significantly in identifying health issues early, allowing prompt veterinary intervention.
Furthermore, the consistent use of the animal welfare scoring sheet helps researchers maintain comprehensive records necessary for audits and evaluations.
Key Features of the Laboratory Animal Clinical Record Sheet
The Laboratory Animal Clinical Record Sheet includes several impactful features designed for efficient health monitoring. Key components of the form are:
-
Blank fields for daily health observations.
-
Checkboxes for scoring various health parameters.
-
A signature line for researcher approval.
These features are integral to ensuring that the research meets the highest standards of animal care.
Who Needs the Laboratory Animal Clinical Record Sheet?
The primary users of this essential form are researchers working within laboratories that involve animal subjects, particularly sheep. It is required in various scenarios, such as health assessments and monitoring during experimental procedures. Researchers play a crucial role in utilizing this form by documenting observations and providing their signatures to confirm the accuracy of the reported data.
How to Fill Out the Laboratory Animal Clinical Record Sheet Online
Filling out the Laboratory Animal Clinical Record Sheet online is straightforward if you follow these steps:
-
Gather all necessary information regarding the animals being monitored.
-
Access the form on a compatible device.
-
Fill out each field diligently, ensuring that scoring is accurate.
-
Review the completed form for completeness before submission.
Accuracy in data entry is critical for reflecting the true health status of the sheep.
How to Sign and Submit the Laboratory Animal Clinical Record Sheet
Understanding the signing and submission requirements is key to effective process management. Researchers can opt for either a digital or a wet signature to validate their documentation. Submission methods should conform to the established guidelines specific to researchers in the U.S., and it's vital to confirm receipt of the form after submission for tracking purposes.
Security and Compliance for the Laboratory Animal Clinical Record Sheet
Data privacy and security are major considerations when handling the Laboratory Animal Clinical Record Sheet. This form employs 256-bit encryption to protect sensitive information and complies with HIPAA and GDPR regulations. Maintaining confidentiality is crucial for the protection of health information, and researchers must adhere to best practices for securely managing submitted forms.
Sample of a Completed Laboratory Animal Clinical Record Sheet
A practical reference for users is the sample of a filled-out Laboratory Animal Clinical Record Sheet. This example highlights key sections and demonstrates the expected level of detail in entries. Accurate documentation not only aids in compliance but also enhances overall research outcomes by ensuring that all health parameters are properly addressed.
How pdfFiller Can Assist with Your Laboratory Animal Clinical Record Sheet
pdfFiller provides valuable assistance for users handling the Laboratory Animal Clinical Record Sheet. This platform enables seamless form editing, eSigning, and online filling without the need for downloads. Users can benefit from comprehensive support and resources available through pdfFiller, making the process of managing this essential form easier and more efficient.
How to fill out the laboratory animal clinical record
-
1.To access the Laboratory Animal Clinical Record Sheet on pdfFiller, visit the pdfFiller website and use the search bar to find the form by its name or number.
-
2.Once the form appears, click on it to open. Familiarize yourself with the layout, which includes blank fields for data entry and checkboxes.
-
3.Before starting, gather all necessary information, such as the animal's identification, daily health observation data, and scoring parameters.
-
4.Begin filling out the form by entering the sheep's identification and any observed health indicators into the designated fields, making sure to check all relevant conditions.
-
5.Use the scoring tables provided in the form to assess the sheep's overall condition based on the daily observations. Ensure each checkmark corresponds with accurate health details.
-
6.When you've completed the scoring tables and observation inputs, review all filled sections for accuracy and clarity. Confirm that all fields are appropriately filled.
-
7.Once confirmed, sign the form in the designated area to validate that the information is correct and complete.
-
8.Finally, save your work by clicking the save button. You can also download the completed form as a PDF or submit it directly through pdfFiller, depending on how you wish to handle it.

Who is eligible to complete the Laboratory Animal Clinical Record Sheet?
The form can be completed by researchers involved in animal health studies, veterinarians managing animal welfare, and other authorized personnel working in laboratory settings.
Are there any specific deadlines for submitting this form?
While there may not be universal deadlines, researchers should complete and submit the form promptly to ensure timely veterinary assessments and interventions.
What methods are available for submitting the completed form?
The completed Laboratory Animal Clinical Record Sheet can be submitted electronically through pdfFiller or downloaded and physically submitted depending on your institution's policies.
What supporting documents are needed with this form?
Typically, no additional documents are required. However, researchers should have daily observation records on hand to complete the form accurately.
What common mistakes should be avoided when filling the form?
Common mistakes include omitting crucial data, failing to sign the form, or inaccurately scoring the animal's condition. Double-check all entries.
How long does it take for the form to be processed after submission?
Processing times may vary by institution. However, researchers can expect feedback or required actions within a reasonable timeframe especially in urgent situations.
Can the form be modified after initial submission?
Yes, if changes are necessary after submission, a revised form can be filled out and submitted. Always document the reasons for the revision.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.