Last updated on Apr 2, 2015

Get the free Registration Certificate In Vitro Testing
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is In Vitro Testing Form
The Registration Certificate In Vitro Testing is a government document used by healthcare providers to register for the use of byproduct material for in vitro clinical or laboratory tests.
pdfFiller scores top ratings on review platforms




Who needs In Vitro Testing Form?
Explore how professionals across industries use pdfFiller.
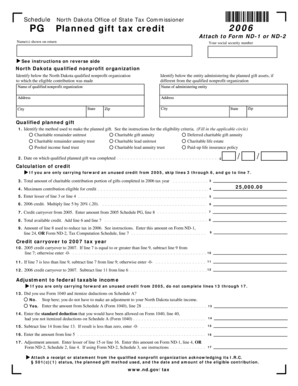
Comprehensive Guide to In Vitro Testing Form
What is the Registration Certificate In Vitro Testing?
The Registration Certificate In Vitro Testing is a critical document designed for physicians, clinical laboratories, and hospitals involved in in vitro testing. This form serves to register for the legal use of byproduct material in laboratory tests, ensuring compliance with safety regulations. Issued by the U.S. Atomic Energy Commission, it plays a vital role in maintaining industry standards.
This in vitro testing form, also referred to as the registration certificate form, confirms that the entities using byproduct materials are operating under government guidelines, enhancing accountability in laboratory practices.
Purpose and Benefits of the Registration Certificate In Vitro Testing
The primary purpose of the registration certificate is to establish a legal framework for entities conducting clinical testing. Physicians, clinical laboratories, and hospitals must complete this form to remain compliant with regulatory standards. By properly utilizing this medical testing form or hospital registration form, these organizations ensure safe practices in their operations.
Among the key benefits is the assurance of legal compliance, which protects both the institutions and patients involved. Moreover, it fosters a culture of safety and morality in handling potentially hazardous materials.
Who Needs to Complete the Registration Certificate In Vitro Testing?
Registrants for the Registration Certificate In Vitro Testing primarily include physicians, clinical laboratories, and hospitals. Each entity must assess the specific conditions under which they are required to file this clinical laboratory form. For example, hospitals may have different requirements compared to private labs, influencing who must actively undertake this registration process.
Understanding the distinctions in registration needs is essential for all potential registrants, ensuring adherence to appropriate guidelines.
Eligibility Criteria for the Registration Certificate In Vitro Testing
To qualify for the Registration Certificate In Vitro Testing, entities must meet specific eligibility criteria that often include licensing requirements and operational qualifications. These requirements ensure that only qualified professionals and organizations handle byproduct material.
Restrictions may apply, particularly regarding the type and amount of nuclear material that can be utilized for testing. These limitations are essential to safeguard public health and safety.
How to Fill Out the Registration Certificate In Vitro Testing Online (Step-by-Step)
Filling out the registration certificate form online can be streamlined using pdfFiller. To ensure successful completion, follow these steps:
-
Access the registration certificate form on pdfFiller.
-
Fill in personal and organizational details such as name and address accurately.
-
Ensure compliance checkboxes are marked as necessary.
-
Review for any common pitfalls, such as missing signatures.
-
Submit the form digitally through the platform.
Each field has specific guidelines that must be carefully followed to avoid delays in processing.
Common Errors and How to Avoid Them When Filling Out the Form
When completing the registration certificate form, mistakes can lead to significant delays or rejections. Some common errors include:
-
Incomplete fields or missing signatures.
-
Incorrectly entered information that does not match official documents.
-
Failing to certify compliance with health and safety regulations.
To avoid these issues, double-check all entries for accuracy and ensure that the form is thoroughly reviewed before submission.
Where and How to Submit the Registration Certificate In Vitro Testing
After completing the registration certificate form, the submission can occur through various methods such as online submissions via pdfFiller or by mail. Registrants should be aware of critical deadlines to ensure their applications are timely and processed without delays.
Understanding the processing times can also help registrants plan accordingly, providing peace of mind in the registration process.
What Happens After You Submit the Registration Certificate In Vitro Testing?
Once the registration certificate has been submitted, applicants can expect a confirmation of receipt. This confirmation provides assurance that the application is being processed. To track the application status, registrants can refer to communication from the issuing authority, ensuring transparency throughout the review process.
In some cases, follow-up actions may be required depending on the review of the submitted form.
How to Correct or Amend the Registration Certificate In Vitro Testing
If corrections or amendments to the registration certificate form are needed after submission, it is essential to follow a systematic approach. Begin by identifying the specific errors that require rectification and gather necessary documentation to support your amendments.
Keeping the information updated is pivotal for compliance, ensuring that all regulatory standards are continually met.
Utilizing pdfFiller for the Registration Certificate In Vitro Testing
pdfFiller offers robust features that facilitate the completion and management of the registration certificate form. Key capabilities include the ability to edit text and images, create fillable forms, and eSign documents securely.
With a focus on security, pdfFiller employs 256-bit encryption and complies with HIPAA and GDPR standards, ensuring that sensitive documents are managed safely and effectively.
How to fill out the In Vitro Testing Form
-
1.Access pdfFiller and search for 'Registration Certificate In Vitro Testing' in the forms section.
-
2.Open the form in pdfFiller's editor once you locate it.
-
3.Begin by gathering essential information, such as your name, address, and compliance certifications.
-
4.Navigate through the form, filling in each blank field methodically using the provided prompts.
-
5.Use pdfFiller's tools to check off relevant checkboxes as required in the form.
-
6.Review your completed form for any potential errors or missed fields.
-
7.Once satisfied with the information entered, finalize the form using the 'Save' option.
-
8.Download the completed form to your device, or choose to submit it directly through pdfFiller if available.

Who is eligible to use the Registration Certificate In Vitro Testing?
Eligibility typically includes healthcare providers such as physicians, clinical laboratories, and hospitals that intend to use byproduct materials for in vitro testing.
What documents are needed to complete the form?
You will need your personal identification, address details, and any necessary compliance documents related to byproduct materials prior to filling out the registration form.
How is the completed form submitted?
The completed Registration Certificate In Vitro Testing can be submitted online via pdfFiller or printed and mailed directly to the U.S. Atomic Energy Commission, depending on the instructions provided.
Are there any deadlines for submitting this registration form?
Deadlines may vary based on institutional requirements; however, it is advisable to submit the form well in advance of the intended use of byproduct materials to ensure compliance.
What common mistakes should be avoided when filling out this form?
Ensure that all fields are completed accurately, double-check for any typos in your name or address, and make sure to sign the form where required to prevent rejection.
How long does it take to process this registration?
Processing times can vary; typically, expect a few weeks for review and approval. Contact the issuing authority for specific time frames.
Can the form be notarized?
No, notarizing is not required for this form. Ensure all information is accurate before submission, as it is legally binding without a notary.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.