Last updated on Mar 30, 2026

Quality Medical Devices Agreement Template free printable template
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Quality Medical Devices Agreement Template
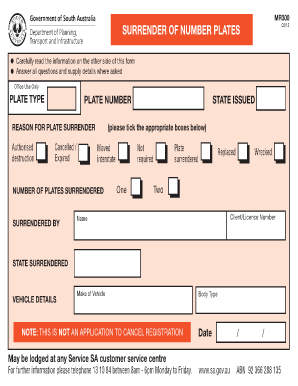
The Quality Medical Devices Agreement is a business contract used by manufacturers and distributors to outline terms for supplying medical devices and ensuring adherence to quality standards.
pdfFiller scores top ratings on review platforms




It is very good for the most part; however, the pop-ups are very distracting. Also, when filling up a table with numbers, is is hard to line them up.
so far so good. very easy and great support
love it, just what I needed for my work.
just trying to figure things out. But it has helped me.
I like it so far. It is providing me with what I need and I can already think of other ways for it to benefit my business.
I haven't quite figured it out yet without having to go to support
Who needs Quality Medical Devices Agreement Template?
Explore how professionals across industries use pdfFiller.
Quality Medical Devices Agreement Template Guide
This guide elaborates on how to effectively utilize a Quality Medical Devices Agreement Template form. Ensuring compliance with industry standards and legal obligations is crucial in the medical realm to mitigate risks associated with device quality and safety.
What is a Quality Medical Devices Agreement?
A Quality Medical Devices Agreement (QAA) is a formal contract between manufacturers and distributors that outlines the quality assurance expectations and responsibilities related to medical devices. Such agreements play a pivotal role in the medical industry, ensuring that all parties meet specific quality standards and regulatory requirements.
-
A legal document defining the quality parameters for medical devices and responsibilities between involved parties.
-
It ensures compliance with necessary regulations, bolsters product quality, and protects stakeholders.
-
Terms often include 'quality assurance,' 'regulatory compliance,' and 'defective products handling.'
What are the essential components of a Quality Medical Devices Agreement?
-
Clearly defines the start date of the agreement and specifies the parties engaged in the agreement.
-
Outlines the main goal of the agreement, usually to ensure quality and safety measures are in place.
-
Covers key terms used throughout the document to avoid ambiguity.
-
Defines which medical devices are included under the agreement.
-
Defines specific quality control measures that must be adhered to by all parties involved.
What key clauses should be included?
-
Details the standards that manufacturers must adhere to, such as ISO standards.
-
Locates mandatory compliance points with local and international laws.
-
Establishes the process for managing defective products and any potential liability.
-
Defines what each party is entitled to and responsible for within the partnership.
How to fill out the Quality Medical Devices Agreement Template?
-
Carefully read the template and fill in information in a logical order, following the prompts.
-
Prepare key documents like company licenses and specifications prior to filling out the template.
-
Always double-check details and legal terms or consult a legal expert when necessary.
What interactive tools can help manage your Quality Medical Devices Agreement?
-
Easily edit PDFs, add digital signatures, and ensure document integrity all from one platform.
-
Share agreements in real-time with collaborators and assign action items to keep projects moving forward.
-
Monitor revisions made to the agreement and document all discussions surrounding it.
What common mistakes should be avoided in Quality Medical Devices Agreements?
-
Failing to incorporate essential regulatory compliance can lead to legal pitfalls and product recalls.
-
Ambiguities can result in disputes; clear definitions provide clarity and direction.
-
Regular reviews are essential to keep your agreements relevant and legally sound.
What are some examples of Quality Medical Devices Agreements?
-
Comparing various agreements helps in understanding diverse structuring and specific needs.
-
Insight into how the most effective agreements are designed can guide one’s drafting.
-
Integrating proven practices from leading companies enhances quality in your agreements.
How does regulatory framework affect Quality Medical Devices Agreements?
-
ISO 13485 and FDA regulations set the quality benchmarks that all manufacturers must comply with.
-
Be aware of local laws and regulations in your operating region that pertain to medical devices.
-
Strict adherence to standards is paramount for market acceptance and customer safety.
What are post-agreement procedures?
-
Understand what both parties are expected to fulfill after the agreement is signed.
-
Know how to properly make adjustments to the agreement as needed.
-
Prioritize clear communication channels and established processes for managing conflicts.
How to fill out the Quality Medical Devices Agreement Template
-
1.To access the Quality Medical Devices Agreement on pdfFiller, navigate to the given URL or search for this form in the search bar on the homepage of pdfFiller.
-
2.Once on the form, use the intuitive interface to view the document and the available fields that need completion. Familiarize yourself with the layout and headings.
-
3.Before beginning to fill out the form, gather necessary information such as the agreement date, manufacturer and distributor names, and product details.
-
4.Start filling in the blank fields, such as 'Enter Agreement Date: [MM/DD/YYYY]'. Click into each field to type or make selections as required. For example, enter the names of the Manufacturer and Distributor.
-
5.Follow the instructions provided within the document carefully, particularly for sections such as responsibilities and quality assurance measures. Ensure to fill this out accurately to reflect the agreement between both parties.
-
6.Double-check all entered information for accuracy. Review the filled sections for completion and clarity, ensuring that all required fields are adequately addressed.
-
7.Once finalized, you can save your document directly on pdfFiller by clicking the save icon or selecting ‘Download’ to obtain a copy of your completed form.
-
8.To submit the agreement, follow any specific submission method outlined in the document, or use the email function in pdfFiller to send it directly to the relevant parties.
Who is eligible to complete the Quality Medical Devices Agreement?
Both manufacturers and distributors of medical devices can use this agreement. It is crucial that all parties involved are legally recognized entities capable of entering a binding contract.
Is there a deadline for completing this agreement?
While this form does not specify a deadline, it is advisable to complete it promptly to align with business operations and comply with regulatory standards in medical device distribution.
How can I submit the Quality Medical Devices Agreement once completed?
The completed agreement can be submitted directly through email or as per the specific methods indicated within the document. Ensure all parties sign before submission to validate the contract.
What supporting documents are required with this agreement?
Typically, additional documents may include identification of both parties, business licenses, quality assurance certifications, and any specific compliance documentation related to medical device regulations.
What are common mistakes to avoid when filling out this form?
Common mistakes include leaving fields blank, not providing clear specifications for medical devices, and neglecting to review each party's responsibilities. Ensure all areas are accurately completed before finalizing.
How long will it take to process the Quality Medical Devices Agreement?
Processing time varies based on the parties involved. Once all signatures are obtained, the agreement takes effect, but internal review processes may introduce additional time, plan accordingly.
Are there any fees associated with using the Quality Medical Devices Agreement?
This agreement itself does not incur fees; however, depending on the platform used for signing or filing, there may be administrative or service charges. Check pdfFiller's pricing for details.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.