Last updated on Mar 29, 2026
Get the free Sample Box Location Log Sheet
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is sample box location log
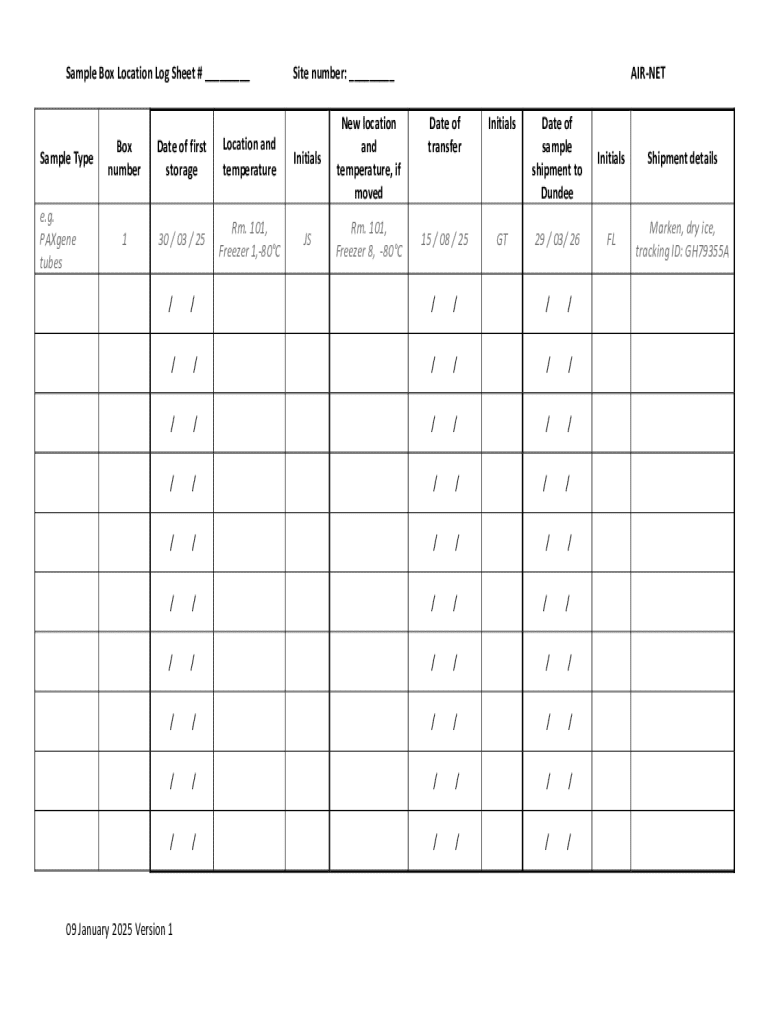
The Sample Box Location Log Sheet is a document used by sample handlers to track the location, storage conditions, and transfer details of biological samples.
pdfFiller scores top ratings on review platforms




Who needs sample box location log?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to sample box location log
What is the Sample Box Location Log Sheet?
The Sample Box Location Log Sheet is a vital form used for the effective tracking and management of biological samples. This form ensures that every sample is accounted for throughout its lifecycle, contributing significantly to sample handling processes. It serves as a comprehensive tool for documenting the storage and transfer of biological samples, thus enhancing laboratory efficiency.
This log sheet plays a crucial role in biological sample tracking and helps maintain organized records that are essential in various healthcare and research settings. By utilizing the sample box location log sheet, professionals can ensure compliance and enhance overall operational effectiveness.
Purpose and Benefits of Using the Sample Box Location Log Sheet
The primary purpose of the Sample Box Location Log Sheet is to maintain accurate records of sample storage and handling, which is critically important in laboratory settings. By documenting sample locations, users can significantly enhance safety and compliance, thereby mitigating risks associated with sample mismanagement.
Implementing this log sheet offers numerous benefits, such as facilitating streamlined sample management processes. It allows laboratory personnel to quickly access sample information, track movement, and ensure that protocols are followed in accordance with regulations, thereby benefiting healthcare professionals and researchers alike.
Key Features of the Sample Box Location Log Sheet
The Sample Box Location Log Sheet showcases various essential features that aid in sample management. Each log sheet includes detailed fields that capture important information such as sample type, box number, and specific storage conditions. This ensures all necessary data is documented clearly.
Additionally, the log sheet emphasizes the importance of initials and dates, which serve as accountability markers. By recording these details, users can ensure thorough tracking of all biological samples, thereby supporting integrity throughout the storage process.
Who Needs the Sample Box Location Log Sheet?
This log sheet is beneficial for a wide range of professionals operating in healthcare, research, and laboratory environments. Key roles such as Sample Handlers and Lab Technicians require efficient sample tracking to uphold safety and quality standards in their operations.
Given its importance, any individual involved in the management of biological samples should utilize this form. It aids in maintaining structured documentation necessary for compliance and operational success in laboratories and medical settings.
How to Fill Out the Sample Box Location Log Sheet Online
Filling out the Sample Box Location Log Sheet online can be streamlined into a few straightforward steps. Begin by accessing the form in your preferred PDF editor, such as pdfFiller, which allows for easy online completion without software downloads.
-
Enter the sample type and box number in their respective fields.
-
Input the date of first storage along with the specific location where the sample is kept.
-
Add any shipment details necessary for tracking.
-
Ensure to initial and date your entry for verification.
Accurate data entry is paramount to prevent errors and ensure compliance during handling.
Security and Compliance for the Sample Box Location Log Sheet
User privacy and data security are paramount when it comes to handling biological sample data. The Sample Box Location Log Sheet incorporates stringent security measures, including data encryption, to protect sensitive information.
This form also complies with regulations such as HIPAA and GDPR, ensuring that all personal and medical data is handled securely. By emphasizing these security features, users can trust that their records are managed with the highest standards of confidentiality and integrity.
How to Download and Share the Sample Box Location Log Sheet
Accessing and sharing the Sample Box Location Log Sheet is straightforward, especially when utilizing pdfFiller. To download the log sheet, simply select the download option to retrieve it in PDF format.
For sharing completed forms, users have various options to distribute the log sheets securely, ensuring that sensitive information is protected throughout the transfer process.
What Happens After You Submit the Sample Box Location Log Sheet?
Following the submission of the Sample Box Location Log Sheet, users will receive confirmation of their form submission. It is crucial to keep track of these records for verification and compliance purposes.
Additionally, users should be aware of the duration and procedures involved after submission to ensure continued oversight and accountability in handling samples.
Common Errors and How to Avoid Them
Common mistakes often occur when completing the Sample Box Location Log Sheet, potentially leading to issues in sample management. Some typical errors include neglecting to fill out required fields or providing incomplete information.
To enhance accuracy and compliance, users should double-check their entries and follow guidelines meticulously. Understanding common pitfalls associated with this biological sample tracking form enables users to prevent errors effectively.
Your Solution for Completing the Sample Box Location Log Sheet
For a seamless experience in completing the Sample Box Location Log Sheet, consider using pdfFiller. This platform offers cloud-based editing capabilities, allowing users to easily fill and sign the log sheet without any necessary downloads.
By leveraging pdfFiller's intuitive features, including eSigning and convenient sharing options, users can effectively manage their sample tracking needs with confidence and ease.
How to fill out the sample box location log
-
1.To access and open the Sample Box Location Log Sheet on pdfFiller, first visit the pdfFiller website and locate the search bar. Enter 'Sample Box Location Log Sheet' in the search field to find the correct form. Once located, click on the form to open it.
-
2.Navigating through pdfFiller is user-friendly. Once the form is open, you will see various fields such as sample type, box number, and date of first storage. Click on each field to fill in details. You can use the tab key to move efficiently between fields.
-
3.Before starting to fill out the form, gather all necessary information. This includes the sample type, the corresponding box number, date of first storage, and location details. Prepare any additional information regarding shipment details and initials for documentation.
-
4.After entering all the required information, review the details you have filled in. Ensure accuracy for each field in the Sample Box Location Log Sheet to prevent errors. Consider checking for completeness and consistency in your entries.
-
5.Once satisfied with the information entered, you can save the form on pdfFiller. Click on the save button located at the top right of the interface. Choose to download the completed form as a PDF or submit it directly through pdfFiller to your intended recipient.

Who is eligible to use the Sample Box Location Log Sheet?
The Sample Box Location Log Sheet can be used by anyone involved in the handling, tracking, and storage of biological samples, including laboratory technicians, healthcare professionals, and clinical researchers.
Are there deadlines for submitting this form?
There are generally no specific overall deadlines for the Sample Box Location Log Sheet. However, timely completion is essential to maintain accurate tracking of biological samples. Consult with your laboratory or institution for any specific timelines that may apply.
How can I submit the Sample Box Location Log Sheet?
You can submit the Sample Box Location Log Sheet either electronically through pdfFiller or print it out to mail or fax to the relevant department. Ensure you follow your institution's submission protocols.
What supporting documents do I need with this form?
Typically, no additional supporting documents are required with the Sample Box Location Log Sheet. However, depending on your organization, you may need to attach any relevant storage or transfer documentation to ensure compliance.
What common mistakes should I avoid when filling out this form?
Common mistakes include omitting required fields, entering incorrect sample details, and not double-checking for accuracy. Make sure all fields are filled out legibly and entirely before submission.
How long does it take to process the submitted Sample Box Location Log Sheet?
Processing times can vary based on your organization’s policies. Generally, if submitted electronically, it can be reviewed within a few business days. Check with your institution for specific processing timelines.
Is notarization required for this form?
No, notarization is not required for the Sample Box Location Log Sheet. This form serves as an internal tracking document for biological samples, so signatures from authorized personnel suffice.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.





















