Last updated on Apr 10, 2026
Get the free Polish Adverse Event Reporting Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is polish adverse event reporting
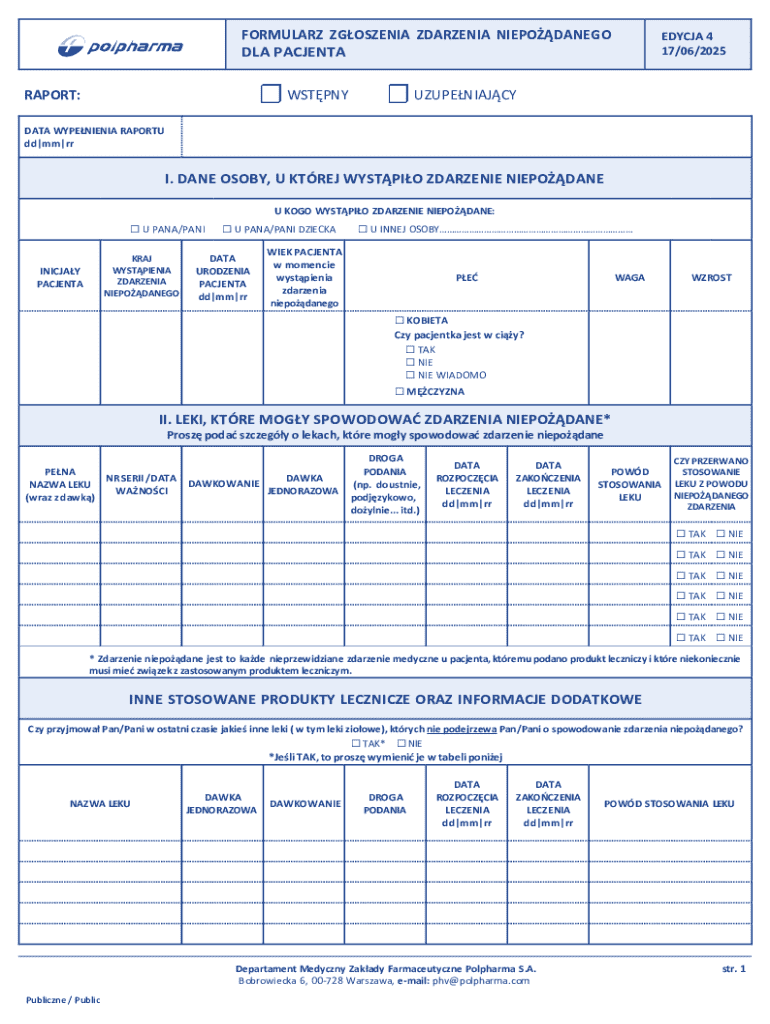
The Polish Adverse Event Reporting Form is a healthcare document used by patients and healthcare professionals to report unexpected medical events related to pharmaceutical products in Poland.
pdfFiller scores top ratings on review platforms




Who needs polish adverse event reporting?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to polish adverse event reporting
What is the Polish Adverse Event Reporting Form?
The Polish Adverse Event Reporting Form is an essential tool for health and safety reporting, designed to collect detailed information about adverse events that occur in medical settings. Adverse events refer to incidents that result in harm to patients, and their implications can significantly affect overall health outcomes and patient safety. This form plays a critical role in ensuring accountability within healthcare practices, allowing for systematic reporting and analysis to improve patient care.
Purpose and Benefits of the Polish Adverse Event Reporting Form
The primary objectives of the Polish Adverse Event Reporting Form revolve around enhancing patient safety and monitoring health outcomes effectively. By utilizing the form, healthcare providers, patients, and regulatory bodies can benefit in various ways:
-
Improves the identification and resolution of adverse events.
-
Ensures that healthcare practices align with safety regulations.
-
Facilitates data collection for improved analysis and reporting.
Moreover, the form streamlines the adverse event reporting process, making it more efficient for all parties involved.
Eligibility Criteria for the Polish Adverse Event Reporting Form
The Polish Adverse Event Reporting Form is intended for use by various stakeholders in the healthcare system, including:
-
Healthcare providers who witness or manage adverse events.
-
Patients who experience unexpected medical events.
Specific situations warrant filing a report, particularly when an adverse event occurs that may be linked to a medicinal product. Additionally, there may be restrictions based on the location of the incident or the type of adverse event being reported.
How to Fill Out the Polish Adverse Event Reporting Form Online (Step-by-Step)
To fill out the Polish Adverse Event Reporting Form online, follow these detailed instructions:
-
Access the form through pdfFiller.
-
Locate the key sections that must be completed.
-
Fill in all required fields with accurate information.
-
Utilize the editing features on pdfFiller to correct any mistakes.
-
Review the completed form for thoroughness before submission.
pdfFiller offers digital features that enhance the filling out process, making it user-friendly and efficient.
Common Errors and How to Avoid Them in the Polish Adverse Event Reporting Form
When completing the Polish Adverse Event Reporting Form, users often make several common mistakes that can lead to delays or inaccuracies:
-
Omitting required fields or providing insufficient details.
-
Failing to review the information before submission.
To avoid these errors, it's essential to provide detailed and accurate information and review the completed form thoroughly prior to submitting it.
Submission Methods and Delivery for the Polish Adverse Event Reporting Form
Once you have completed the Polish Adverse Event Reporting Form, several submission methods are available:
-
Online submission directly through pdfFiller.
-
Mailing a hard copy to the appropriate regulatory agency.
It is important to be aware of any timelines for submission as well as potential fees that may apply. After submitting, you can track the status of your submission through the designated agency.
What to Expect After Submitting the Polish Adverse Event Reporting Form
After submitting the Polish Adverse Event Reporting Form, users can expect certain follow-up actions:
-
Receipt of a confirmation notice acknowledging the report.
-
Possible communication from the agency for additional information or clarification.
If amendments or corrections to the submitted details are necessary, guidance will typically be provided on how to proceed.
Security and Compliance When Using the Polish Adverse Event Reporting Form
When utilizing the Polish Adverse Event Reporting Form via pdfFiller, users can be assured of data security and privacy. The platform complies with relevant regulations, including HIPAA and GDPR, ensuring that personal information remains protected. Furthermore, pdfFiller employs robust encryption measures, enhancing secure document handling processes.
How pdfFiller Can Enhance Your Experience with the Polish Adverse Event Reporting Form
Using pdfFiller to complete the Polish Adverse Event Reporting Form offers various advantages:
-
Easy editing capabilities to ensure accuracy.
-
eSigning options that streamline the submission process.
-
Intuitive design suitable for both novice and experienced users.
These features contribute to a user-friendly experience while ensuring that the form is completed efficiently.
Ready to Get Started on Your Polish Adverse Event Reporting Form?
Begin your process by utilizing pdfFiller, which enables users to fill out the form seamlessly. The platform's features promote an efficient and secure experience, making it easy for anyone to manage this important healthcare documentation.
How to fill out the polish adverse event reporting
-
1.Access the Polish Adverse Event Reporting Form on pdfFiller's website by searching for the form name or navigating directly to the healthcare forms section.
-
2.Once you locate the form, click on it to open it in the pdfFiller interface where you will see a preview of the document.
-
3.Before filling out the form, gather all necessary information such as patient details, specific medical event occurrences, and medication names involved in the incident.
-
4.Use your mouse or keyboard to navigate through the fillable fields on the form. Click or tap on each field to enter the required information accurately, following the prompts provided.
-
5.For sections that require checkboxes or multiple choice answers, simply click the relevant options to mark your choices.
-
6.Carefully review all the filled fields for accuracy and completeness to ensure the information provided is correct before finalizing the document.
-
7.Once you are satisfied with your entries, proceed to save your work using the save option, ensuring your data is not lost.
-
8.You can then download the completed form to your device or directly submit it through pdfFiller if submission options are enabled.
-
9.Keep a copy for your records after downloading or submitting to track the reporting process.

Who is eligible to use the Polish Adverse Event Reporting Form?
Any healthcare professional or patient who has experienced or witnessed an unexpected medical event related to pharmaceutical products in Poland is eligible to use the form for reporting.
Is there a deadline for submitting an adverse event report?
While there is no strict deadline stated for the Polish Adverse Event Reporting Form, it is recommended to submit reports as soon as possible to ensure timely monitoring and safety evaluations.
How should the completed form be submitted?
The completed form can be submitted through pdfFiller's submission option or printed and mailed directly to the medical department of Polpharma S.A. for proper processing.
What supporting documents are required with the form?
Generally, no additional supporting documents are needed when submitting the Polish Adverse Event Reporting Form. However, detailed information about the incident may enhance the report.
What are common mistakes to avoid when completing the form?
Ensure that all fields are filled accurately and completely. A common mistake is providing incomplete patient information or missing details about the medical event and medications involved.
How long does it take to process an adverse event report?
The processing time for adverse event reports can vary based on the complexity of the report and the procedures of the receiving organization, but expect at least a few weeks for initial reviews.
What should I do if I encounter issues while filling out the form on pdfFiller?
If you face any issues while filling out the form, refer to pdfFiller’s help or support section for troubleshooting guidance, or contact their customer service for immediate assistance.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.






















