Last updated on Jul 23, 2015

Get the free Integrated Research Application System Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is IRAS Form
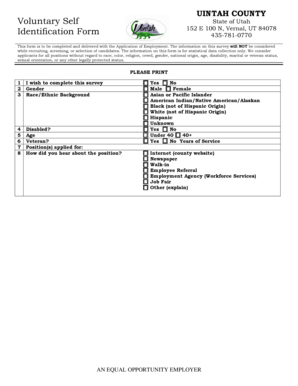
The Integrated Research Application System Form is an application form used by researchers in the UK to obtain approval for health and social care research projects.
pdfFiller scores top ratings on review platforms




Who needs IRAS Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to IRAS Form
What is the Integrated Research Application System Form?
The Integrated Research Application System (IRAS) Form serves as a crucial tool in the UK health and social care research sector. It collects essential details about research projects, encompassing study design and ethical considerations. This form plays a significant role in the research application process by enabling ethical review and approval.
Included in the form are critical components that guide researchers, such as objectives, study methodology, and participant rights. Understanding these elements is imperative for submitting a successful application to the UK Research Ethics Committee.
Purpose and Benefits of the Integrated Research Application System Form
The IRAS Form is vital for researchers seeking ethical approval for health and social care studies. Its standardized application process ensures efficiency and clarity, reducing complexities in proposal submissions. By using this form, researchers contribute to the protection of participant rights and help ensure research integrity.
The benefits of employing the IRAS include streamlined communication between stakeholders and enhanced compliance with ethical guidelines, which are essential elements in health research applications.
Who Needs the Integrated Research Application System Form?
Several key personnel are required to complete the IRAS Form, primarily the Chief Investigators and Sponsor Contacts. These roles are fundamental as they provide oversight and accountability during the research application process.
Additionally, other stakeholders such as research team members and institutional representatives may also be involved, depending on the type of research being proposed. This collaborative approach ensures that all aspects of the study are adequately covered.
How to Fill Out the Integrated Research Application System Form Online
Filling out the IRAS Form online can be done effectively using pdfFiller. Here’s how to go about it:
-
Access the form in pdfFiller and fill in the project title and Chief Investigator details accurately.
-
Ensure all required sections are completed, paying close attention to multi-field inputs and checkboxes.
-
Attach any necessary documents as per the guidelines provided in the form.
Accurate data entry is paramount as it influences the processing speed of your application, making the understanding of each required field essential.
Field-by-Field Instructions for the Integrated Research Application System Form
Each field on the IRAS Form is designed to capture specific information vital for ethical review. For instance, the question 'Is your project research?' must be answered carefully.
Common mistakes to avoid include leaving mandatory fields blank and misinterpreting the guidelines for each section. It's important to provide clear, compliant information to facilitate a smooth review process.
How to Sign and Submit the Integrated Research Application System Form
When it comes to signing the IRAS Form, digital signatures are accepted via pdfFiller, ensuring a convenient submission process. There are various methods for submission including online uploads and email options, providing flexibility based on your preferences.
Be mindful of important deadlines to maintain your research timeline and consider tracking your submission status to ensure everything proceeds smoothly.
Consequences of Not Filing or Late Filing the Integrated Research Application System Form
Failure to file the IRAS Form or late submissions can lead to significant delays in your research start dates. There are potential ethical implications and administrative penalties that could arise from improper filing practices.
Adhering to filing deadlines is critical to avoid complications that could derail your research project. Ensuring timely submission protects you from unwanted setbacks.
Security and Compliance for the Integrated Research Application System Form
When handling sensitive information through the IRAS Form, pdfFiller employs robust security measures including 256-bit encryption, ensuring compliance with GDPR and other regulations. This dedication to data protection safeguards personal and research-related information.
It is essential to understand and maintain compliance with local guidelines, which pdfFiller addresses throughout the document handling process, providing peace of mind for researchers.
Engage with pdfFiller for Your Integrated Research Application System Form Needs
pdfFiller offers a user-friendly platform that simplifies the process of completing the IRAS Form. Key features like eSigning and fillable forms enhance the experience and efficiency of form completion.
With easy access from any browser, pdfFiller makes managing your application a straightforward task, encouraging users to take advantage of these capabilities for optimal results.
How to fill out the IRAS Form
-
1.Begin by accessing pdfFiller and searching for the Integrated Research Application System Form using the search bar.
-
2.Once you locate the form, click on it to open it within the pdfFiller interface.
-
3.Before filling out the form, gather all necessary information such as project title, research methodologies, and ethical considerations.
-
4.Navigate through the form by clicking on each fillable field. Use the tab key to move quickly between sections and fill in all required information.
-
5.Answer questions thoughtfully, ensuring clarity and completeness. Pay attention to sections that require checkboxes or specific signatures.
-
6.Once you have completed all fields, review your entries carefully for accuracy and completeness.
-
7.Make sure to finalize the document by saving your changes. Choose the save option to store it in your pdfFiller account for easy access later.
-
8.If you need to download a copy, select the download option to get it in your preferred format.
-
9.Once finalized, you may submit the form directly through the instructions provided by your institution or organization.

What are the eligibility requirements for using the Integrated Research Application System Form?
Researchers associated with health and social care projects in the UK are eligible to use this form. It is specifically designed for Chief Investigators and their teams.
What is the typical processing time for the approvals once the form is submitted?
Processing times can vary significantly depending on the ethics review board. Generally, it may take several weeks to obtain feedback or approval.
What submission methods are available for the Integrated Research Application System Form?
The form is typically submitted through the relevant ethics committee's portal or via electronic submission as per institutional guidelines outlined in the document.
What supporting documents are required when submitting this form?
You may need to provide supporting materials such as study protocols, consent forms, and information sheets detailing the research process and ethical considerations.
What common mistakes should I avoid while filling out the Integrated Research Application System Form?
Ensure that all required fields are filled out completely and accurately. Common mistakes include incomplete signatures and unclear project descriptions.
Are there any fees associated with submitting this form?
Generally, there are no fees directly associated with submitting the Integrated Research Application System Form; however, check with your institution for any internal processing fees.
How do I know if I need to notarize the form before submission?
This form does not require notarization. However, check with your institution’s specific requirements to ensure compliance with their policies.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.