Last updated on Mar 10, 2015

Get the free AEC-483 Registration Certificate for In Vitro Testing
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is AEC-483 Form
The AEC-483 Registration Certificate for In Vitro Testing is a government form used by physicians, clinical laboratories, and hospitals in the United States to apply for a registration number to use byproduct materials for in vitro testing.
pdfFiller scores top ratings on review platforms




Who needs AEC-483 Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to AEC-483 Form
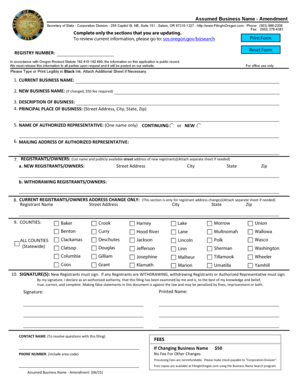
What is the AEC-483 Registration Certificate for In Vitro Testing?
The AEC-483 form serves as a registration certificate for entities involved in in vitro testing, specifically designed for physicians, clinical laboratories, and hospitals. Its primary purpose is to ensure compliance with regulatory standards when using byproduct materials for clinical testing. Obtaining this registration is vital as it establishes legitimacy in clinical testing and addresses the safety of byproduct materials.
This form plays a key role in the overall clinical testing landscape, facilitating proper oversight and regulation by the U.S. Atomic Energy Commission. It confirms that practitioners are authorized to conduct specific testing procedures using designated materials, ultimately supporting patient safety and health standards.
Purpose and Benefits of the AEC-483 Registration Certificate for In Vitro Testing
The registration certificate in vitro testing serves several crucial functions that enhance safety and regulatory compliance. It assures that clinical laboratories and hospitals adhere to strict standards necessary for performing diagnostic tests, thereby protecting patient health. By obtaining this certification, stakeholders benefit from increased trust and credibility in their operations.
Physicians and hospitals can also enjoy improved operational efficiency and clearer regulatory pathways when they possess the clinical laboratory registration required by law. This streamlined process not only supports better patient outcomes but also helps in mitigating legal risks associated with unauthorized testing practices.
Who Needs the AEC-483 Registration Certificate for In Vitro Testing?
Primary users of the AEC-483 form include physicians, clinical laboratories, and hospitals that intend to utilize byproduct materials for in vitro tests. Entities operating without this registration may face legal consequences, making it necessary for them to obtain this certificate to ensure lawful operation.
Particularly, any clinical laboratory registration is imperative for those performing diagnostic tests, while hospitals are required to secure the hospital registration form to maintain compliance with healthcare regulations.
Eligibility Criteria for the AEC-483 Registration Certificate for In Vitro Testing
To be eligible for the AEC-483 certificate, applicants must meet specific criteria, including having the necessary certifications and possessing radiation measuring instruments. This ensures that all applicants are qualified to handle byproduct materials safely and according to industry standards.
These eligibility requirements are designed to ensure that only entities equipped with the proper knowledge and equipment engage in in vitro testing, thereby promoting adherence to health and safety regulations.
How to Fill Out the AEC-483 Registration Certificate for In Vitro Testing Online (Step-by-Step)
Filling out the AEC-483 Registration Certificate for In Vitro Testing online can be straightforward if you follow these steps:
-
Access the online registration portal for the AEC-483 form.
-
Complete all required fields, including name, address, and contact information.
-
Provide details about your radiation measuring instruments and ensure they adhere to required specifications.
-
Review the form for accuracy and completeness before submission.
-
Submit the form electronically and note any confirmation numbers provided.
It is essential to avoid common pitfalls such as leaving blank fields or providing incorrect information, as these may delay the registration process.
Required Documents and Supporting Materials for the AEC-483 Registration Certificate
To submit the AEC-483 form, applicants must prepare certain documents and materials. Required items include:
-
A completed AEC-483 form.
-
Proof of eligibility, such as certification documents.
-
Details outlining the radiation measuring instruments in use.
-
Any additional documents specified by the U.S. Atomic Energy Commission.
It is important to note that the specific filing requirements may vary depending on the type of applicant, necessitating careful attention to documentation to avoid any processing delays.
Submission Methods for the AEC-483 Registration Certificate
There are various methods available for submitting the AEC-483 form to the U.S. Atomic Energy Commission. These methods include:
-
Electronic submission via the official online portal.
-
Paper submission via mail to the designated office.
When submitting, it's crucial to adhere to specified timeframes and to track submissions effectively. Avoiding delays can be achieved by ensuring all documents are complete and submitted correctly, as well as monitoring submission confirmation communications.
Security and Compliance for the AEC-483 Registration Certificate for In Vitro Testing
Security and compliance during the handling of sensitive documents, such as the AEC-483 Registration Certificate, is paramount. Safeguarding confidentiality helps to protect patient information and maintain institutional integrity.
pdfFiller employs 256-bit encryption and adheres to HIPAA and GDPR regulations, ensuring document security throughout the registration process. This commitment to security enables users to manage sensitive information with confidence.
How pdfFiller Can Help with the AEC-483 Registration Certificate
pdfFiller offers a range of features that assist users in efficiently completing the AEC-483 form. Tools available on the platform include options for editing text and images, creating fillable forms, and utilizing eSigning capabilities.
These resources simplify the process of filling out the registration form, enabling users to submit their applications quickly and accurately while maintaining the integrity of their documentation.
Next Steps After Submitting the AEC-483 Registration Certificate
Once the AEC-483 form submission is complete, applicants can expect to receive updates regarding their application status. Initial tracking may involve monitoring confirmation communications and responses from the U.S. Atomic Energy Commission.
If necessary, users should be prepared to correct or amend their applications. Familiarity with the correction process will facilitate timely adjustments and help ensure that the registration is finalized without complications.
How to fill out the AEC-483 Form
-
1.To begin, visit pdfFiller and log into your account or create a new one if you don’t have an account.
-
2.Search for 'AEC-483 Registration Certificate for In Vitro Testing' in the pdfFiller search bar.
-
3.Once you find the form, click on it to open it in the pdfFiller editor.
-
4.Carefully read through the form to familiarize yourself with the required fields.
-
5.Before filling out the form, gather necessary information including your name, address, and details about your radiation measuring instruments.
-
6.Use the text boxes in pdfFiller to input your information accurately in each designated field.
-
7.Ensure that you complete all required fields and check the appropriate boxes for type of registrant.
-
8.Review the form thoroughly using pdfFiller’s preview feature. Ensure all information is accurate and complete.
-
9.Once you are satisfied with your entries, save the form to your pdfFiller account.
-
10.Choose the option to download the form or submit it directly through pdfFiller to the U.S. Atomic Energy Commission in triplicate.

Who is eligible to complete the AEC-483 form?
The AEC-483 form is intended for physicians, clinical laboratories, and hospitals intending to use byproduct materials for in vitro testing. All applicants must certify that they are equipped with the necessary radiation measuring instruments for compliance.
What is the submission method for the AEC-483 form?
The AEC-483 form must be submitted in triplicate to the U.S. Atomic Energy Commission. You can either download the completed form from pdfFiller and mail it or, if allowed, submit electronically.
Are there any deadlines for submitting the AEC-483 Registration Certificate?
While specific deadlines may vary, it is advised to submit your AEC-483 form as soon as you have completed it to avoid any delays in receiving your registration. Check the U.S. Atomic Energy Commission’s guidelines for any time-sensitive information.
What supporting documents are required with the AEC-483 form?
Typically, the AEC-483 form does not require additional documentation, but registrants should ensure their radiation measuring instruments are compliant. It's best to check the U.S. Atomic Energy Commission’s instructions for any updates.
What common mistakes should I avoid when filling out the AEC-483 form?
Common mistakes include leaving required fields blank, failing to check the appropriate registrant type boxes, or filling in incorrect information. Double-checking entries before submission can prevent rejections.
How long does it take to process the AEC-483 application?
Processing times can vary. Typically, applications submitted to the U.S. Atomic Energy Commission may take several weeks. Contact them directly for more precise timelines.
Do I need to notarize the AEC-483 form?
No, the AEC-483 form does not require notarization, making it simpler for registrants. Ensure all information is accurately provided instead.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.