Last updated on Jun 17, 2015

Get the free FDA Drug Establishment Registration Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is FDA Drug Registration
The FDA Drug Establishment Registration Form is a government document used by drug manufacturers and distributors to register their establishments with the U.S. Food and Drug Administration (FDA).
pdfFiller scores top ratings on review platforms




Who needs FDA Drug Registration?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to FDA Drug Registration
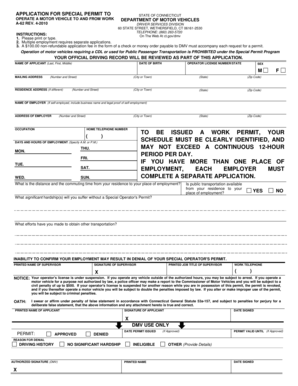
What is the FDA Drug Establishment Registration Form?
The FDA Drug Establishment Registration Form is a critical document for the pharmaceutical industry, facilitating compliance with the U.S. Food and Drug Administration (FDA). This form serves to register establishments that manufacture and distribute drugs, ensuring they adhere to regulatory requirements. By maintaining accurate and updated records, this registration process plays a pivotal role in the integrity of drug establishment operations.
The primary mission of the FDA Drug Establishment Registration Form is to ensure that all involved parties operate within the legal standards established by the FDA. This document is central to maintaining regulatory compliance, which is vital for the safety and efficacy of drugs in the market.
Purpose and Benefits of the FDA Drug Establishment Registration Form
The FDA Drug Establishment Registration Form is necessary because it establishes a legal framework for drug manufacturers and distributors. By completing this registration, businesses confirm their commitment to compliance with FDA regulations. Non-compliance can lead to serious legal implications, including fines and penalties.
Utilizing the FDA compliance form boosts a business's operational integrity, instilling confidence in consumers and regulatory bodies alike. The registration process not only protects the interests of the businesses but also ensures that products meet safety standards.
Who Needs to Complete the FDA Drug Establishment Registration Form?
The target audience for the FDA Drug Establishment Registration Form includes various professionals and entities in the pharmaceutical sector. Key stakeholders required to register are manufacturers, distributors, and other related entities. Each role necessitates a thorough understanding of the responsibilities involved in this regulatory process.
-
Authorizing Officials, who must sign on behalf of the organization
-
Distributors, responsible for the distribution of drug products
Both roles are crucial for maintaining compliance and ensuring that all operations meet FDA guidelines.
How to Fill Out the FDA Drug Establishment Registration Form Online (Step-by-Step)
Filling out the FDA Drug Establishment Registration Form online using pdfFiller is straightforward. Here are the steps to complete the process effectively:
-
Access the FDA form 2656 via the pdfFiller platform.
-
Fill in the necessary fields, ensuring all provided information is accurate.
-
Utilize the platform’s features, such as templates and auto-fill, for efficiency.
-
Gather all required information beforehand to streamline the process.
-
Review for any potential errors before submitting the form.
Field-by-Field Instructions for the FDA Drug Establishment Registration Form
Each section of the FDA Drug Establishment Registration Form requires careful attention. Understanding essential fields helps avoid common mistakes such as incorrect data entry or neglecting required signatures. Key areas to focus on include:
-
Firm Information: Ensure accurate names and addresses.
-
Compliance Mailing Address: Confirm the responsibility for receiving compliance notices.
Users are advised to refer to specific guidelines within the form to navigate complex sections efficiently, particularly concerning the drug labeling code.
Submission Methods for the FDA Drug Establishment Registration Form
Once the FDA Drug Establishment Registration Form is completed, there are several submission options available. Users can submit the form via:
-
Online submission through the pdfFiller platform.
-
Traditional mail to the FDA, ensuring adherence to submission deadlines.
-
Electronic platforms if applicable.
It is crucial to include all required documents and be aware of any associated fees during the submission process.
Consequences of Not Filing or Late Filing the FDA Drug Establishment Registration Form
Failing to file the FDA Drug Establishment Registration Form or submitting it late can bring significant consequences. Legal penalties may include hefty fines and the potential for operational delays. Such repercussions can severely impact business continuity and regulatory standing.
Timely submission plays a vital role in maintaining compliance status with the FDA, ensuring that companies remain in good standing as drug manufacturers or distributors.
Security and Compliance for the FDA Drug Establishment Registration Form
pdfFiller prioritizes document security throughout the registration process. With features like 256-bit encryption, users can be assured that their sensitive information is protected. The platform complies with regulatory standards such as HIPAA and GDPR, ensuring that data privacy is maintained at all times.
Utilizing secure document handling practices reassures businesses that their compliance efforts are safeguarded during registration.
How pdfFiller Can Help with Filling Out the FDA Drug Establishment Registration Form
pdfFiller brings numerous advantages for users filling out the FDA Drug Establishment Registration Form. Key features that enhance the filling and eSigning experience include:
-
Intuitive form templates to reduce errors.
-
eSigning functionality for seamless approval processes.
Additionally, pdfFiller offers customer service support for inquiries and personalized assistance, making it easier for users to navigate the registration process.
Get Started with Your FDA Drug Establishment Registration Form Today!
Utilizing pdfFiller for your FDA Drug Establishment Registration Form can streamline the registration process. The platform not only facilitates easy completion and submission, but it also ensures your documents are secured. Experience the benefits of using pdfFiller to maintain compliance and operational integrity today.
How to fill out the FDA Drug Registration
-
1.Begin by visiting pdfFiller and searching for 'FDA Drug Establishment Registration Form' in the search bar.
-
2.Once located, click on the form to open it in pdfFiller's editing interface.
-
3.Before filling out the form, gather all necessary information, including details about your establishment, compliance addresses, and signatures from authorized officials.
-
4.Start filling in the form by clicking on each required field, following the prompts to provide detailed information about your firm and site.
-
5.Use the checkbox options where indicated, ensuring all required fields are completed accurately.
-
6.As you complete the form, regularly save your progress to prevent loss of information.
-
7.Once all fields are filled and accurate, review the entire form carefully to ensure all information is correct and complete.
-
8.Finalize the form by adding necessary electronic signatures from authorized officials and distributors.
-
9.To save your completed form, click on the 'Save' option, then choose to download it in your preferred format.
-
10.For submission, follow the guidelines provided on the FDA website or your chosen submission method, ensuring you send the form to the correct office.

Who is eligible to submit the FDA Drug Establishment Registration Form?
Eligible submitters include drug manufacturers and distributors who need to register their establishments with the FDA. Authorized officials from these entities must sign the form.
What are the common mistakes to avoid when completing this form?
Common mistakes include leaving required fields blank, providing incorrect or outdated information, and failing to obtain the necessary signatures from authorized officials.
How should I submit the FDA Drug Establishment Registration Form?
The form should be submitted according to the specific submission guidelines provided by the FDA. Typically, submissions can be made electronically or via mail, depending on current regulations.
Are there any deadlines for submitting the FDA Drug Establishment Registration Form?
Yes, there are deadlines for registration, especially when facilities are newly established or if there are changes in ownership or operations. It's crucial to check the FDA's current guidelines for specific timelines.
What supporting documents are required with the FDA Drug Establishment Registration Form?
Supporting documents may include proof of business operation, compliance certificates, and any other documentation requested by the FDA, depending on your specific registration requirements.
How long does it take to process the FDA Drug Establishment Registration Form?
Processing times can vary based on the volume of submissions the FDA receives, but it typically takes several weeks to a few months. It’s best to submit well ahead of any deadlines.
Is notarization required for this form?
No, notarization is not required for the FDA Drug Establishment Registration Form, but it does require signatures from authorized officials.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.