Last updated on Mar 22, 2015

Get the free Form AEC-483 Registration Certificate
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is AEC-483 Form
The Form AEC-483 Registration Certificate is a document used by physicians, clinical laboratories, and hospitals to obtain authorization for in vitro testing with byproduct material under U.S. Atomic Energy Commission regulations.
pdfFiller scores top ratings on review platforms




Who needs AEC-483 Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to AEC-483 Form
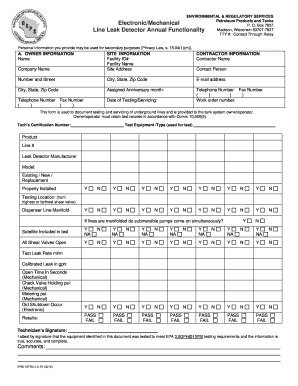
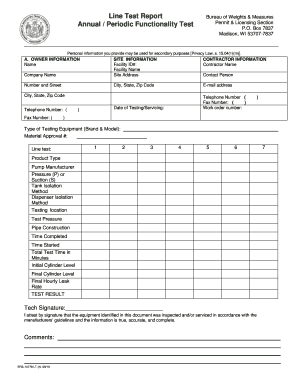
What is the Form AEC-483 Registration Certificate?
The Form AEC-483 serves as a registration certificate related to in vitro testing with byproduct materials, governed by regulations set forth by the U.S. Atomic Energy Commission. This form is crucial for entities like physicians, clinical laboratories, and hospitals, as it authorizes the possession and use of small amounts of byproduct material for clinical testing. Understanding the details of the registration certificate is essential for compliance and operational efficiency within medical settings.
Purpose and Benefits of the Form AEC-483 Registration Certificate
Obtaining the Form AEC-483 offers numerous advantages, including legal authorization to utilize byproduct material within the parameters defined by the U.S. Atomic Energy Commission. This registration certificate ensures compliance with federal regulations outlined in 10 CFR 31, which governs the use of such materials in clinical practices. By securing this certificate, institutions can operate within legal boundaries and improve their operational credibility.
Eligibility Criteria for the Form AEC-483 Registration Certificate
To successfully fill out the Form AEC-483, applicants need to meet specific eligibility criteria. Qualified applicants typically include:
-
Physicians engaged in clinical or laboratory testing.
-
Clinical laboratories licensed to operate with byproduct materials.
-
Hospitals that utilize such materials in patient care.
It is essential for applicants to ensure they meet any additional requirements outlined by the U.S. Atomic Energy Commission.
How to Fill Out the Form AEC-483 Registration Certificate Online
Filling out the Form AEC-483 online can significantly streamline the submission process. Here’s a step-by-step guide using pdfFiller:
-
Access the pdfFiller platform and locate the Form AEC-483 template.
-
Fill in the required fields, such as name, address, and other pertinent details.
-
Utilize the instructions provided within the form to ensure all sections are completed correctly.
-
Review the form for accuracy before submitting it electronically.
By following these steps, users can ensure a smooth and efficient submission process.
Common Errors and How to Avoid Them When Filling the Form AEC-483
Applicants often encounter several common pitfalls when completing the Form AEC-483. Frequent errors include:
-
Inaccurate information in key fields, such as registrant name and address.
-
Failure to check the appropriate box for the type of entity registering.
To mitigate these risks, it is advisable to meticulously review the form before submission and ensure all information is accurate and complete.
Submission Methods for the Form AEC-483 Registration Certificate
The Form AEC-483 can be submitted through various methods, including:
-
Online submission via platforms like pdfFiller.
-
Traditional mail to the designated authority.
Understanding the deadlines associated with each submission method is critical for timely processing. Applicants should familiarize themselves with any important dates relevant to their submission.
What Happens After You Submit the Form AEC-483 Registration Certificate
Once the Form AEC-483 is submitted, registrants can expect specific processes to unfold. Key details include:
-
The review process typically involves a waiting period for approval.
-
Registrants can track and confirm the status of their application via the submitting authority’s channels.
Understanding these steps helps set realistic expectations during the approval phase.
How pdfFiller Can Help with the Form AEC-483 Registration Certificate
pdfFiller offers a range of features that simplify the completion of the Form AEC-483. Users can enjoy:
-
Easy editing and filling of the form online.
-
eSigning capabilities to streamline the approval process.
-
Secure document management to comply with regulatory standards, including HIPAA and GDPR.
These features help ensure that sensitive information is handled securely while enhancing user experience.
Sample or Example of a Completed Form AEC-483 Registration Certificate
Providing examples or templates of a completed Form AEC-483 can be invaluable for users. Key components to review when filling out the form include:
-
Ensuring the accuracy of registration details.
-
Understanding the significance of each section of the form.
Sample versions can serve as helpful guides for new applicants.
Final Thoughts on Completing the Form AEC-483 Registration Certificate
Utilizing pdfFiller for filling and managing the Form AEC-483 can greatly enhance the efficiency of the process. Timely and accurate completion of the form underpins successful regulatory compliance for medical professionals and institutions.
How to fill out the AEC-483 Form
-
1.Access pdfFiller and search for the Form AEC-483 Registration Certificate to open the document.
-
2.Familiarize yourself with the form layout to locate required fields and sections.
-
3.Gather necessary information, including your name, address, and relevant registration details before starting.
-
4.Begin filling in the form by entering your name and address in the designated fields.
-
5.Use the checkboxes to select the appropriate type of registrant: physician, clinical laboratory, or hospital.
-
6.Ensure all required fields are completed accurately according to the guidelines provided.
-
7.Review the filled form for accuracy, making sure all information matches your supporting documents.
-
8.Finalize the form by clicking the 'Save' option to keep a copy with your inputs.
-
9.Download the completed form or submit it directly through pdfFiller if required by your state regulations.

Who is eligible to complete the Form AEC-483?
Eligible individuals include physicians, clinical laboratories, and hospitals that require authorization for in vitro testing involving byproduct material under U.S. Atomic Energy Commission regulations.
What is the deadline for submitting the AEC-483 Registration Certificate?
Deadlines may vary based on your specific state regulations. It is advisable to check with your local Atomic Energy Commission office or relevant authorities.
How do I submit the completed Form AEC-483?
You can submit the completed form online through pdfFiller or by printing it out and mailing it to the appropriate regulatory authority. Ensure you follow the submission guidelines for your area.
What supporting documents are required with the AEC-483 form?
Typically, you may need to provide identification, proof of address, and any previous licensing information, depending on the specific requirements in your state.
What common mistakes should I avoid when filling out this form?
Common mistakes include missing signatures, incomplete fields, or inaccuracies in the registrant's information. Always double-check before submission.
How long does processing take for the AEC-483 Registration Certificate?
Processing times can vary, but applicants should expect a response within several weeks after submission. It's advisable to confirm details with the regulatory office.
What should I do if I receive a rejection for my application?
If your application is rejected, review the feedback provided for errors or missing information. Address those issues before resubmitting the form.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.