Last updated on Oct 22, 2015

Get the free Laboratory Consent Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Lab Consent
The Laboratory Consent Form is a medical consent document used by participants to provide voluntary consent for health profile and laboratory analysis conducted by Clinical Reference Laboratory and Healics Inc.
pdfFiller scores top ratings on review platforms




Who needs Lab Consent?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Lab Consent
What is the Laboratory Consent Form?
The Laboratory Consent Form is a critical document used in healthcare settings to obtain voluntary consent from participants before undergoing lab analysis. This form serves multiple purposes, including outlining the rights of participants and ensuring that they understand the procedures involved. It is primarily associated with institutions such as Clinical Reference Laboratory and Healics Inc. Consent plays a vital role in medical processes, reinforcing the ethical obligation of healthcare providers to inform and protect patients.
Purpose and Benefits of the Laboratory Consent Form
The Laboratory Consent Form is essential for protecting the rights of participants during lab analysis. By detailing the risks involved and the sample collection process, the form enables participants to make informed decisions regarding their health. Obtaining voluntary consent is not only a legal requirement but also fosters trust between participants and healthcare providers. The clarity provided by the form contributes to an ethical healthcare environment.
Key Features of the Laboratory Consent Form
This form includes several key features that enhance its usability and effectiveness. It contains fillable fields such as the Participant’s Signature, Date, and Examiner Name, allowing for clear documentation of consent. Optional sections include checkboxes for specific acknowledgments and initial fields where participants can indicate their understanding. Moreover, adherence to security regulations ensures that the form is compliant with healthcare standards.
Who Needs the Laboratory Consent Form?
The Laboratory Consent Form must be filled out by both participants and examiners involved in medical procedures. Typical scenarios needing this form include blood sample collection and other lab analyses where participant consent is paramount. Ensuring that participants fully understand the form is crucial for their engagement and compliance with the outlined procedures.
How to Fill Out the Laboratory Consent Form Online
Completing the Laboratory Consent Form online is a straightforward process. Follow these steps:
-
Access the form on pdfFiller's platform.
-
Carefully fill in all required fields, including the Participant’s Signature and Examiner Name.
-
Ensure all relevant checkboxes are marked accordingly, based on individual circumstances.
-
Review the form to avoid common errors that may delay processing.
-
Submit the form as instructed on the platform.
Security and Compliance for the Laboratory Consent Form
The safety and compliance of the Laboratory Consent Form are of utmost importance. It employs 256-bit encryption and aligns with HIPAA and GDPR regulations to protect sensitive participant data. Security measures implemented during the use of pdfFiller help safeguard health information, ensuring that participants' details are managed safely and responsibly.
What Happens After You Submit the Laboratory Consent Form?
Once the Laboratory Consent Form is submitted, participants can expect a streamlined process. Processing times may vary, and participants will receive notifications regarding the status of their consent. The pdfFiller platform also provides tracking mechanisms, allowing users to monitor their submissions closely. Understanding possible outcomes and next steps after submission is crucial for participant awareness.
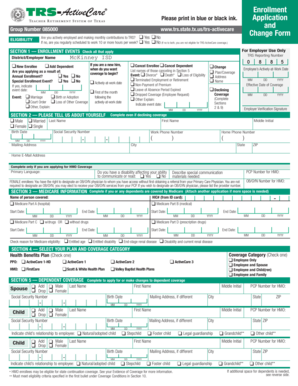
Sample or Example of a Completed Laboratory Consent Form
A visual reference can significantly aid users in understanding the Laboratory Consent Form. An example of a completed form illustrates the layout and key sections effectively. Essential areas of the form, such as the Participant’s Signature and Declaration of Understanding, can be annotated for clarity, emphasizing the significance of each component in the context of obtaining informed consent.
Your Partner in Completing the Laboratory Consent Form
pdfFiller is your go-to resource for completing the Laboratory Consent Form quickly and securely. The platform offers tools for editing and eSigning the form, ensuring access from any device without the need for downloads. Utilizing a cloud-based solution enhances convenience and fosters the accurate completion of the Laboratory Consent Form, promoting peace of mind for participants.
How to fill out the Lab Consent
-
1.Access pdfFiller in your web browser and log into your account or create a free account if you don't have one.
-
2.Use the search bar at the top of the page to locate the Laboratory Consent Form. You can also find it under Healthcare Forms in the appropriate category.
-
3.Open the form by clicking on it. Familiarize yourself with the layout and available fillable fields displayed within pdfFiller's interface.
-
4.Gather all necessary information before starting, such as the participant’s details, examiner's information, and specifics required for the blood sample collection process.
-
5.Fill in each field methodically. Start with the participant's name and address, then move on to the signature field. Ensure that all data is entered accurately.
-
6.Utilize the checkboxes within the form to indicate if the sample has been obtained and for the participant's initials where necessary.
-
7.Once all required fields are completed, review the form to ensure accuracy and completeness. Pay special attention to the signature and examiner details.
-
8.Finalize the form by saving your changes. You can download the completed form in various formats or submit it directly through pdfFiller, if applicable.
-
9.Follow any on-screen prompts to save or submit the form. It's recommended to keep a copy for your records.

Who is eligible to use the Laboratory Consent Form?
The Laboratory Consent Form is designed for individuals participating in health profile and lab analysis programs. Eligible users include adult participants and legal guardians for minors.
Is there a deadline for submitting this form?
While specific deadlines may vary depending on the testing program, participants are typically advised to submit the Laboratory Consent Form prior to their scheduled laboratory appointment.
How do I submit the Laboratory Consent Form once completed?
You can submit the form directly through pdfFiller if applicable. Alternatively, you can save it to your device and email or deliver it to the relevant healthcare provider.
What supporting documents do I need when completing this form?
Generally, you may need to provide identification, health insurance information, or other documents as specified by the healthcare provider overseeing the laboratory analysis.
What common mistakes should I avoid when filling out this form?
Common mistakes include missing required signatures, neglecting to initial checkboxes, or providing incorrect personal information. Always double-check your entries before submission.
How long does it take to process the Laboratory Consent Form?
Processing times will depend on the laboratory or healthcare provider. Typically, consent forms are processed quickly, but it’s wise to check for specific timelines directly with the organization.
What if I have concerns about the risks involved in the testing?
Participants are encouraged to discuss any concerns regarding risks with their healthcare provider prior to signing the Laboratory Consent Form to ensure they are fully informed.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.