Last updated on Sep 30, 2015

Get the free Radiopharmaceutical Vendor Qualification Checklist
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Radiopharmaceutical Checklist
The Radiopharmaceutical Vendor Qualification Checklist is a checklist used by nuclear pharmacies to evaluate and ensure compliance of vendors supplying radiopharmaceuticals.
pdfFiller scores top ratings on review platforms




Who needs Radiopharmaceutical Checklist?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Radiopharmaceutical Checklist
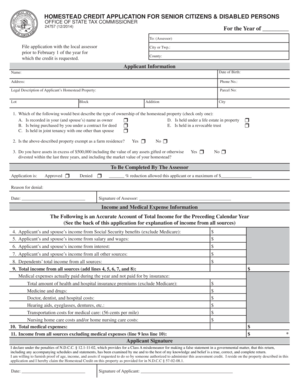
What is the Radiopharmaceutical Vendor Qualification Checklist?
The Radiopharmaceutical Vendor Qualification Checklist is an essential tool for nuclear pharmacies used to evaluate potential vendors supplying radiopharmaceuticals. This checklist serves a critical purpose in ensuring compliance with regulatory standards and maintaining safety measures. Its structured format assists pharmacy compliance teams in making informed decisions while assessing vendor capabilities.
Purpose and Benefits of the Radiopharmaceutical Vendor Qualification Checklist
This checklist is necessary for ensuring quality and safety in medication administration. By utilizing the checklist, pharmacies can assess vendor compliance and verify supporting documentation. Such evaluations are vital to uphold high standards in nuclear pharmacy settings and mitigate risks associated with radiopharmaceutical use.
Key Features of the Radiopharmaceutical Vendor Qualification Checklist
The checklist includes several notable features designed to enhance effectiveness:
-
Sections dedicated to regulatory compliance, ensuring adherence to industry standards.
-
Quality measures that reflect patient safety criteria essential for medication handling.
-
Checkboxes and fillable fields that facilitate easy completion of the form.
Who Needs the Radiopharmaceutical Vendor Qualification Checklist?
This checklist is targeted at various professionals including pharmacy compliance officers, quality assurance teams, and vendor management personnel. It is essential for anyone involved in the procurement of radiopharmaceuticals, as they play a crucial role in upholding compliance and ensuring safety policies are met.
Required Documents and Supporting Materials
Completing the checklist necessitates several supporting documents. Important materials include:
-
Vendor certifications that verify compliance with industry standards.
-
Safety data sheets crucial for understanding potential risks.
-
Compliance reports that demonstrate adherence to regulations.
Providing comprehensive documentation helps avoid compliance issues that could adversely affect medication administration.
How to Fill Out the Radiopharmaceutical Vendor Qualification Checklist Online (Step-by-Step)
To accurately complete the checklist using pdfFiller, follow these steps:
-
Access the Radiopharmaceutical Vendor Qualification Checklist on pdfFiller.
-
Fill out the required fields, marking checkboxes where applicable.
-
Review your entries for accuracy before finalizing the form.
-
Save your completed checklist for submission.
Be mindful of common pitfalls like omitting required fields or neglecting to gather supporting documents, as these can hinder form completion.
Digital Signature vs. Wet Signature Requirements
There are important distinctions between digital and wet signatures when handling the checklist. Digital signatures, facilitated by pdfFiller, enhance security and streamline the submission process, whereas traditional wet signatures may delay compliance. Understanding these differences is crucial for effective documentation.
How to Submit the Radiopharmaceutical Vendor Qualification Checklist
Submitting the completed checklist can be done through various methods, including:
-
Email submission directly to regulatory bodies.
-
Using an online portal for streamlined processing.
Keep track of your submission status to ensure all compliance requirements have been met efficiently.
Security and Compliance when Using the Radiopharmaceutical Vendor Qualification Checklist
Handling sensitive information with care is paramount. pdfFiller employs robust security measures, including 256-bit encryption, to protect user data. Additionally, compliance with regulations such as HIPAA and GDPR is maintained, ensuring confidentiality for vendor and patient data alike.
Get Started with the Radiopharmaceutical Vendor Qualification Checklist Using pdfFiller
Utilizing pdfFiller for completing the Radiopharmaceutical Vendor Qualification Checklist offers numerous advantages. Users benefit from enhanced efficiency, security, and ease of use. Accessing the checklist on the pdfFiller platform simplifies the process, making it an ideal choice for nuclear pharmacies looking to streamline vendor evaluations.
How to fill out the Radiopharmaceutical Checklist
-
1.Start by accessing pdfFiller and logging into your account. If you don’t have an account, create one to proceed.
-
2.Use the search bar to find the 'Radiopharmaceutical Vendor Qualification Checklist.' Click on the document to open it.
-
3.Familiarize yourself with the form layout. Identify all sections, including regulatory compliance, quality measures, and safety guidelines.
-
4.Gather necessary information such as vendor details, compliance standards, and any required supporting documentation before filling out the form.
-
5.Using the editing tools provided by pdfFiller, click on each checkbox to indicate compliance, and fill in any required fields with relevant details.
-
6.Follow the instructions on the form for each section, ensuring to confirm compliance and note any supporting documents needed.
-
7.Once you have completed the checklist, review all your responses. Make sure there are no missing fields or unchecked boxes.
-
8.Finalize the form by saving your changes. You can either download the checklist for your records or submit it directly to the required department.
-
9.If you intend to submit electronically, select the submission option on pdfFiller and follow the prompts to ensure proper sending.

Who should fill out the Radiopharmaceutical Vendor Qualification Checklist?
The checklist should be filled out by nuclear pharmacies, pharmacy compliance officers, or any authorized personnel responsible for evaluating vendors supplying radiopharmaceuticals.
What supporting documents are required for this form?
You may need to gather vendor compliance certifications, quality assurance documents, and safety procedure manuals to accompany the checklist.
Are there any deadlines for submitting the checklist?
While specific deadlines can vary, it is ideal to complete and submit the checklist prior to vendor engagement to ensure compliance and safety.
Can I submit the checklist electronically?
Yes, you can submit the Radiopharmaceutical Vendor Qualification Checklist electronically through pdfFiller, ensuring proper protocols are followed for vendor evaluation.
What are common mistakes to avoid when completing the checklist?
Common mistakes include leaving checkboxes unchecked, omitting required supporting documentation, and providing incomplete information on vendor compliance.
How long does it take to process the completed checklist?
Processing times can vary based on the reviewing department, but aim for a few business days for evaluation after submission.
Is notarization required for this form?
No, the Radiopharmaceutical Vendor Qualification Checklist does not require notarization, making it easier for you to complete and submit.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.