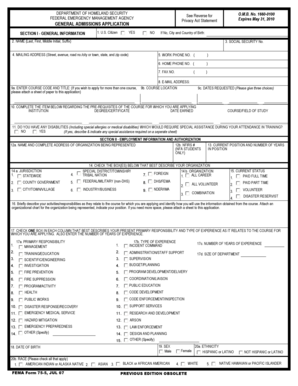Last updated on Mar 21, 2015

Get the free NIH Proposal Summary and Data Record
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is NIH Proposal Form
The NIH Proposal Summary and Data Record is a government form used by institutions to submit proposals for contracts or grants to the National Institutes of Health (NIH). Its primary purpose is to collect detailed project information for funding applications.
pdfFiller scores top ratings on review platforms




Who needs NIH Proposal Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to NIH Proposal Form
What is the NIH Proposal Summary and Data Record?
The NIH Proposal Summary and Data Record is a critical form utilized by institutions to submit proposals for grants and contracts with the National Institutes of Health (NIH). This form captures essential information such as the legal name of the institution, address, and project details.
The primary purpose of this document is to streamline the NIH grant application process by ensuring that all necessary information is accurately collected upfront. By gathering crucial details in a standardized format, it aids in compliance and project approval, ultimately facilitating more efficient funding decisions.
Information required in the form is significant; for instance, the legal name is essential for official records, while project details help evaluators understand the objectives and scope of the proposed research.
Purpose and Benefits of the NIH Proposal Summary and Data Record
This form plays an integral role in the funding application process, allowing researchers to present their proposals in a coherent format. The NIH Proposal Summary and Data Record not only serves as a submission requirement but also acts as a tool for clarity and transparency throughout the evaluation process.
The benefits of using this form include:
-
Streamlined proposal submissions to save time and reduce errors.
-
Ensured compliance with NIH requirements to enhance approval chances.
-
Facilitated project approval by providing comprehensive project insights to reviewers.
Who Needs the NIH Proposal Summary and Data Record?
The responsibility of submitting the NIH Proposal Summary and Data Record typically falls on two key roles: the Principal Investigator and the Business Representative. Both are required to sign the form to validate the submission.
Various institutions or entities, such as universities, nonprofit organizations, and research facilities, must adhere to this requirement to apply for NIH contracts or grants. Understanding the roles involved and the types of institutions that need to submit helps streamline the process for all parties involved.
Key Features of the NIH Proposal Summary and Data Record
The NIH Proposal Summary and Data Record includes several important components, ensuring clarity in submissions. Key features include:
-
Fillable fields for capturing detailed project information.
-
Signature requirements from both the Principal Investigator and the Business Representative.
-
Detailed descriptions of the project and estimated costs to provide a comprehensive overview for reviewers.
These elements are crucial for aligning proposals with NIH standards and expectations.
How to Fill Out the NIH Proposal Summary and Data Record Online (Step-by-Step)
Completing the NIH Proposal Summary and Data Record can be straightforward when following a structured approach. Here is a guide for filling it out:
-
Gather necessary information such as the legal name of the institution, project title, and estimated budget.
-
Access the form through the appropriate online platform.
-
Fill in the required fields, ensuring accuracy in each section.
-
Review all entries for completeness and correctness before submission.
-
Obtain required signatures from the Principal Investigator and the Business Representative.
By following these steps, applicants can ensure a thorough and compliant submission process.
Field-by-Field Instructions for the NIH Proposal Summary and Data Record
Understanding each field in the NIH Proposal Summary and Data Record is vital for accurate completion. Key fields include:
-
Legal Name: Ensure the institution's legal name is correctly entered as it appears in official documents.
-
Contract Type: Select the type of contract you are proposing to clarify the nature of the funding request.
-
Project Summary: Provide a concise summary that outlines the aims and objectives of the project.
Common mistakes include inaccuracies in the legal name and incomplete project summaries. Double-checking each field can help avoid these pitfalls.
Submission and Delivery of the NIH Proposal Summary and Data Record
Once the NIH Proposal Summary and Data Record is completed, it must be submitted through the designated channels. The submission process involves:
-
Saving the completed form in the specified format.
-
Sending it to the appropriate NIH department via the online portal or email as instructed.
-
Tracking the submission confirmation and following up if necessary.
Understanding the submission process ensures that proposals are received and reviewed promptly.
Security and Compliance for the NIH Proposal Summary and Data Record
The security of sensitive information in the NIH Proposal Summary and Data Record is paramount. pdfFiller implements robust measures to safeguard user data, including:
-
256-bit encryption to protect information during transmission and storage.
-
Compliance with stringent regulations such as HIPAA and GDPR to ensure data privacy.
These practices reinforce the integrity of the form's handling processes and bolster user confidence.
Support from pdfFiller in Completing Your NIH Proposal Summary and Data Record
pdfFiller offers a range of features that can significantly ease the process of completing the NIH Proposal Summary and Data Record. Key offerings include:
-
Editing capabilities to customize form fields as needed.
-
eSigning features for obtaining necessary signatures securely.
-
Document sharing options that enable collaborative completion among stakeholders.
Leveraging these tools can facilitate a smoother experience in preparing and submitting the form.
Get Started with Your NIH Proposal Summary and Data Record Today!
Utilizing pdfFiller for your NIH Proposal Summary and Data Record ensures a seamless filling and submission experience. With user-friendly features and strong security, the platform is designed to assist you every step of the way.
Experience the ease of use and support available to manage this important document effectively.
How to fill out the NIH Proposal Form
-
1.Access the NIH Proposal Summary and Data Record form on pdfFiller by searching for its title in the pdfFiller search bar.
-
2.Open the form, and familiarize yourself with the layout and available fields. Identify where to input required information.
-
3.Gather the necessary project information, including the legal name and address of the offeror, project costs, and details regarding human subjects or vertebrate animals involved.
-
4.Begin filling out the form by clicking on each field and entering the relevant details using pdfFiller's intuitive interface. Utilize the checkboxes for relevant sections.
-
5.As you fill in the form, double-check entries for accuracy, especially in critical components like budget and project description.
-
6.Once you have completed filling out all the required fields, review the entire form carefully, ensuring all necessary sections are filled and signed.
-
7.Finalize your form by clicking the ‘Save’ button to store your progress, and use the ‘Download’ option if you wish to keep a copy.
-
8.Submit the completed form directly through pdfFiller using the provided submission options, or download it to submit via traditional means.

Who is eligible to use the NIH Proposal Summary and Data Record?
The NIH Proposal Summary and Data Record is primarily for Principal Investigators and Business Representatives from institutions looking to apply for NIH grants or contracts. Eligibility typically includes research institutions, universities, and non-profit organizations.
What should I include when filling out the form?
You should provide detailed information about your project, including the project's legal name, address, type of contract proposed, estimated costs, and any involvement of human subjects or vertebrate animals.
What are the submission methods for the NIH Proposal Summary and Data Record?
You can submit the form electronically using pdfFiller or download it and submit a printed copy via mail or fax, depending on NIH submission guidelines.
Are there any common mistakes to avoid when submitting the form?
Common mistakes include omitting required signatures, providing inaccurate or incomplete project details, and failing to double-check the eligibility criteria specific to the submission.
What supporting documents might I need to submit with this form?
Supporting documents can include budget justifications, letters of support, project narratives, and Institutional Review Board approvals if human subjects or vertebrate animals are involved.
How long does it take to process the NIH Proposal Summary and Data Record?
Processing times for NIH proposals can vary widely based on the application type and NIH review schedules. It's advisable to submit well in advance of funding deadlines to accommodate any delays.
What if I have questions while completing the form?
If you have questions, you can refer to NIH guidelines, consult with your institution's grant office, or reach out to current NIH contacts for specific clarification on form requirements.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.