Last updated on Apr 2, 2026
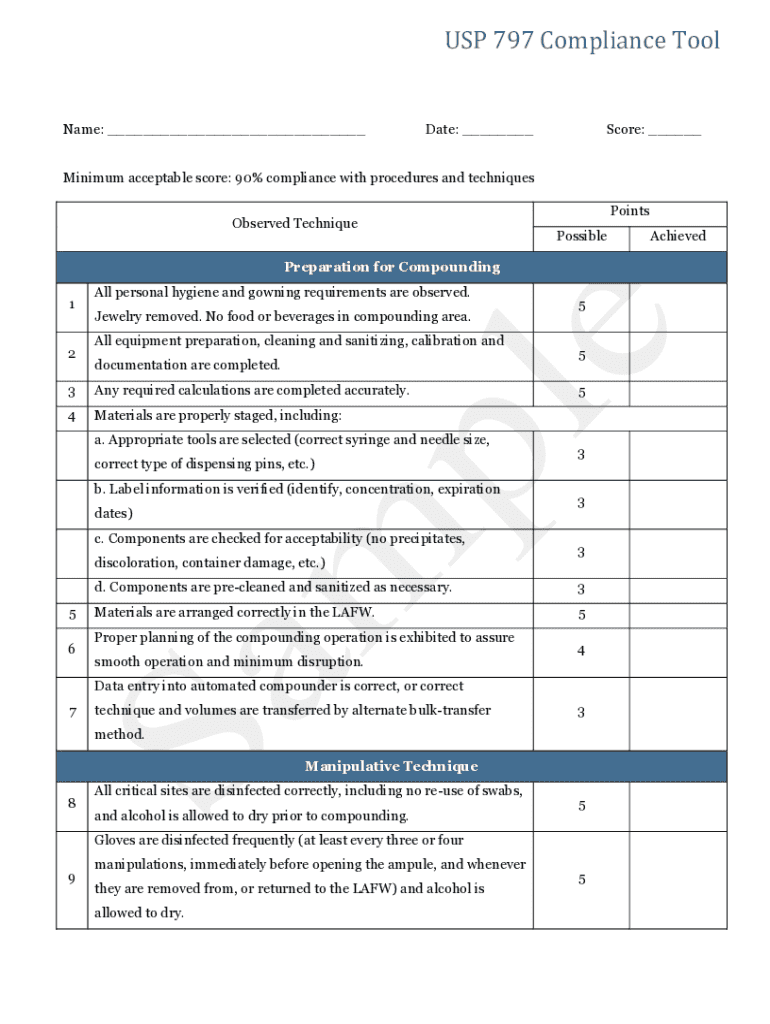
Get the free USP 797 Compliance Tool
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is usp 797 compliance tool
The USP 797 Compliance Tool is a digital document used by healthcare facilities to ensure compliance with sterile compounding standards established by the United States Pharmacopeia.
pdfFiller scores top ratings on review platforms




Who needs usp 797 compliance tool?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to usp 797 compliance tool
What is the USP 797 Compliance Tool?
The USP 797 Compliance Tool serves a crucial role in ensuring that pharmaceutical practices adhere to the standards set for sterile compounding. By complying with USP 797 requirements, healthcare providers can minimize the risk of contamination, ensuring patient safety. This tool facilitates the management of necessary documentation efficiently, particularly through the support of pdfFiller, which streamlines the process of filling and tracking compliance documents.
Purpose and Benefits of the USP 797 Compliance Tool
Utilizing the USP 797 Compliance Tool is essential for pharmacies and healthcare institutions to comply with stringent safety regulations. This tool offers numerous benefits, including improved patient safety, reduced risk of errors, and enhanced operational efficiency. Non-compliance can lead to severe risks, such as fines or legal issues; thus, leveraging this tool mitigates such consequences effectively.
Key Features of the USP 797 Compliance Tool
-
User-friendly interface that simplifies navigation and form-filling.
-
Cloud-based accessibility allows users to access documents from any device.
-
Collaboration features enable multiple users to work on compliance documents simultaneously.
-
Built-in templates for various forms related to USP 797 compliance.
-
Security features to protect sensitive information with 256-bit encryption.
Who Needs the USP 797 Compliance Tool?
The primary users of the USP 797 Compliance Tool include pharmacy professionals such as pharmacists and pharmacy technicians, along with healthcare administrators. These roles are integral to ensuring compliance with USP 797 regulations, as they directly handle sterile compounding processes. Organizations involved in pharmaceutical care, including hospitals and outpatient clinics, should implement this compliance tool to promote safety and adhere to guidelines.
How to Fill Out the USP 797 Compliance Tool Online (Step-by-Step)
-
Log in to your pdfFiller account and locate the USP 797 Compliance Tool.
-
Review the instructions provided at the top of the form to understand the requirements.
-
Fill in the required fields, ensuring accuracy in all entries.
-
Use the built-in help features for assistance with any complex sections.
-
Save your progress frequently to avoid losing any information.
-
Once completed, review the form carefully before submitting.
Common Errors and How to Avoid Them
Some users may encounter common errors when completing the USP 797 Compliance Tool, such as incorrect data entry or overlooking required fields. To avoid these pitfalls, users should follow practical strategies, including reviewing each section thoroughly as they proceed. A comprehensive review checklist can help ensure that all necessary information is captured before submission.
Where and How to Submit the USP 797 Compliance Tool
Submitting the USP 797 Compliance Tool involves specific processes depending on your institution's requirements. This may include electronic submission via a designated portal or mailing a physical copy to the appropriate regulatory body. Be mindful of any deadlines and fees associated with the submission process, ensuring that all documentation arrives on time to avoid compliance issues.
Tracking Your Submission and What Happens Next
After submitting the USP 797 Compliance Tool, users can track their submission status through the designated submission platform. Expect typical processing times, which can vary, and stay alert for any communications regarding the status of your compliance document. In the event of issues or required follow-ups, be prepared to address them promptly to ensure ongoing compliance.
Maintaining Compliance: Security and Record Retention
Document security is paramount when handling the USP 797 Compliance Tool. Proper record retention practices are required for compliance documentation, ensuring that records are securely stored for the necessary duration. pdfFiller enhances this process through its advanced security features, safeguarding sensitive information against unauthorized access.
Getting Started with pdfFiller for Your USP 797 Compliance Tool Needs
pdfFiller simplifies the completion of the USP 797 Compliance Tool, making the process efficient and user-friendly. By leveraging the platform's features, users can enjoy a seamless form-filling experience. For additional support, resources are available on pdfFiller's website to assist users in navigating their compliance documentation effectively.
How to fill out the usp 797 compliance tool
-
1.Access the USP 797 Compliance Tool on pdfFiller by searching for the form in the provided search bar or selecting it from the categories listed.
-
2.Once opened, familiarize yourself with pdfFiller's interface, which consists of the document viewing area and editing toolbars on the side.
-
3.Before starting, gather necessary information such as sterile compounding procedures, facility layouts, and current compliance standards to fill out the form accurately.
-
4.Begin filling out the form by clicking on the fields which will prompt text boxes for you to enter required data such as contact information and compliance measures.
-
5.Utilize features like text fields, checkboxes, or dropdown menus to complete sections accordingly, ensuring all information is correct and thorough.
-
6.Review the filled form in pdfFiller thoroughly. Use the preview mode to ensure that everything appears as desired and double-check all entries for accuracy.
-
7.Once finalized, save your document by selecting the appropriate option in pdfFiller. You may also choose to download a copy or submit it directly if needed.

Who is eligible to use the USP 797 Compliance Tool?
Any pharmacy, healthcare provider, or facility involved in sterile compounding can use the USP 797 Compliance Tool, particularly those subject to regulatory standards.
What are the deadlines for filing the compliance information?
Deadlines may vary based on local regulations or internal policies regarding compliance. It is recommended to check with your regulatory authority for specific requirements.
How do I submit the completed USP 797 Compliance Tool?
The completed form can typically be submitted via email to your regulatory authority or uploaded directly through institutional portals as per local guidelines.
What supporting documents are required with the USP 797 Compliance Tool?
Supporting documents may include protocol manuals, staff training records, and facility layouts. Check with your regulatory authority for specific needs.
What common mistakes should I avoid when filling this form?
Ensure all contact information is correct, double-check compliance measures, and avoid leaving fields blank unless specified. Review all entries before finalization.
How long does it take to process the compliance submissions?
Processing times can differ by jurisdiction; typically, it may take anywhere from a few days to several weeks depending on the volume of submissions.
Are there any fees associated with submitting the USP 797 Compliance Tool?
Fees may depend on the regulatory body's policies. Some may charge for processing or inspections, so confirm with your authority for precise fee structures.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.