Last updated on Apr 4, 2026
Get the free Controlled Substance Inventory Annual Summary Report
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is controlled substance inventory annual
The Controlled Substance Inventory Annual is a report used by healthcare facilities to document and verify the quantities of controlled substances in their possession annually.
pdfFiller scores top ratings on review platforms




Who needs controlled substance inventory annual?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to controlled substance inventory annual
What is the Controlled Substance Inventory Annual?
The Controlled Substance Inventory Annual form is a critical document that health care entities must complete to maintain compliance with regulations governing controlled substances. This form serves as a comprehensive record of all controlled substances held by the entity, making it essential for accurate inventory management.
Maintaining an accurate inventory is vital for compliance with various federal and state regulations. Non-compliance can lead to severe penalties, including fines and legal repercussions, which underscores the necessity of diligent inventory practices.
Entities managing controlled substances are required to understand and adhere to applicable regulations to avoid potential violations and ensure proper handling of these sensitive materials.
Purpose and Benefits of the Controlled Substance Inventory Annual
The Controlled Substance Inventory Annual serves multiple essential purposes. First and foremost, it ensures compliance with both federal and state regulations regarding controlled substances. By accurately maintaining records, organizations can effectively prevent misuse and diversion of these substances.
Additionally, proper inventory management facilitates smoother audits and inspections by regulatory authorities, streamlining the verification process. This not only enhances operational efficiency but also builds trust with regulatory bodies.
Who Needs the Controlled Substance Inventory Annual?
Entities such as pharmacies, hospitals, and other healthcare facilities are required to complete the Controlled Substance Inventory Annual form. These organizations hold responsibility for maintaining compliance with regulations and ensuring the safe handling of controlled substances.
Individual roles may vary, with designated staff members tasked with oversight of the inventory process. Special considerations must be made for different types of facilities, as the specific requirements may differ based on the nature and volume of controlled substances handled by each entity.
When and How to File the Controlled Substance Inventory Annual
Filing deadlines for the Controlled Substance Inventory Annual are crucial to ensure compliance. Typically, entities are required to submit the form annually, with key deadlines established by regulatory authorities.
-
Prepare necessary documentation and data related to controlled substances.
-
Utilize pdfFiller to fill out the form online, ensuring all information is accurate.
-
Submit the form through the preferred method: online, via mail, or in-person.
Understanding these deadlines and submission methods is critical to avoid potential penalties for late filing.
How to Complete the Controlled Substance Inventory Annual Online
Completing the Controlled Substance Inventory Annual online via pdfFiller is straightforward. Users should begin by familiarizing themselves with the sections within the form, ensuring they have all necessary information ready.
Organizing data prior to filling out the form is essential. Tips include preparing inventory lists and ensuring all figures are accurate. Following the instructions on pdfFiller will help users navigate the form efficiently, resulting in a smooth completion process.
Common Errors and How to Avoid Them
When filling out the Controlled Substance Inventory Annual form, individuals often encounter common errors. Mistakes may range from inaccurate data entry to incomplete sections, which can lead to compliance issues.
To avoid these pitfalls, it is recommended to double-check entries before submission. Implementing a review process for all data ensures accuracy and helps maintain compliance with regulatory standards.
Security and Compliance for the Controlled Substance Inventory Annual
Utilizing pdfFiller to manage the Controlled Substance Inventory Annual ensures document security through robust features. The platform incorporates 256-bit encryption, safeguarding sensitive information contained within the form.
Moreover, pdfFiller is compliant with relevant laws, including HIPAA and GDPR, providing peace of mind when handling sensitive data. Understanding these security measures is crucial for organizations that must protect patient and operational information.
Tracking Your Submission and What Happens Next
Once the Controlled Substance Inventory Annual form has been submitted, it is essential for entities to track the submission process. Confirming that the submission has been received by the regulatory body is the first step in ensuring compliance.
Organizations should also be aware of what to expect after submission, such as processing timelines and potential responses from regulatory authorities. Should any issues arise with the submission, it is important to have a clear procedure for addressing these concerns promptly.
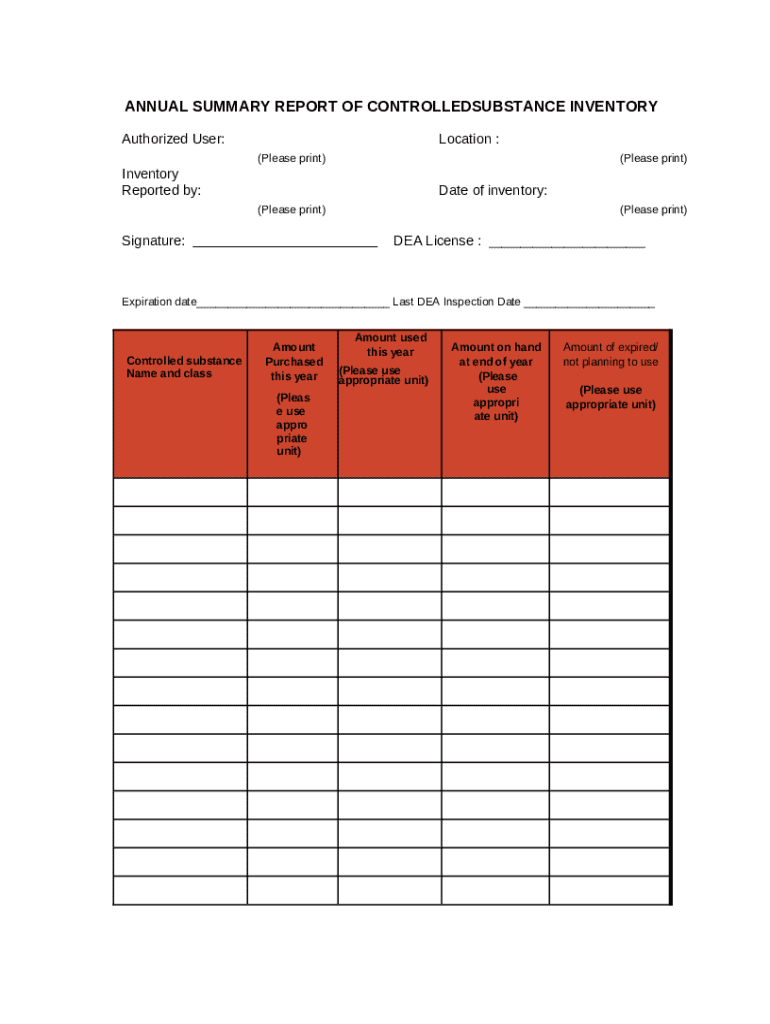
Sample Controlled Substance Inventory Annual Form
For clarity and guidance, having access to a sample Controlled Substance Inventory Annual form can be invaluable. This example highlights key fields and necessary data entry points, serving as a reference for users.
Utilizing a completed form as a guide can help minimize errors and streamline the filling process, ensuring that users understand the expectations and requirements associated with the inventory form.
Effortless Filing with pdfFiller
pdfFiller offers an enhanced filing experience for the Controlled Substance Inventory Annual. The platform is designed to support users through features such as eSigning, document sharing, and efficient editing.
Additionally, pdfFiller provides extensive user support and resources, empowering entities to navigate the form-filling process with confidence. Leveraging such tools can significantly enhance compliance and operational efficiency.
How to fill out the controlled substance inventory annual
-
1.Begin by logging into your pdfFiller account. If you do not have an account, create one to gain access.
-
2.Once signed in, use the search bar to locate the 'Controlled Substance Inventory Annual' form. Click on the form to open it.
-
3.Review the form fields carefully. Familiarize yourself with the required information such as drug names, quantities, and storage locations.
-
4.Gather necessary documentation, including previous inventory reports and purchase records, to ensure accurate completion of the form.
-
5.In pdfFiller's editing interface, click on the fields to enter the required data. Type in the current quantities of each controlled substance you have on hand.
-
6.If applicable, use the dropdowns and selection boxes to choose the correct options pertaining to each substance's handling and storage.
-
7.Double-check all entries for accuracy and completeness. It's essential to ensure that no information is missing to avoid processing delays.
-
8.After ensuring everything is filled out correctly, scroll to the bottom of the form where you can review all your entries.
-
9.Once verified, save your form. You can choose to download it directly to your device or use pdfFiller’s sharing features to send it through email.
-
10.If submitting electronically, follow the prompts for submission via pdfFiller. Otherwise, print the form for your records and manual submission if needed.

Who is required to complete the Controlled Substance Inventory Annual?
Healthcare facilities that handle controlled substances must complete this inventory form annually to maintain regulatory compliance and ensure accurate tracking of these medications.
What is the deadline for submitting the inventory report?
The specific deadline for submitting the Controlled Substance Inventory Annual varies by state regulations. Generally, it's due at the end of your inventory year or as specified by your local regulatory body.
How do I submit the Controlled Substance Inventory Annual form?
You can submit the form either electronically using pdfFiller or by printing it and sending it directly to the designated regulatory authority via mail or fax, depending on local laws.
Are there any supporting documents needed when filing this form?
While typically no additional documents are required, it is advisable to have previous inventory records and purchase receipts on hand to provide context and verification for the quantities reported.
What are common mistakes to avoid when completing this inventory form?
Common mistakes include providing incorrect quantities, overlooking missing substances, or failing to double-check entries. Ensure that all medications are accounted for and accurately reflected on the form.
How long does it take to process the inventory report once submitted?
Processing times for the Controlled Substance Inventory Annual can vary based on the regulatory agency. Generally, you can expect a response within a few weeks after submission.
Can I amend the Controlled Substance Inventory Annual after submission?
If you need to amend the form after submission, you should contact the regulatory body to learn about their amendment policy and specific procedures to correct any errors.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.