Last updated on Mar 21, 2015

Get the free CDC Q Fever Case Report
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Q Fever Report
The CDC Q Fever Case Report is a healthcare form used by physicians and health departments to document and report cases of Q Fever, a bacterial infection.
pdfFiller scores top ratings on review platforms




Who needs Q Fever Report?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Q Fever Report
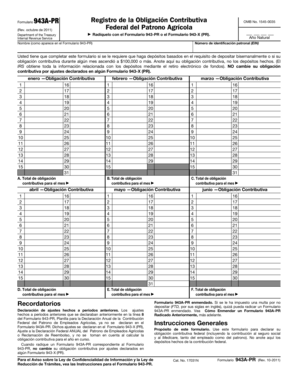
What is the CDC Q Fever Case Report?
The CDC Q Fever Case Report is a significant healthcare form used to document and report cases of Q Fever, a bacterial infection with considerable implications for public health. This form serves as a crucial tool for medical professionals and health departments to record essential patient and physician information. Additionally, it underscores the need for thorough documentation of Q Fever cases to facilitate disease monitoring and management.
-
Q Fever, caused by the bacterium Coxiella burnetii, can lead to severe health complications.
-
The CDC Q Fever Case Report compiles critical details necessary for effective public health response.
Purpose and Benefits of the CDC Q Fever Case Report
This form is vital for physicians and health departments as it aids in tracking and managing Q Fever cases in the community. Monitoring Q Fever is essential not only for individual patient outcomes but also for the overall public health strategy. The CDC Q Fever Case Report enhances patient care and supports better data collection, thereby improving responses to communicable diseases.
-
Enables effective tracking of Q Fever cases, contributing to public health safety.
-
Supports enhanced patient care through detailed record-keeping.
Who Needs the CDC Q Fever Case Report?
The CDC Q Fever Case Report is primarily utilized by physicians and state health department officials. These roles are critical as they must complete the form accurately to ensure comprehensive reporting of Q Fever cases. Moreover, designated professionals are required to provide signatures, which validate the information submitted.
-
Physicians responsible for diagnosing and reporting Q Fever cases.
-
State health department officials who oversee public health efforts.
How to Fill Out the CDC Q Fever Case Report Online
Filling out the CDC Q Fever Case Report online is a straightforward process. Users should first gather necessary patient information, including the patient's name and symptom details. The form features fillable fields that enhance ease of use, allowing for a clear and efficient completion process.
-
Access the CDC Q Fever Case Report form through a secure platform.
-
Input patient-specific information and clinical details as required.
-
Review each field to ensure accuracy before submission.
Key Features of the CDC Q Fever Case Report
The CDC Q Fever Case Report contains several sections that are essential for effective documentation. Important areas include clinical findings, laboratory data, and patient demographics, which must be recorded accurately for comprehensive case tracking. Ensuring all relevant sections are completed thoroughly is crucial for optimal data integrity.
-
Incorporates detailed fields for clinical findings and laboratory results.
-
Captures accurate demographic information and final diagnosis, enhancing data utility.
Submission Methods for the CDC Q Fever Case Report
Submitting the CDC Q Fever Case Report can be accomplished through various methods. Health professionals should choose the most suitable submission option based on convenience and regulatory standards. Clarity on where to send the completed form and the submission method used is critical for proper documentation.
-
Online submission for quicker processing and convenience.
-
Available mail and fax options for traditional submission methods.
Common Errors and How to Avoid Them When Filing the CDC Q Fever Case Report
Filing the CDC Q Fever Case Report requires attention to detail, as common errors can significantly affect data quality. Users should be aware of frequent mistakes and implement strategies for error minimization before submission.
-
Common mistakes include incomplete fields or inaccurate patient information.
-
Double-check entries and utilize a validation checklist for accuracy.
Security and Compliance Considerations for the CDC Q Fever Case Report
Data security is paramount when handling the CDC Q Fever Case Report. The form is designed with robust security measures, ensuring that sensitive patient information is well-protected. Compliance with regulations like HIPAA and GDPR reinforces trust in the reporting process.
-
Utilizes 256-bit encryption to protect data integrity.
-
Ensures adherence to healthcare data protection regulations.
How pdfFiller Makes Completing the CDC Q Fever Case Report Easier
pdfFiller provides numerous benefits for users completing the CDC Q Fever Case Report. The platform’s advanced features assist in document management, making the process more accessible and efficient for healthcare professionals.
-
Editing, eSigning, and sharing capabilities enhance user experience.
-
Cloud storage ensures secure and organized document management.
What to Do After Submitting the CDC Q Fever Case Report
After submitting the CDC Q Fever Case Report, it is important to follow up to ensure the data has been received and processed correctly. Users should be aware of their options for checking submission status or amending the report if necessary.
-
Steps to verify the status of the submission should be followed promptly.
-
Resources are available for support regarding any inquiries or necessary amendments.
How to fill out the Q Fever Report
-
1.Access the CDC Q Fever Case Report form on pdfFiller by searching for its name or navigating to the healthcare forms section.
-
2.Once the form is open, review the fields that need to be filled out carefully. Familiarize yourself with the structure and the types of information required.
-
3.Collect necessary patient information such as 'Patient's name', demographic details, clinical findings, and laboratory data before you start filling in the form.
-
4.Use pdfFiller's interactive interface to click into each fillable field. Input the required data accurately and check for any prompts or tooltips for guidance.
-
5.Take special note of the checkboxes for various clinical signs and symptoms, ensuring you mark all that are relevant to the case being reported.
-
6.Once all fields are completed, review your data for accuracy. Double-check details like spelling, numerical entries, and required documentation.
-
7.Finalize the form by adding signatures in the designated areas, which are crucial for validation by the physician and the health department official.
-
8.After reviewing and signing, use pdfFiller options to save, download, or submit the completed form electronically as per the guidelines.

Who is eligible to complete the CDC Q Fever Case Report?
The CDC Q Fever Case Report can be completed by licensed physicians and state health department officials. It's essential that the form is filled accurately to ensure proper disease monitoring.
What information is required to complete the form?
Essential information includes the patient's name, patient demographics, physician details, clinical findings, laboratory results, and final diagnosis. Make sure to gather this data before starting the form.
How do I submit the CDC Q Fever Case Report?
After filling out the form on pdfFiller, you can submit it electronically through the platform, or print it to submit via regular mail to the designated health department.
What are the common mistakes to avoid when filling out the form?
Ensure all fields are filled correctly to avoid incomplete submissions. Double-check that all required signatures are obtained and information is accurate to prevent delays in processing.
Is notarization required for the CDC Q Fever Case Report?
No, notarization is not required for this form. However, it does require signatures from both the physician and a state health department official.
How long does it take to process the CDC Q Fever Case Report?
Processing times for the CDC Q Fever Case Report can vary depending on the health department. Typically, expect between one to two weeks for processing after submission.
Are there any fees associated with submitting this form?
There are typically no fees associated with submitting the CDC Q Fever Case Report, but it is advisable to confirm with your local health department regarding any specific regulations.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.