Last updated on Mar 21, 2015

Get the free NHANES Lead Dust Study Informed Consent
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is NHANES Lead Consent
The NHANES Lead Dust Study Informed Consent is a healthcare document used by the US to obtain consent from respondents for the collection of household dust samples for lead testing.
pdfFiller scores top ratings on review platforms




Who needs NHANES Lead Consent?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to NHANES Lead Consent
What is the NHANES Lead Dust Study Informed Consent?
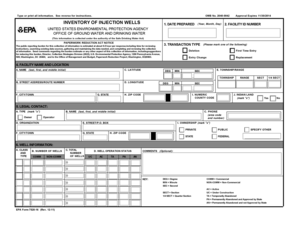
The NHANES Lead Dust Study Informed Consent form is a crucial document that defines participant involvement in the lead dust study. This form plays a significant role in informing respondents about the study's purpose and their rights prior to participation. It encompasses an overview of the NHANES Lead Dust Study and outlines the various stages of the process involved.
The informed consent form is essential for ensuring ethical standards are met and that participants understand the implications of their involvement. Participants must acknowledge their agreement, which serves to protect their rights throughout the study.
Purpose and Benefits of the NHANES Lead Dust Study Informed Consent
The primary objective of the NHANES Lead Dust Study Informed Consent is to ensure that participants are fully aware of the study's goals and potential health benefits. By clarifying these objectives, the form enhances participant understanding of how their involvement can lead to advancements in public health policies regarding lead exposure.
Participating in the NHANES Lead Dust Study contributes not only to personal knowledge but also provides noteworthy advantages for overall home environments. Understanding lead exposure can help inform families and communities about necessary safety measures.
Key Features of the NHANES Lead Dust Study Informed Consent
This informed consent form includes specific features that ensure clarity and transparency. Participants encounter fillable fields such as:
-
Respondent name
-
Signature of the respondent
-
Date
-
Signature of the interviewer
Additionally, the form outlines participant protection measures, reinforcing their rights throughout the lead dust study participation process. Effective communication about these features guarantees participants are well-informed.
Who Needs the NHANES Lead Dust Study Informed Consent?
The NHANES Lead Dust Study Informed Consent form is necessary for those who meet specific eligibility criteria to participate as respondents. Individuals are typically selected based on their demographics, such as location and potential exposure to lead.
An interview by the study team plays a key role in guiding participants through the informed consent process, emphasizing the importance of their signature. This validates their consent and confirms their willingness to engage in the study.
How to Fill Out the NHANES Lead Dust Study Informed Consent Online
Completing the NHANES Lead Dust Study Informed Consent online is a straightforward process. Here are the steps to ensure accurate completion:
-
Access the consent form via pdfFiller.
-
Fill in the required fields carefully, including your name and date.
-
Provide your signature and the interviewer's signature as required.
-
Review all entries to confirm accuracy before submission.
Each field is designed for clear input, making it easy for participants to navigate through the form completing the necessary details.
Submission Methods for the NHANES Lead Dust Study Informed Consent
Once the NHANES Lead Dust Study Informed Consent form is completed, participants have various methods of submission. They can choose to submit the form online, print it out for mail or fax submission. Each option accommodates different preferences for document handling.
It is important to be aware of any deadlines or timeliness associated with submissions to ensure participation in the study.
Security and Compliance for the NHANES Lead Dust Study Informed Consent
Participants can feel assured about the security and confidentiality of their data when handling the NHANES Lead Dust Study Informed Consent form. Comprehensive security measures are in place to protect sensitive information, including encryption and secure storage.
The form complies with regulations such as HIPAA and GDPR, further enhancing trust in the safeguarding of personal data and upholding participant privacy.
What Happens After You Submit the NHANES Lead Dust Study Informed Consent?
After submitting the NHANES Lead Dust Study Informed Consent form, participants can expect confirmation of receipt from the research team. Processing times may vary, but the study team aims to keep participants informed of their application status.
Following approval, participants will receive documentation regarding the next steps in the study, ensuring continuous communication and engagement throughout the process.
Common Errors and How to Avoid Them When Filling Out the NHANES Lead Dust Study Informed Consent
To ensure a smooth submission process, it is vital to avoid common errors when filling out the NHANES Lead Dust Study Informed Consent form. Frequent mistakes include:
-
Missing signatures in designated fields
-
Providing incorrect or incomplete information
-
Overlooking the date field
Taking time to double-check each section can enhance accuracy and facilitate effective participation in the study.
Enhance Your Experience with pdfFiller for the NHANES Lead Dust Study Informed Consent
Utilizing pdfFiller can greatly enhance your experience in completing the NHANES Lead Dust Study Informed Consent form. This platform allows for efficient document management and provides eSigning capabilities.
pdfFiller ensures users have protection while handling forms online, facilitating an easy and user-friendly completion experience.
How to fill out the NHANES Lead Consent
-
1.To begin, access pdfFiller and search for the NHANES Lead Dust Study Informed Consent form in the document library.
-
2.Once located, click to open the form in pdfFiller's editor.
-
3.Review the form to familiarize yourself with the content and the required fields.
-
4.Before filling out the form, gather all necessary information, including the respondent's name, signature, and the date.
-
5.Start by entering the respondent’s printed name in the designated field.
-
6.Next, have the respondent sign in the provided area to indicate their consent.
-
7.Then, enter the date when the respondent signed the form.
-
8.The interviewer should follow the same steps, starting with their name and signature.
-
9.After completing all required fields, take a moment to review the form for accuracy.
-
10.Ensure all necessary signatures are present and that all fields are properly completed.
-
11.Once satisfied, save the form within pdfFiller and choose the option to download it if needed.
-
12.Finally, submit the completed form as per the instructions provided by the NHANES study, either electronically or via mail.

Who is eligible to sign the NHANES Lead Dust Study Informed Consent?
Individuals residing in homes selected for the NHANES study are eligible to sign the consent form, ensuring they are informed of the study's purpose and procedures.
What is the process for submitting the NHANES consent form?
Once completed, the NHANES consent form can be submitted electronically through pdfFiller or mailed to the designated address provided by NHANES.
Are there any deadlines for signing the consent form?
Yes, participants should sign and submit the consent form promptly after being selected for the study. Specific deadlines will be provided by the study coordinators.
What common mistakes should be avoided when completing the form?
Common mistakes include failing to sign the form, missing required fields, or submitting the form with incorrect personal information. Double-check all entries before submission.
Is there a fee associated with participating in the NHANES study?
Participation in the NHANES study and signing the consent form is free of charge. Participants are not required to pay any fees.
What should I do if I need assistance while filling out the form on pdfFiller?
If you need help, pdfFiller offers support through their help center, which includes tutorials and customer service support to guide you through the form completion.
How can I ensure that my privacy is protected during the study?
The NHANES Lead Dust Study follows strict protocols to protect participants' privacy. Your information will be kept confidential and used only for research purposes.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.