Last updated on Sep 10, 2015

Get the free MESA Participant Tracking Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is MESA Tracking Form
The MESA Participant Tracking Form is a medical history document used by researchers to record and update participant information for the Multi-Ethnic Study of Atherosclerosis.
pdfFiller scores top ratings on review platforms




Who needs MESA Tracking Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to MESA Tracking Form
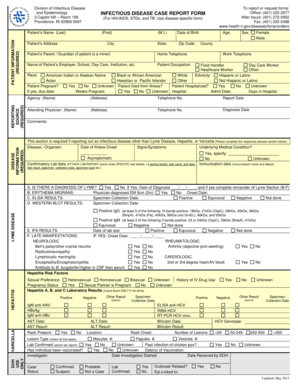
What is the MESA Participant Tracking Form?
The MESA Participant Tracking Form is a vital document used in the Multi-Ethnic Study of Atherosclerosis to collect and update participant information. This form is essential for tracking various details, such as changes in addresses and healthcare providers. It enables researchers to maintain accurate records, which are crucial for the ongoing research associated with the study.
The form captures key participant data and changes, ensuring that researchers have the most up-to-date information to analyze trends in health and disease among diverse populations.
Purpose and Benefits of the MESA Participant Tracking Form
The MESA Participant Tracking Form serves multiple purposes that benefit both participants and researchers. Accurate participant tracking is essential for the continuation of research activities, as it ensures data integrity and reliability.
Maintaining updated medical histories through this form provides healthcare providers with necessary insights into participant health, allowing for more tailored care. Furthermore, the form enhances the overall research process by ensuring that participant information changes, such as new healthcare providers or addresses, are documented effectively.
Key Features of the MESA Participant Tracking Form
This form is equipped with a variety of key features and fields that facilitate user input. Notably, it includes sections such as:
-
Participant Id#
-
Date
-
Street address
-
Healthcare provider updates
The design includes blank fields and checkboxes that allow participants to easily enter their information. Additionally, the document incorporates security measures to protect sensitive data entered within the form.
Who Needs the MESA Participant Tracking Form?
The MESA Participant Tracking Form is primarily designed for participants involved in the MESA study, making it critical for them to fill out this document during their enrollment or follow-up processes. Healthcare providers may also require updated participant information for effective patient management and care coordination.
Other stakeholders, such as researchers and data managers, can also benefit from the accurate and updated data provided through this form, ensuring comprehensive insights for the study.
How to Fill Out the MESA Participant Tracking Form Online (Step-by-Step)
Filling out the MESA Participant Tracking Form online can be done efficiently by following these steps:
-
Access the MESA Participant Tracking Form on the pdfFiller platform.
-
Complete fields such as 'Participant Id#', 'Date', and 'Street address' accurately.
-
Use checkboxes for any applicable updates.
-
Review all information to ensure accuracy before submission.
-
Utilize pdfFiller features for easy data entry and validation.
These practices will help in submitting a well-completed medical history update form efficiently.
Submission Methods and Keeping Your Information Secure
Once the MESA Participant Tracking Form is completed, electronic submission can be made conveniently through pdfFiller. The platform ensures the safety of your information by employing 256-bit encryption and compliance with HIPAA regulations.
Participants are also provided with instructions on how to confirm their form submission and track the status of their updates, reinforcing the importance of maintaining information security throughout the process.
Common Errors and How to Avoid Them
Filling out the MESA Participant Tracking Form can often present challenges; however, being aware of common errors can help mitigate these issues. Typical mistakes include:
-
Incomplete fields that may delay processing.
-
Incorrect formatting when entering contact information.
-
Failure to review all entries before submission.
To avoid these pitfalls, participants should follow the instructions carefully, ensuring all changes are documented in the database, thus minimizing errors and enhancing data reliability.
What Happens After You Submit the MESA Participant Tracking Form
After submitting the MESA Participant Tracking Form, participants can expect a defined processing time for their updates. They can also check the status of their application or information via the appropriate channels.
Maintaining open communication with relevant healthcare providers is crucial to address any questions or further updates required after submission. Remaining proactive helps ensure that all participant information changes are properly reflected in the medical history update form.
Experience the Ease of Using pdfFiller for Your MESA Participant Tracking Form
pdfFiller significantly enhances the experience of handling the MESA Participant Tracking Form with its key capabilities. Users can easily edit, sign, and share documents from any web browser, ensuring a seamless process for managing forms.
The platform's cloud-based features offer flexibility and accessibility, enabling participants to fill out forms at their convenience. Additionally, with a focus on security and compliance, pdfFiller prioritizes the safe handling of sensitive information associated with the Atherosclerosis study form.
How to fill out the MESA Tracking Form
-
1.Open pdfFiller and search for 'MESA Participant Tracking Form' using the search bar.
-
2.Click on the form title to load the document into the pdfFiller interface.
-
3.Familiarize yourself with the form layout, locating fields like 'Participant Id#', 'Date', and 'Street address.'
-
4.Before filling the form, gather required information such as personal identifiers and secondary contact details.
-
5.Start filling out the fields by clicking in each blank space and typing the relevant information.
-
6.Use checkboxes provided for selections, ensuring you mark only those that apply.
-
7.Refer to the instructions indicating 'Enter all changes into the MESA database' as you make updates.
-
8.After all fields are completed, review the entries carefully for accuracy and completeness.
-
9.Save your work by clicking on the 'Save' button located at the top right corner.
-
10.Once satisfied, you can either download the completed form or submit it directly through pdfFiller using the available options.

Who can fill out the MESA Participant Tracking Form?
The form should be completed by participants of the Multi-Ethnic Study of Atherosclerosis or authorized healthcare providers who manage participant data.
Are there deadlines for submitting this form?
While specific deadlines may vary by study phase, it is important to submit the MESA Participant Tracking Form promptly following any changes to ensure accurate tracking.
How do I submit the MESA Participant Tracking Form?
You can submit the form directly through pdfFiller by using the submission options provided once you've completed and saved your document.
What supporting documents are needed with the form?
Generally, you may need to provide identification or health history documents; however, specific requirements can vary based on your role in the study.
What are common mistakes to avoid when filling out the form?
Be careful not to leave any required fields blank, and ensure that all information is current and accurately reflects your details to avoid processing delays.
How long does it take to process the form after submission?
Processing times may vary, but generally, you can expect confirmation of your submission within a few days, depending on the study's specific administrative protocols.
What if I need to make changes after submitting the form?
If corrections are necessary after submission, contact the study coordinator to request updates, as maintaining accurate participant information is crucial.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.