Last updated on Apr 25, 2026
Get the free IRB Amendment Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is irb amendment form
The IRB Amendment Form is a document used by researchers to request modifications to previously approved research protocols or studies.
pdfFiller scores top ratings on review platforms




Who needs irb amendment form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to irb amendment form
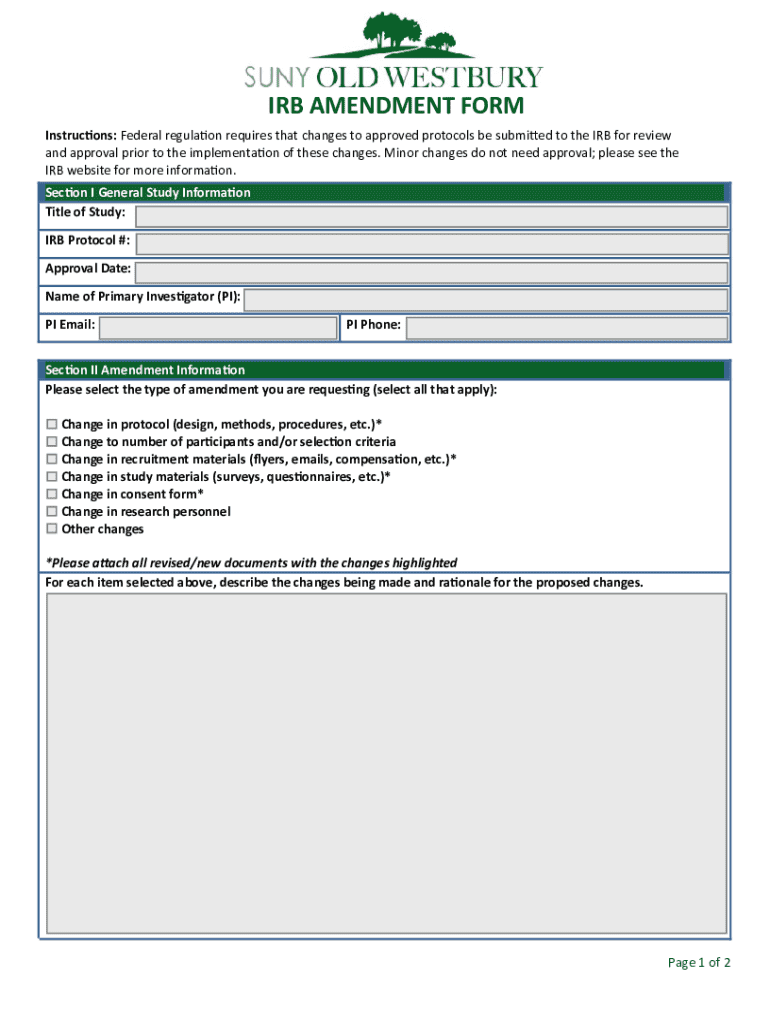
What is the IRB Amendment Form?
The IRB amendment form serves as a crucial document in the research process, facilitating necessary changes to an existing research protocol. Its role in research governance is vital as it maintains ethical and regulatory compliance, ensuring that any modifications, such as alterations in research design or participant information, are properly reviewed and approved.
Researchers must submit amendments to keep their studies aligned with the standards of Institutional Review Boards (IRBs), which oversee the ethical conduct of research involving human subjects. By doing so, researchers uphold the integrity of their work and protect participant rights, making the IRB amendment form indispensable in the research lifecycle.
Purpose and Benefits of the IRB Amendment Form
The IRB amendment form is designed to ensure compliance with ethical standards in research, serving not only as a record of changes but also as a mechanism to safeguard participant rights. By submitting this form, researchers can enhance the quality of their research and contribute to the overall integrity of scientific inquiry.
Failing to submit an amendment may lead to significant repercussions, including delays in research timelines or non-compliance with ethical guidelines. Understanding the benefits of the IRB amendment form is essential for researchers who aim for successful and responsible research outcomes.
Key Features of the IRB Amendment Form
When filling out the IRB amendment form, users will encounter several key components designed to facilitate thorough documentation of changes. Key sections include descriptions of amendments and any modifications in participant information, which are essential for IRB review.
Providing clear and concise information about changes not only aids the IRB in their review process but also accommodates diverse types of research, allowing for tailored adjustments to fit specific study needs.
Who Needs to Use the IRB Amendment Form?
The IRB amendment form is necessary for a variety of researchers and institutions involved in research projects that require oversight. Typically, academic institutions, hospitals, and independent research organizations are required to submit this form when changes to their approved protocols occur.
Eligibility criteria for using the form vary by institution, but it generally applies to researchers who have received prior IRB approval. Common scenarios necessitating an amendment include changes in study design, shifting of study locations, or alterations in participant demographics.
How to Fill Out the IRB Amendment Form Online: Step-by-Step Guide
Completing the IRB amendment form online through pdfFiller can be streamlined by following these steps:
-
Access the IRB amendment form via the pdfFiller platform.
-
Gather all necessary information regarding amendments and participant changes.
-
Carefully fill out each section of the form, ensuring clarity and accuracy.
-
Review the form to check for any errors or omissions.
-
Submit the completed form according to your institution’s guidelines.
By preparing your information beforehand and paying close attention to critical sections, you can enhance the accuracy of your submission.
Common Errors and How to Avoid Them
Various mistakes can occur during the completion of the IRB amendment form, hindering the submission process. Common errors include incomplete sections, unclear descriptions of changes, and overlooking required signatures.
To avoid these pitfalls, double-check your entries and ensure that all necessary information is included before submission. Taking the time to verify details can prevent delays in the review process and contribute to successful outcomes.
Submission Methods and Delivery Options for the IRB Amendment Form
Once the IRB amendment form is completed, it can be submitted through several methods depending on institutional policies. These options typically include online submissions through platforms like pdfFiller, mailing the form, or forwarding it via email.
Be aware of any deadlines associated with your submission to ensure timely processing. Understanding the specific requirements of your institution will streamline the submission process.
What Happens After You Submit the IRB Amendment Form?
After submitting the IRB amendment form, the review process begins, which may take varying amounts of time based on the nature of the changes. Researchers can typically track the status of their submissions through their institution’s IRB portal or contact for updates.
Possible outcomes following submission include approval of the amendment or requests for additional modifications, highlighting the importance of thorough and accurate information in the initial submission.
Secure Your IRB Amendment Form with pdfFiller
To enhance the security of your IRB amendment form, utilizing pdfFiller offers robust encryption protocols, ensuring that sensitive data remains protected. This cloud-based platform not only provides ease of access but also eliminates the need for installations, making it convenient for researchers.
With pdfFiller, users benefit from a comprehensive document management process that supports them in creating, editing, and submitting their forms securely, reinforcing the importance of safeguarding important research documents.
How to fill out the irb amendment form
-
1.To access the IRB Amendment Form on pdfFiller, visit the platform's homepage and search for the form using the search bar.
-
2.Once located, select the form to open it in the pdfFiller editor.
-
3.Before you start filling out the form, gather relevant information such as the original protocol, details of the proposed amendments, and any required supporting documents.
-
4.In the pdfFiller interface, click on each field to enter the necessary information, ensuring that you accurately describe the changes being proposed.
-
5.Utilize pdfFiller's tools to add comments, checkboxes, or additional text as required. Make sure to complete all mandatory fields to avoid submission issues.
-
6.After filling in all the required fields, take a moment to review your entries for accuracy and ensure all information is complete.
-
7.Use pdfFiller’s review features to double-check your form against the requirements before finalizing it.
-
8.Once everything is correct, choose to save a copy of your form. You can download it in your desired format or submit it directly through pdfFiller’s submission options.

Who is eligible to use the IRB Amendment Form?
The IRB Amendment Form is designed for researchers and students who hold approval for prior research studies involving human subjects and need to amend their protocols for compliance.
What is the deadline for submitting the IRB Amendment Form?
Deadlines for submission vary by institution, but it is advisable to submit your amendment as early as possible to allow time for review and potential adjustments.
How do I submit the completed IRB Amendment Form?
Completed forms can typically be submitted electronically via your institution's research compliance portal or printed and delivered to your IRB office, depending on their specific submission policies.
What supporting documents are needed with the IRB Amendment Form?
Supporting documents may include the original protocol, data collection instruments, and any changes to consent forms or recruitment materials. Check with your IRB for specific requirements.
What are common mistakes to avoid when completing the form?
Common mistakes include failing to complete all required fields, providing insufficient justification for changes, and not adhering to formatting guidelines set by the IRB.
How long does it take to process the IRB Amendment Form?
Processing times can vary widely, typically ranging from a week to several months, depending on the complexity of the amendment and the IRB's workload.
What should I do if my amendment request is denied?
If your amendment is denied, carefully review the feedback provided by the IRB. You may need to revise your proposal according to their suggestions and resubmit the form.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.






















