Last updated on Feb 14, 2015

Get the free Confidential Financial Disclosure Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Financial Disclosure Form
The Confidential Financial Disclosure Form is a financial disclosure document used by clinical investigators to reveal financial interests related to clinical studies.
pdfFiller scores top ratings on review platforms




Who needs Financial Disclosure Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Financial Disclosure Form
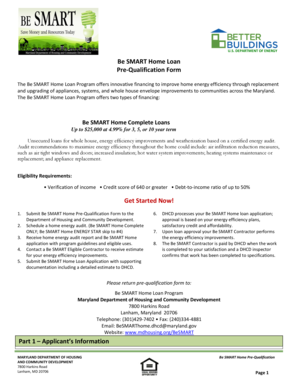
What is the Confidential Financial Disclosure Form?
The Confidential Financial Disclosure Form is a critical document used by clinical investigators to disclose any financial interests related to clinical research. This form serves to ensure transparency and integrity in clinical studies, as it requires investigators to report financial arrangements with pharmaceutical companies that may influence study outcomes. Accurately completing this financial disclosure form is essential for maintaining the credibility of the research process.
Disclosing financial interests is vital because it helps to prevent conflicts of interest that could compromise the integrity of clinical studies. The form plays a key role in upholding ethical standards within the clinical research community.
Why is the Confidential Financial Disclosure Form Important?
The necessity of the Confidential Financial Disclosure Form cannot be overstated, as it is integral to maintaining transparency in clinical research. Undisclosed financial interests can have detrimental effects on study integrity, potentially leading to biased outcomes and undermining public trust in the research process.
Regulatory bodies, including the FDA, mandate financial disclosures from clinical investigators to ensure accountability and adherence to ethical guidelines. Failure to comply with these regulations can result in significant consequences for both the investigator and the study itself.
Who Needs to Complete the Confidential Financial Disclosure Form?
The Confidential Financial Disclosure Form is primarily required for individuals involved in clinical investigation, including clinical investigators and key study personnel. These individuals must submit the form annually to disclose their financial interests and relationships that could influence their research activities.
By completing this form, clinical investigators foster a culture of transparency and accountability within the research environment, which is crucial for ethical clinical practice.
How to Fill Out the Confidential Financial Disclosure Form Online
Completing the Confidential Financial Disclosure Form online is a straightforward process. Follow these steps to ensure your submission is accurate and compliant:
-
Access the online form and review the instructions carefully.
-
Fill in each required field, ensuring that all information is complete and correct.
-
Double-check for any missing details or potential errors in your entries.
-
Submit the form electronically according to the specified guidelines.
For best results, familiarize yourself with tips for accuracy and compliance to avoid complications during submission.
Common Mistakes to Avoid When Filing the Confidential Financial Disclosure Form
When completing the Confidential Financial Disclosure Form, it is crucial to avoid several common mistakes that can lead to complications or rejection of your submission:
-
Omitting essential financial interests that may raise conflicts.
-
Errors in personal information, such as incorrect contact details.
-
Failing to sign the form electronically before submission.
To help mitigate these issues, utilize a validation checklist that guides you through a cross-check of your entries prior to submission.
How to eSign the Confidential Financial Disclosure Form
eSigning the Confidential Financial Disclosure Form is an important step in the submission process. Understanding the differences between digital and wet signatures can help ensure compliance:
-
A digital signature is an electronic form of a signature that is legally recognized.
-
A wet signature is a handwritten signature on a physical document.
Follow the specific instructions provided for electronically signing the form to ensure your submission is valid and accepted by regulatory authorities.
Where to Submit the Confidential Financial Disclosure Form
After completing the Confidential Financial Disclosure Form, it is essential to know the correct submission methods:
-
Online: Submit through the designated platform for immediate processing.
-
Mail: Send the completed form via postal service if required.
Be aware of important deadlines and processing times to ensure timely submission and avoid delays in your research activities.
Data Security and Compliance When Using the Confidential Financial Disclosure Form
Ensuring data security is paramount when filling out the Confidential Financial Disclosure Form. pdfFiller prioritizes the protection of sensitive information with robust security measures:
-
256-bit encryption safeguards your data during transmission.
-
Compliance with regulations such as HIPAA and GDPR further protects user information.
These measures help provide peace of mind while handling confidential financial details associated with clinical studies.
Utilizing pdfFiller for Your Confidential Financial Disclosure Form Needs
pdfFiller is an invaluable resource for managing your Confidential Financial Disclosure Form. The platform offers various capabilities that enhance the form-filling process:
-
User-friendly interface allows for seamless editing and submission.
-
Features such as eSignature integration simplify the signing process.
-
Document storage options keep all your submissions organized and easily accessible.
Leverage pdfFiller to streamline your workflow and maintain compliance with financial disclosure requirements.
Next Steps After Submitting the Confidential Financial Disclosure Form
Once you have submitted the Confidential Financial Disclosure Form, there are important next steps to consider:
-
Track the submission status through the specified platform to ensure approval.
-
If necessary, follow the procedures for making corrections or amendments to your submission.
Staying informed about your submission status can help you address any potential issues promptly.
How to fill out the Financial Disclosure Form
-
1.Access pdfFiller and search for the Confidential Financial Disclosure Form to begin.
-
2.Once you've located the form, click to open it in the pdfFiller interface.
-
3.Before filling out the form, gather necessary information about any financial interests or arrangements with pharmaceutical companies relevant to your studies.
-
4.Use the document's checkboxes to indicate the appropriate disclosures and fill in any blank fields with accurate information.
-
5.Review each section carefully to ensure all details are complete and correct before finalizing.
-
6.Utilize pdfFiller’s editing tools to make adjustments and clearly assess your disclosures.
-
7.Once satisfied with your entries, save your progress and download the form if required.
-
8.Finally, submit the completed form according to your institution's submission guidelines or keep a copy for your records.

Who is eligible to use the Confidential Financial Disclosure Form?
The Confidential Financial Disclosure Form is necessary for clinical investigators who are engaged in research involving financial interests, particularly those working on studies that involve pharmaceutical companies.
What are the deadlines for submitting the form?
The form must be submitted annually as part of the investigator's registration packet. Ensure you check specific submission deadlines set by your institution or regulatory body.
How should I submit the completed form?
After filling out the form, you can submit it electronically through the required channels of your sponsoring organization or print it out for physical submission, depending on their instructions.
What supporting documents are required with the form?
Typically, additional documentation is not required for this form. However, you may need to provide supplementary evidence of your financial interests if specifically requested by your institution or sponsor.
What common mistakes should I avoid when completing the form?
Ensure all financial interests are accurately reported, especially any relationships with pharmaceutical companies. Avoid leaving fields blank and double-check your information for errors before submission.
How long does it take to process the form after submission?
Processing time can vary based on the institution or regulatory body. Generally, allow several weeks for review, but check with your specific organization for exact timelines.
Are there fees associated with this form?
There are typically no fees directly associated with completing the Confidential Financial Disclosure Form. However, fees may apply for the submission process based on your institution's policies.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.